Advanced Synthesis of N-Substituted-3-(Benzenesulfonyl)Quinoxaline-2-Amines for Oncology Applications
The landscape of oncology drug discovery is constantly evolving, with a particular emphasis on small molecule inhibitors that target critical signaling pathways. Patent CN101602737B introduces a significant advancement in this field by disclosing a novel class of N-substituted-3-(benzenesulfonyl)quinoxaline-2-amine compounds. These heterocyclic structures represent a promising frontier in the development of next-generation antitumor therapeutics, specifically designed to inhibit key kinases involved in cell proliferation. The patent details a robust and scalable synthetic methodology that transforms simple, commercially available precursors into highly functionalized bioactive molecules. As a leading entity in fine chemical manufacturing, we recognize the immense potential of this technology to streamline the production of high-purity pharmaceutical intermediates. The structural versatility offered by this scaffold allows for extensive SAR (Structure-Activity Relationship) exploration, making it an invaluable asset for R&D teams aiming to optimize potency and selectivity against resistant tumor strains.
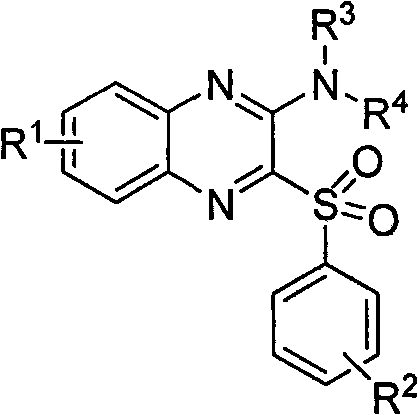
Furthermore, the preliminary pharmacological data associated with these compounds indicates exceptional efficacy, with certain derivatives exhibiting IC50 values in the low micromolar to nanomolar range against diverse cancer cell lines including prostate, lung, colon, and leukemia models. This level of bioactivity underscores the importance of securing a reliable supply chain for these complex intermediates. For procurement managers and supply chain heads, understanding the synthetic accessibility of these molecules is paramount. The patented process eliminates the need for exotic catalysts or extreme reaction conditions, thereby reducing the barrier to entry for commercial-scale production. By leveraging this intellectual property, manufacturers can ensure a consistent flow of high-quality materials essential for preclinical and clinical development programs, ultimately accelerating the timeline from benchtop discovery to patient availability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex quinoxaline derivatives has often been plagued by inefficiencies that hinder large-scale manufacturing. Traditional routes frequently rely on multi-step sequences involving harsh reagents, toxic solvents, or expensive transition metal catalysts that require rigorous removal to meet pharmaceutical purity standards. In many legacy processes, the introduction of the sulfonyl group at the 3-position of the quinoxaline ring can be particularly challenging, often resulting in poor regioselectivity or low yields due to competing side reactions. Additionally, conventional methods may necessitate cryogenic temperatures or extended reaction times, which drastically increase energy consumption and operational costs. The purification of intermediates in these older pathways often involves tedious chromatographic separations or recrystallizations that are not easily adaptable to kilogram or ton-scale production. These bottlenecks create significant vulnerabilities in the supply chain, leading to unpredictable lead times and inflated costs for the final active pharmaceutical ingredients (APIs).
