Advanced Atmospheric Racemization Technology for Commercial Scale Production of R-Lipoic Acid
Advanced Atmospheric Racemization Technology for Commercial Scale Production of R-Lipoic Acid
The pharmaceutical and nutraceutical industries have long recognized the superior bioactivity of R-(+)-Lipoic Acid compared to its S-(-)-enantiomer, driving a critical demand for efficient, high-purity manufacturing processes. Patent CN102442994A introduces a groundbreaking methodology that addresses the historical inefficiency of chiral resolution by implementing a robust thermal racemization cycle for the unwanted S-isomer. This innovation transforms what was traditionally considered chemical waste into a valuable feedstock, fundamentally altering the economic landscape for producing this vital antioxidant intermediate. By operating under normal atmospheric pressure rather than the harsh high-pressure conditions of legacy methods, this technology offers a safer, more scalable pathway for reliable pharmaceutical intermediates supplier networks globally. The strategic integration of this racemization loop ensures that manufacturers can achieve near-total atom economy, significantly reducing the environmental footprint while maximizing output from raw materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of R-(+)-Lipoic Acid has been plagued by inherent inefficiencies associated with classical chiral resolution techniques, where the maximum theoretical yield is strictly capped at 50%. Traditional approaches often involve resolving racemic lipoic acid and discarding or stockpiling the less active S-(-)-enantiomer, leading to substantial material loss and inflated production costs. Furthermore, existing methods for recycling the S-isomer, such as those described in US Patent 5621117, frequently require extreme reaction conditions, including high-pressure apparatuses that demand significant capital investment and rigorous safety protocols. These legacy processes not only complicate the supply chain by necessitating specialized equipment but also introduce potential bottlenecks in commercial scale-up of complex pharmaceutical intermediates due to slower batch cycles and higher energy consumption. The reliance on expensive chiral reagents for direct asymmetric synthesis further exacerbates cost issues, making it difficult to produce high-purity OLED material or pharmaceutical grade ingredients at a competitive price point without compromising on quality or safety standards.
The Novel Approach
The methodology disclosed in the patent data presents a paradigm shift by utilizing a simple yet highly effective thermal racemization technique that operates under standard atmospheric pressure. Instead of discarding the S-(-)-Lipoic Acid obtained from the resolution mother liquor, this novel approach dissolves the isomer in specific high-boiling solvents and subjects it to prolonged reflux at temperatures ranging from 110°C to 220°C. This process effectively converts the optically active S-enantiomer back into a racemic mixture, which can then be re-fed into the resolution cycle, thereby breaking the 50% yield barrier. The simplicity of using common aromatic solvents like toluene or xylene eliminates the need for exotic catalysts or dangerous high-pressure reactors, drastically simplifying the process flow. This streamlined workflow not only enhances the overall yield to approximately 85% but also aligns perfectly with green chemistry principles by minimizing waste generation and optimizing resource utilization for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Thermal Racemization of Lipoic Acid
The core of this technological advancement lies in the thermal instability of the chiral center in Lipoic Acid when subjected to elevated temperatures in appropriate solvent systems. The mechanism involves the reversible opening of the dithiolane ring or the temporary formation of a planar intermediate at the chiral carbon, allowing for the interconversion between the R and S configurations until thermodynamic equilibrium is reached. By maintaining the reaction mixture at reflux temperatures between 110°C and 220°C for durations exceeding 15 hours, the system ensures complete equilibration, resulting in a racemate with an optical rotation value close to zero degrees. The choice of solvent plays a pivotal role in this mechanism, as solvents like diphenyl ether or trimethylbenzene provide the necessary thermal stability and solvation power to facilitate this configurational inversion without degrading the sensitive disulfide bond. Understanding this mechanistic pathway is crucial for R&D teams aiming to optimize reaction times and solvent ratios to achieve the highest possible purity specifications.
