Advanced Synthesis of Multi-Target Evodiamine Derivatives for Commercial Oncology Applications
The pharmaceutical landscape is continuously evolving towards multi-target therapeutic strategies to combat complex diseases like cancer, and patent CN108794474B represents a significant breakthrough in this domain by disclosing a novel class of evodiamine derivatives. These compounds are engineered to simultaneously inhibit Topoisomerase 1, Topoisomerase 2, and Histone Deacetylase, offering a robust mechanism to suppress tumor growth while minimizing the risks associated with traditional combination therapies. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediates supplier partners, understanding the structural versatility and synthetic accessibility of these molecules is crucial for integrating them into next-generation oncology pipelines. The patent details a comprehensive preparation method that ensures high purity and structural integrity, which are paramount for downstream drug development and regulatory approval processes in global markets.
This technical insight report analyzes the commercial and scientific viability of these evodiamine derivatives, focusing on their potential to streamline cost reduction in pharmaceutical intermediates manufacturing. By leveraging the specific synthetic routes outlined in the intellectual property, manufacturers can optimize production workflows to achieve substantial cost savings without compromising on the stringent quality standards required for active pharmaceutical ingredients. The following sections will dissect the mechanistic advantages, synthetic protocols, and supply chain benefits, providing a holistic view for decision-makers aiming to secure a competitive edge in the high-purity API intermediate sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional anticancer treatments often rely on single-target inhibitors or physical mixtures of multiple drugs, which frequently encounter significant hurdles such as the emergence of drug resistance and complex pharmacokinetic interactions. When separate agents are administered, the body metabolizes each compound differently, leading to unpredictable plasma concentrations and increased toxicity profiles that can limit the maximum tolerated dose. Furthermore, the need to confirm the rationality of drug compatibility in combination therapies adds layers of complexity to clinical trials, extending development timelines and inflating research budgets considerably. Single-target approaches also fail to address the heterogeneous nature of tumor cells, allowing resistant clones to survive and proliferate despite initial treatment success. These limitations underscore the urgent need for unified molecular entities that can engage multiple pathological pathways simultaneously without the logistical burden of polypharmacy.
The Novel Approach
The innovative strategy presented in patent CN108794474B overcomes these challenges by integrating multiple pharmacophores into a single evodiamine scaffold, creating a multi-target agent that simplifies the treatment regimen while enhancing therapeutic efficacy. This molecular design ensures that the absorption, distribution, metabolism, and excretion processes are uniform across all active components, thereby reducing the occurrence of adverse drug-drug interactions and improving patient compliance. By targeting Top1, Top2, and HDAC concurrently, the derivative induces synergistic anti-tumor effects that are greater than the sum of individual inhibitions, effectively blocking tumor cell cycles and inducing apoptosis more potently than single-agent therapies. This approach not only streamlines the clinical development pathway but also offers a more predictable safety profile, making it an attractive candidate for rapid translation from bench to bedside in the competitive oncology market.
Mechanistic Insights into Multi-Target Top1/Top2/HDAC Inhibition
The core innovation lies in the precise structural modification of the evodiamine parent nucleus, which serves as a potent scaffold for engaging distinct enzymatic targets within the cancer cell. The general structure, as defined in the patent, incorporates specific substituents at the R1 and R2 positions that modulate the electronic and steric properties of the molecule, optimizing its binding affinity for the active sites of Topoisomerases and HDAC enzymes. The inclusion of a hydroxamic acid moiety or similar zinc-binding groups in the side chain is critical for chelating the zinc ion in the HDAC active site, thereby preventing the deacetylation of histones and reactivating tumor suppressor genes. Simultaneously, the planar aromatic system of the evodiamine core facilitates intercalation with DNA, stabilizing the Top1-DNA and Top2-DNA cleavage complexes and preventing religation, which leads to lethal DNA double-strand breaks. This dual mechanism of epigenetic modulation and DNA damage induction creates a formidable barrier against tumor survival and proliferation.