The Novel Approach
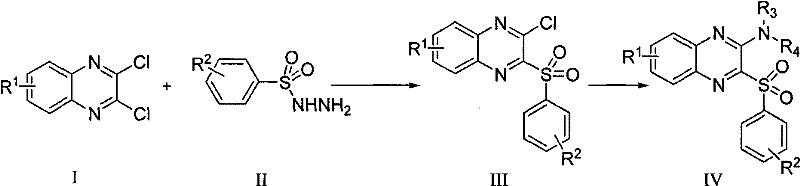
In stark contrast, the methodology outlined in patent CN101602737B presents a streamlined, two-step synthetic strategy that effectively circumvents these traditional hurdles. The core innovation lies in the sequential nucleophilic substitution of a 2,3-dichloroquinoxaline scaffold. Initially, the reaction with substituted benzenesulfonyl hydrazides in an alcohol solvent under reflux conditions selectively installs the sulfonyl moiety while retaining a reactive chlorine handle for further functionalization. This intermediate is then subjected to a second substitution with various amine compounds, allowing for rapid diversification of the molecular library. This approach is characterized by its operational simplicity, utilizing common solvents like ethanol, toluene, or isopropanol, and moderate heating conditions that are easily managed in standard reactor vessels. The result is a highly efficient pathway that minimizes waste generation and maximizes atom economy, providing a clear route for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Nucleophilic Aromatic Substitution on Quinoxaline Scaffold
The chemical elegance of this synthesis is rooted in the differential reactivity of the chlorine atoms on the 2,3-dichloroquinoxaline ring system. The first step involves a nucleophilic attack by the benzenesulfonyl hydrazide on the electron-deficient quinoxaline core. The nitrogen atoms within the pyrazine ring exert a strong electron-withdrawing effect, activating the adjacent carbon-chlorine bonds towards nucleophilic aromatic substitution (SNAr). Under reflux in an alcoholic medium, the hydrazide acts as a potent nucleophile, displacing one of the chlorine atoms to form the stable 2-chloro-3-(arylsulfonyl)quinoxaline intermediate. The choice of alcohol solvent is critical here, as it not only solubilizes the reactants but also participates in proton transfer events that facilitate the departure of the chloride leaving group. This step is highly regioselective, ensuring that the sulfonyl group is installed precisely at the 3-position, setting the stage for the subsequent transformation.
The second mechanistic phase involves the displacement of the remaining chlorine atom at the 2-position by an amine nucleophile. This step demonstrates remarkable versatility, accommodating both aromatic amines (such as substituted anilines) and aliphatic cyclic amines (like morpholine or piperidine). The reaction can be driven thermally in inert solvents like toluene at elevated temperatures (up to 180°C) or accelerated using microwave irradiation in protic solvents. The mechanism proceeds through a Meisenheimer-like complex where the amine attacks the C-2 position, followed by the elimination of the chloride ion. This flexibility allows chemists to fine-tune the physicochemical properties of the final molecule, such as solubility and metabolic stability, by simply swapping the amine component. Furthermore, the ability to form physiologically acceptable salts post-synthesis ensures that the final drug substance meets the stringent requirements for bioavailability and formulation stability required in modern oncology therapies.
How to Synthesize N-Substituted-3-(Benzenesulfonyl)Quinoxaline-2-Amines Efficiently
Executing this synthesis requires careful attention to stoichiometry and temperature control to maximize yield and purity. The process begins with the preparation of the key intermediate by reacting the dichloroquinoxaline starting material with a slight excess of the sulfonyl hydrazide in ethanol. Following the isolation of the intermediate, the second substitution is performed by mixing the intermediate with the desired amine in a high-boiling solvent. The reaction progress should be monitored via TLC or HPLC to ensure complete conversion before proceeding to workup. Purification is typically achieved through standard column chromatography, yielding the target compound as a solid which can be further processed into salt forms. For detailed operational parameters and safety guidelines, please refer to the standardized protocol below.
- Reflux 2,3-dichloroquinoxaline derivatives with substituted benzenesulfonyl hydrazides in an alcohol solvent (methanol, ethanol, or isopropanol) to obtain 2-chloro-3-(arylsulfonyl)quinoxaline intermediates.
- React the resulting 2-chloro-3-(arylsulfonyl)quinoxaline intermediate with amine compounds (substituted anilines or cyclic secondary amines) in an inert or protic solvent at 40-180°C.
- Purify the target compound via column chromatography and optionally convert into physiologically acceptable salts such as hydrochlorides or acetates.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial strategic benefits for organizations managing the procurement of complex oncology intermediates. The primary advantage lies in the significant simplification of the manufacturing process, which directly translates to enhanced supply chain reliability and reduced operational risk. By utilizing a two-step sequence with readily available starting materials, manufacturers can mitigate the risks associated with sourcing exotic reagents or relying on single-source suppliers for critical catalysts. This decentralization of raw material needs fosters a more resilient supply network, ensuring that production schedules remain uninterrupted even in the face of global market fluctuations. Moreover, the use of common industrial solvents and standard heating equipment means that this process can be easily transferred between different manufacturing sites without requiring specialized infrastructure investments.
