Scalable Production of Chiral Halo-Phenyl Ethanol Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical and agrochemical industries increasingly demand high-purity chiral intermediates to ensure the efficacy and safety of final active ingredients. Patent CN1359887A introduces a groundbreaking method for the production of optically active 2-halo-1-(substituted phenyl)ethanols and oxidized substituted styrenes, addressing critical limitations in existing asymmetric synthesis technologies. This innovative approach utilizes a phthalate ester intermediate strategy combined with optical resolution using photoactive organic amines, offering a robust pathway for manufacturing complex chiral building blocks. Unlike traditional microbial fermentation or metal-catalyzed asymmetric oxidation which often suffer from inconsistent yields and high operational costs, this chemical resolution method provides precise control over stereochemistry. The technology is particularly relevant for the synthesis of ketene S,S-diol derivatives, which serve as vital precursors in the development of novel therapeutic agents and crop protection chemicals. By leveraging this patented methodology, manufacturers can achieve substantial improvements in optical purity while maintaining a scalable and cost-effective production workflow suitable for global supply chains.
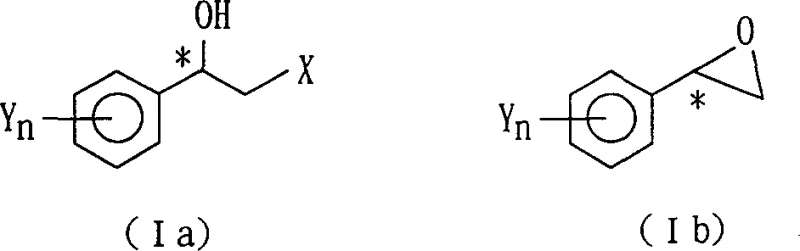
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of photoactive styrene oxides and related chiral alcohols has relied heavily on biological methods or complex asymmetric catalytic systems that present significant commercial hurdles. Microbial production methods, while selective, often require stringent fermentation conditions and extensive downstream processing to isolate the product from biological media, leading to prolonged lead times and variable batch consistency. Furthermore, asymmetric synthesis methods reported by Sharpless and others, although chemically elegant, frequently necessitate the use of expensive chiral ligands and transition metal catalysts that pose contamination risks and require rigorous removal steps to meet pharmaceutical purity standards. These conventional routes often struggle to balance operating efficiency with optical purity, resulting in processes that are difficult to scale from laboratory benchtop to multi-ton commercial production without sacrificing yield. The lack of a universal rule for selecting resolving agents for specific diastereomers has also forced manufacturers into costly trial-and-error development cycles, delaying time-to-market for critical drug intermediates and increasing the overall cost of goods sold.
The Novel Approach
The methodology disclosed in patent CN1359887A circumvents these challenges by employing a diastereomeric resolution strategy centered on phthalate ester derivatives. This novel approach converts racemic 2-halo-1-(substituted phenyl)ethanols into phthalate esters, which are then subjected to optical resolution using readily available photoactive organic amines. The formation of diastereomeric salts allows for separation via fractional crystallization, a unit operation that is well-understood and easily scalable in standard chemical manufacturing facilities. This route eliminates the dependency on precious metal catalysts and complex bioreactors, thereby drastically simplifying the process infrastructure and reducing capital expenditure. The ability to tune the resolution efficiency by selecting specific amines such as alpha-methylbenzylamine or quinidine provides manufacturers with flexibility to optimize for either yield or purity depending on the specific requirements of the downstream application. Moreover, the process includes a racemization step for the unwanted isomer, enabling a dynamic kinetic resolution that maximizes atom economy and minimizes chemical waste, aligning with modern green chemistry principles and environmental compliance standards.