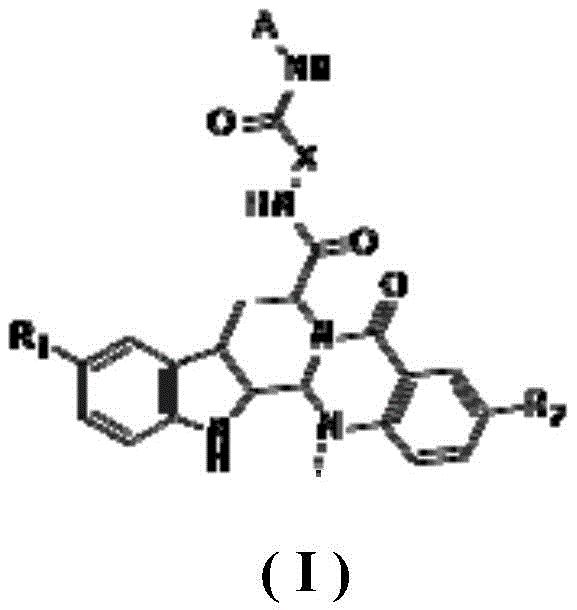
Impurity control is meticulously managed through the selection of high-purity starting materials and optimized reaction conditions that minimize side product formation. The synthetic route avoids the use of transition metal catalysts that often leave toxic residues, instead relying on organic coupling reagents that can be easily removed during workup and purification stages. This attention to detail ensures that the final crystalline products meet the rigorous specifications required for pharmaceutical applications, with well-defined polymorphic forms that guarantee stability and bioavailability. The patent data indicates that specific derivatives, such as compound 10c and compound 22, exhibit nanomolar IC50 values against HDAC1, surpassing the activity of standard controls like SAHA, which validates the effectiveness of the structural design in achieving high-purity pharmaceutical intermediates with superior biological performance.
How to Synthesize Evodiamine Derivative Efficiently
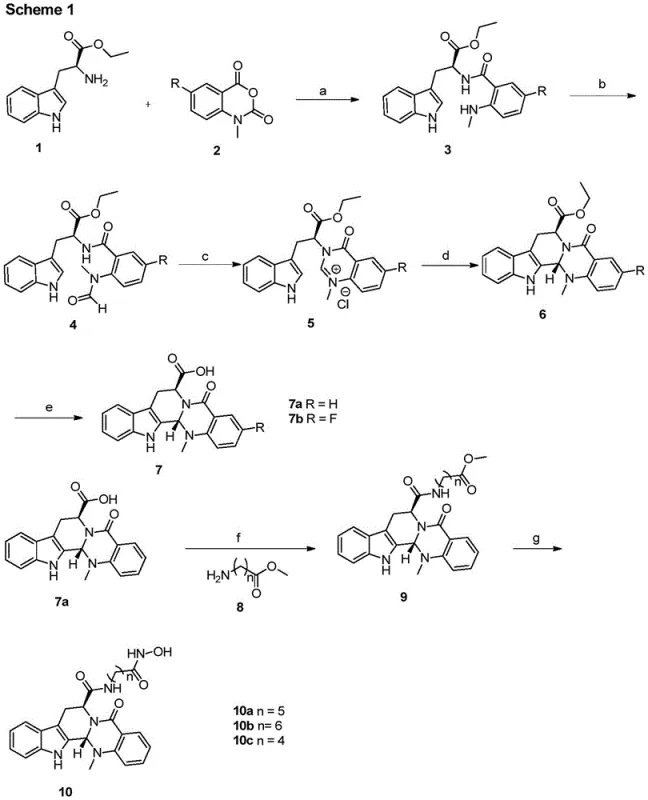
The synthesis of these high-value compounds follows a logical and scalable sequence that begins with the condensation of L-tryptophan ethyl ester hydrochloride and substituted N-methyl isatoic anhydride. This initial step forms the foundational indole-quinazoline skeleton, which is subsequently subjected to cyclization and decarboxylation to yield the key 7-carboxyl evodiamine intermediate. This intermediate serves as a versatile branching point where various side chains can be introduced to tune the biological activity and physicochemical properties of the final drug candidate. The detailed standardized synthesis steps see the guide below, which outlines the specific reagents and conditions necessary to achieve optimal yields and purity levels suitable for commercial production.
- Condense L-tryptophan ethyl ester hydrochloride with substituted N-methyl isatoic anhydride in a pyridine system to form the initial intermediate.
- Perform cyclization and decarboxylation reactions to generate the key 7-carboxyl evodiamine intermediate core structure.