- Cost Reduction in Manufacturing: The economic implications of this technology are profound, primarily driven by the elimination of expensive transition metal catalysts and the reduction of processing steps. Traditional methods often require palladium or copper catalysts which not only add to the raw material cost but also necessitate costly downstream purification to remove trace metal residues to ppm levels. By employing a metal-free nucleophilic substitution strategy, this process inherently lowers the cost of goods sold (COGS). Additionally, the high atom economy and the ability to use cheaper solvents like ethanol contribute to a leaner manufacturing budget. The simplified purification workflow further reduces labor and consumable costs associated with chromatography, making the overall production model significantly more cost-efficient compared to legacy methodologies.
- Enhanced Supply Chain Reliability: Supply chain continuity is a critical concern for pharmaceutical companies, especially when dealing with life-saving oncology drugs. This synthetic route enhances reliability by relying on commodity chemicals that are produced in vast quantities globally. 2,3-Dichloroquinoxaline and various benzenesulfonyl chlorides (precursors to the hydrazides) are established industrial chemicals with stable pricing and availability. This reduces the likelihood of supply shortages that can derail clinical trials or commercial launches. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, leading to more consistent batch-to-batch performance. This predictability allows procurement managers to negotiate better long-term contracts and maintain optimal inventory levels without the fear of sudden production halts.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often introduces unforeseen challenges, but this methodology is inherently designed for scalability. The exothermic nature of the substitution reactions is manageable under standard reflux conditions, and the absence of hazardous reagents simplifies safety protocols. From an environmental standpoint, the process aligns well with green chemistry principles by minimizing waste generation and avoiding toxic heavy metals. The solvents used are either recyclable or easily treatable in standard wastewater facilities, reducing the environmental footprint of the manufacturing operation. This compliance with stringent environmental regulations not only avoids potential fines but also enhances the corporate social responsibility profile of the manufacturing partner, a factor increasingly valued by global pharmaceutical clients.
Frequently Asked Questions (FAQ)
To assist our partners in evaluating the feasibility of integrating these intermediates into their development pipelines, we have compiled answers to common technical inquiries based on the patent data. These questions address the core mechanism, the scope of applicability, and the practical aspects of scaling this chemistry. Understanding these details is crucial for R&D directors and process chemists who are tasked with selecting the most viable synthetic routes for new drug candidates. The following insights are derived directly from the experimental data and claims presented in the intellectual property documentation.
Q: What is the primary biological application of these quinoxaline derivatives?
A: These compounds function as potent antitumor agents, specifically demonstrating significant in vitro anti-proliferation effects against PC-3, A549, HCT116, and HL60 tumor cell lines, likely through PI3K signaling pathway inhibition.
Q: Are the starting materials for this synthesis readily available for industrial scale-up?
A: Yes, the synthesis utilizes commercially accessible precursors such as 2,3-dichloroquinoxaline derivatives and substituted benzenesulfonyl hydrazides, which facilitates reliable supply chain management and cost-effective manufacturing.
Q: How does this novel synthetic route improve upon conventional methods?
A: The patented method offers a streamlined two-step process under relatively mild reflux conditions, avoiding complex catalytic systems and enabling easier purification, which significantly enhances operational simplicity and production efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted-3-(Benzenesulfonyl)Quinoxaline-2-Amine Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure necessary to bring this sophisticated chemistry from the patent literature to commercial reality. Our team of experienced process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We understand that in the competitive landscape of oncology drug development, time is of the essence. Therefore, our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest international standards. Our commitment to quality assurance extends beyond simple analysis; we proactively monitor impurity profiles to ensure that the material is suitable for subsequent GMP manufacturing of the final API.
We invite you to collaborate with us to leverage this innovative synthetic technology for your next-generation antitumor programs. Whether you require custom synthesis for early-stage research or large-scale production for clinical supply, our technical procurement team is ready to support your specific requirements. We encourage you to contact us today to request a Customized Cost-Saving Analysis tailored to your project's unique volume and purity needs. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that will empower your decision-making process. Let us be your trusted partner in navigating the complexities of pharmaceutical intermediate manufacturing, delivering value through science, reliability, and unwavering commitment to excellence.