Mechanistic Insights into Phthalate Ester Optical Resolution
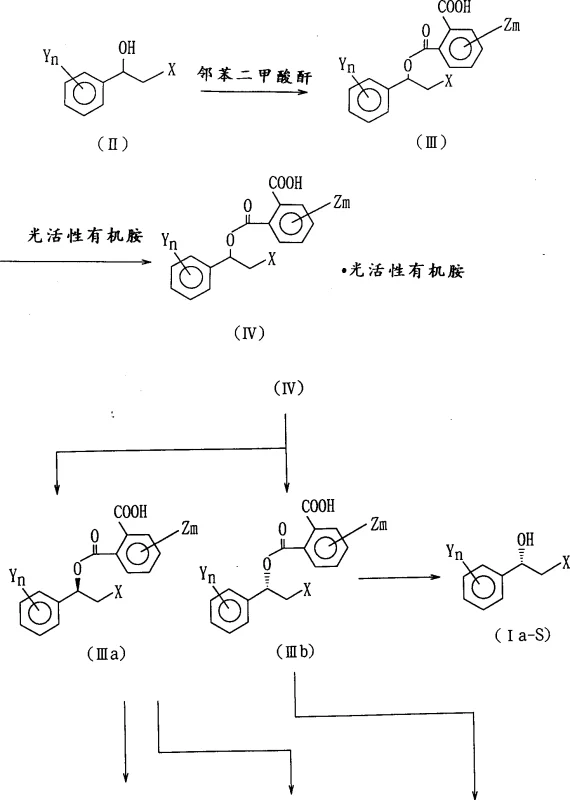
The core of this technology lies in the precise chemical transformation of the racemic alcohol into a diastereomeric salt system that can be physically separated. The process begins with the esterification of the racemic 2-halo-1-(substituted phenyl)ethanol with phthalic anhydride in an inert solvent such as pyridine or toluene, often accelerated by bases like triethylamine or 4-dimethylaminopyridine. This step generates a phthalate ester intermediate that possesses a carboxylic acid group capable of forming stable salts with chiral amines. When reacted with a photoactive organic amine resolving agent, such as (+)-alpha-methylbenzylamine, the racemic phthalate ester forms two distinct diastereomeric salts with different physical properties, particularly solubility. By carefully controlling the solvent system, typically a mixture of ethanol and n-hexane, and the temperature profile, one diastereomer can be selectively crystallized out of the solution while the other remains in the mother liquor. This physical separation is the key to achieving high optical purity, as the crystalline lattice energetically favors the incorporation of one enantiomer over the other, effectively amplifying the stereochemical excess.
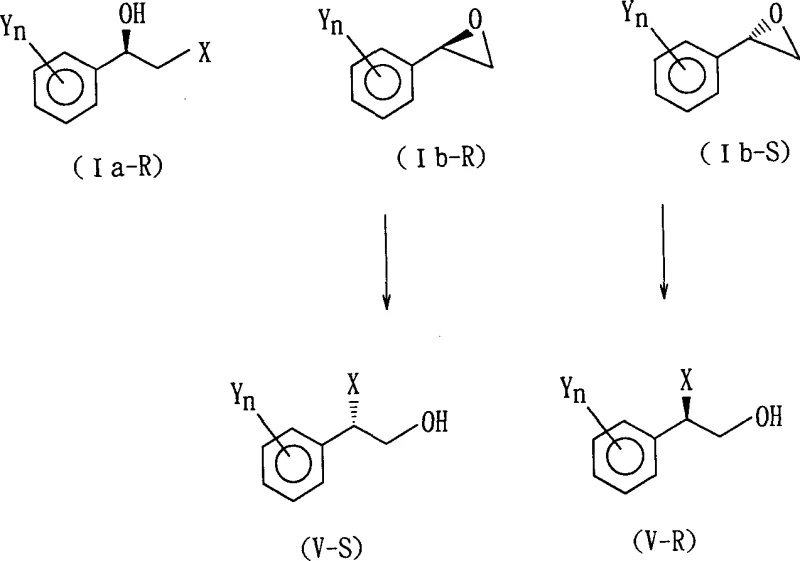
Following the isolation of the desired diastereomeric salt, the free optically active alcohol is recovered through hydrolysis or alcoholysis under acidic or basic conditions. The patent details that this cleavage step can be performed using mineral acids like hydrochloric acid or organic acids like acetic acid in solvents such as dioxane or water-alcohol mixtures. Crucially, the optical purity of the final product is directly correlated with the purity of the crystallized salt, which can be further enhanced through recrystallization cycles if necessary. The mechanism also accounts for the stability of the chiral center during these transformations, ensuring that no racemization occurs during the hydrolysis step provided the reaction conditions are kept within the specified temperature ranges of 0°C to 50°C. For applications requiring the epoxide derivative, the optically active alcohol can be cyclized under basic conditions, or conversely, the epoxide can be ring-opened with hydrogen halides to regenerate the chiral halohydrin with inverted or retained configuration depending on the specific reaction pathway chosen, offering versatile synthetic utility for diverse medicinal chemistry campaigns.
How to Synthesize Optically Active 2-Halo-1-(substituted phenyl)ethanol Efficiently
The synthesis of these high-value chiral intermediates follows a streamlined three-stage protocol designed for maximum operational efficiency and purity control. The process initiates with the derivatization of the racemic starting material to facilitate resolution, followed by the critical separation step using chiral amines, and concludes with the regeneration of the target molecule. Detailed standard operating procedures regarding solvent ratios, temperature gradients, and crystallization times are essential for reproducing the high optical purities reported in the patent data. Manufacturers must pay close attention to the selection of the resolving agent and the solvent system, as these variables dictate the efficiency of the diastereomeric separation. The following guide outlines the fundamental workflow required to implement this technology in a commercial setting, ensuring consistent quality and yield.