- Execute condensation with amino methyl ester compounds using HATU/DIPEA followed by ammoniation with hydroxylamine to yield the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route described in the patent offers significant advantages in terms of raw material availability and process robustness, directly translating to enhanced supply chain reliability. The starting materials, such as L-tryptophan derivatives and isatoic anhydrides, are commodity chemicals that are readily accessible from multiple global suppliers, mitigating the risk of single-source bottlenecks that often plague specialty chemical manufacturing. This abundance ensures that production schedules can be maintained consistently, reducing lead time for high-purity pharmaceutical intermediates and allowing manufacturers to respond swiftly to fluctuating market demands. Furthermore, the avoidance of exotic or hazardous reagents simplifies the logistics of material handling and storage, contributing to a safer and more efficient operational environment.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts and complex purification steps associated with removing heavy metal residues, which significantly lowers the overall cost of goods sold. By utilizing standard peptide coupling reagents like HATU and DIPEA, the synthesis leverages well-established industrial protocols that are easy to implement and scale without requiring specialized equipment or extensive operator training. The high yields reported in the patent examples for key intermediates suggest that material throughput can be maximized, further driving down the unit cost and enabling substantial cost savings for downstream partners. This economic efficiency makes the technology particularly attractive for large-scale production where margin optimization is critical for competitiveness.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for flexible manufacturing strategies where different derivatives can be produced on the same production line with minimal changeover time. This flexibility ensures that supply continuity is maintained even if demand shifts between specific analogues, providing a buffer against market volatility. Additionally, the stability of the intermediates and final products under standard storage conditions reduces the risk of spoilage during transit and warehousing, ensuring that the quality of the commercial scale-up of complex pharmaceutical intermediates remains intact from the factory gate to the customer's facility. This reliability is a key factor for supply chain heads looking to de-risk their vendor portfolios.
- Scalability and Environmental Compliance: The reaction conditions described, such as refluxing in pyridine or stirring at room temperature in DMF, are inherently scalable from gram to kilogram and eventually to metric ton quantities without encountering significant exothermic or safety issues. The waste streams generated are primarily organic solvents that can be recovered and recycled using standard distillation technologies, aligning with modern green chemistry principles and environmental regulations. This compliance reduces the burden of waste disposal costs and facilitates smoother regulatory audits, ensuring that the manufacturing process remains sustainable and viable in the long term. The ability to scale diverse pathways from 100 kgs to 100 MT/annual commercial production is a testament to the industrial readiness of this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these evodiamine derivatives, based on the detailed disclosures within the patent documentation. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this technology into their existing portfolios and assess the potential return on investment. The answers are derived directly from the experimental data and claims provided in the intellectual property, ensuring accuracy and relevance for technical due diligence processes.
Q: What are the primary biological targets of these evodiamine derivatives?
A: The compounds described in patent CN108794474B are designed as triple-target inhibitors, simultaneously acting on Topoisomerase 1 (Top1), Topoisomerase 2 (Top2), and Histone Deacetylase (HDAC) to overcome drug resistance.
Q: Is the synthesis route scalable for commercial production?
A: Yes, the synthesis utilizes standard coupling reagents like HATU and DIPEA and starts from commercially available L-tryptophan derivatives, making it highly suitable for scale-up from kilograms to metric tons.
Q: How does the multi-target approach improve therapeutic efficacy?
A: By inhibiting multiple key links in the tumor disease network simultaneously, these derivatives exhibit synergistic anti-tumor effects and reduce the likelihood of drug-drug interactions compared to combination therapies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Evodiamine Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for complex oncology targets. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch of evodiamine derivative meets stringent purity specifications and complies with international regulatory standards. We understand the critical nature of time-to-market in the pharmaceutical industry and are committed to delivering high-quality intermediates that accelerate your drug development timeline while maintaining the highest levels of safety and efficacy. Our team of expert chemists is ready to collaborate on process optimization to further enhance yields and reduce environmental impact.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project requirements. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower you to make informed decisions about your supply chain strategy. Let us help you navigate the complexities of multi-target inhibitor synthesis and secure a reliable source of high-quality materials for your next breakthrough therapy. Reach out today to discuss how we can support your journey from discovery to commercialization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