- React racemic 2-halo-1-(substituted phenyl)ethanol with phthalic anhydride in an inert solvent to form the corresponding phthalate ester derivative.
- Perform optical resolution by reacting the phthalate ester with a photoactive organic amine resolving agent to form diastereomeric salts, followed by fractional crystallization.
- Hydrolyze or alcoholize the resolved diastereomeric salt using acid or base to recover the free optically active 2-halo-1-(substituted phenyl)ethanol with high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers compelling advantages that directly impact the bottom line and operational resilience. By shifting away from expensive transition metal catalysts and complex bioprocesses, manufacturers can significantly reduce the raw material costs associated with chiral intermediate production. The use of commodity chemicals such as phthalic anhydride and alpha-methylbenzylamine ensures a stable and reliable supply of reagents, mitigating the risk of shortages that often plague specialized chiral catalysts. Furthermore, the scalability of the crystallization-based resolution process allows for seamless transition from pilot plant to full-scale commercial production, reducing the lead time for high-purity pharmaceutical intermediates. The ability to recycle the unwanted enantiomer through racemization further enhances the economic viability of the process by maximizing the utilization of starting materials and minimizing waste disposal costs. This results in a more sustainable and cost-efficient manufacturing model that aligns with the strategic goals of global chemical enterprises seeking to optimize their supply chains.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the reliance on standard organic synthesis equipment drastically lowers the capital and operational expenditures required for production. The process utilizes inexpensive resolving agents and solvents that are readily available in the global chemical market, avoiding the price volatility associated with specialized chiral ligands. Additionally, the high yield and optical purity achieved reduce the need for extensive downstream purification, further cutting processing costs and energy consumption. This economic efficiency makes the technology highly attractive for large-scale manufacturing of cost-sensitive generic pharmaceutical ingredients and agrochemical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the chemical resolution method ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and customer trust. The use of stable chemical reagents rather than biological cultures reduces the risk of production failures due to contamination or strain variability. Moreover, the flexibility to source resolving agents from multiple suppliers enhances supply chain security, preventing bottlenecks that could disrupt the delivery of critical intermediates to downstream customers. This reliability is essential for long-term supply agreements with major pharmaceutical companies that require guaranteed continuity of supply for their drug development pipelines.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as crystallization and filtration that are standard in the fine chemical industry. This facilitates rapid capacity expansion to meet increasing market demand without the need for specialized reactor configurations. The ability to recycle the unwanted isomer minimizes chemical waste generation, supporting environmental sustainability goals and reducing the burden on waste treatment facilities. The use of common solvents also simplifies solvent recovery and recycling systems, contributing to a lower environmental footprint and easier compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this optical resolution technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing accurate guidance for R&D and procurement teams evaluating this synthesis route. Understanding these details is crucial for assessing the feasibility of integrating this method into existing manufacturing workflows and for optimizing the production of chiral intermediates.
Q: What are the advantages of using phthalate esters for optical resolution compared to direct asymmetric synthesis?
A: The phthalate ester method described in patent CN1359887A offers superior operating efficiency and optical purity compared to microbial or Sharpless asymmetric methods. It avoids the need for expensive transition metal catalysts and allows for purification via simple recrystallization of diastereomeric salts, significantly reducing production costs and complexity.
Q: Which resolving agents are most effective for this specific chiral intermediate synthesis?
A: The patent identifies photoactive organic amines such as alpha-methylbenzylamine, quinidine, and cinchonidine as highly effective resolving agents. Specifically, alpha-methylbenzylamine has demonstrated the ability to achieve optical purities exceeding 94% ee after crystallization, providing a robust and reliable method for industrial scale-up.
Q: Can the unwanted enantiomer be recycled to improve overall yield?
A: Yes, the process includes a racemization step where the unwanted enantiomer or mother liquor can be treated with acid catalysts in inert solvents to regenerate the racemic starting material. This dynamic kinetic approach minimizes waste and maximizes the theoretical yield of the desired optically active product, enhancing overall process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Halo-1-(substituted phenyl)ethanol Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chiral intermediate manufacturing, leveraging advanced technologies like the phthalate ester resolution method to deliver superior quality products to the global market. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet the rigorous demands of international pharmaceutical and agrochemical clients. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of optically active 2-halo-1-(substituted phenyl)ethanol meets the highest standards of optical purity and chemical integrity. Our commitment to technical excellence allows us to navigate complex synthetic challenges and provide reliable supply solutions for even the most demanding chiral building blocks.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis route can optimize your supply chain and reduce costs. Request a Customized Cost-Saving Analysis to understand the specific economic benefits for your project. Our experts are ready to provide specific COA data and route feasibility assessments to support your development goals. By partnering with us, you gain access to a wealth of chemical expertise and a dedicated supply chain capable of supporting your growth from clinical trials to commercial launch.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