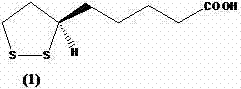

Controlling impurities during this thermal process is equally critical, as prolonged heating can potentially lead to polymerization or decomposition if not carefully monitored. The patent data indicates that the resulting racemized product exhibits an optical rotation of ±1 degree, demonstrating high efficiency in the stereochemical conversion. To further ensure purity, the process incorporates a recrystallization step using hexane, which effectively removes trace impurities and residual solvents that might have formed during the high-temperature reflux. This dual focus on efficient racemization and rigorous purification ensures that the recycled material meets the stringent quality standards required for downstream pharmaceutical applications. For technical directors, this implies a robust process capable of consistent batch-to-batch reproducibility, minimizing the risk of off-spec material that could disrupt the supply of high-purity pharmaceutical intermediates.
How to Synthesize R-(+)-Lipoic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step framework for implementing this recycling strategy in a production environment. It begins with the resolution of racemic Lipoic Acid using R-(+)-phenylethylamine to isolate the desired R-salt, followed by the acidification and recovery of the S-isomer from the mother liquor. The critical innovation lies in the subsequent treatment of this S-isomer, where it is dissolved in a solvent like toluene and refluxed to regenerate the racemic starting material. This cyclical process allows manufacturers to continuously feed the 'waste' stream back into the production line, maximizing the utility of every gram of raw material purchased. Detailed standardized synthetic steps for this procedure are provided in the guide below to assist process engineers in replicating these results.
- Dissolve S-(-)-Lipoic Acid in a high-boiling solvent such as toluene or xylene and reflux at 110-220°C for over 15 hours to achieve racemization.
- Resolve the resulting racemic Lipoic Acid using R-(+)-phenylethylamine to precipitate the desired R-(+)-Lipoic Acid salt.
- Recycle the mother liquor containing S-(-)-Lipoic Acid back into the racemization step to maximize overall yield and minimize waste.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this atmospheric racemization technology translates into tangible strategic advantages that go beyond mere technical novelty. By eliminating the need for high-pressure equipment, the barrier to entry for manufacturing is lowered, allowing for more flexible production scheduling and reduced maintenance downtime. The ability to recycle the S-enantiomer means that the effective cost of goods sold is significantly reduced, as the raw material input is utilized much more efficiently than in traditional 50% yield processes. This efficiency gain is particularly impactful in the context of volatile raw material markets, providing a buffer against price fluctuations and ensuring more stable pricing for long-term contracts. Furthermore, the simplified process flow reduces the complexity of the supply chain, minimizing the number of distinct unit operations and the associated logistical overhead.
- Cost Reduction in Manufacturing: The elimination of expensive high-pressure reactors and the recycling of the S-isomer lead to substantial capital and operational expenditure savings. By converting what was previously waste into a usable feedstock, the overall material cost per kilogram of final product is drastically lowered without the need for complex catalytic systems. This qualitative improvement in atom economy directly impacts the bottom line, making the production of R-(+)-Lipoic Acid more economically viable even at lower market prices. Additionally, the use of common, recoverable solvents like toluene further contributes to cost optimization by reducing solvent purchase and disposal expenses.
- Enhanced Supply Chain Reliability: The robustness of the atmospheric pressure process ensures higher equipment availability and reduces the risk of production stoppages due to safety incidents or equipment failure. Since the method relies on standard glass-lined or stainless steel reactors commonly found in fine chemical plants, sourcing contract manufacturing organizations becomes easier and more competitive. This accessibility enhances supply continuity, ensuring that downstream customers receive their orders on time without the delays often associated with specialized high-pressure processing slots. The simplified logistics of handling standard solvents and ambient pressure conditions also streamline the transportation and storage of intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from pilot batches to multi-ton commercial production without the engineering challenges posed by high-pressure scale-up. From an environmental perspective, the recycling of the S-isomer significantly reduces the volume of chemical waste requiring treatment, aligning with increasingly strict global environmental regulations. This reduction in waste discharge not only lowers disposal costs but also enhances the corporate sustainability profile of the manufacturer. The ability to operate with lower energy intensity compared to high-pressure alternatives further supports green manufacturing initiatives and reduces the overall carbon footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this racemization technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: What is the primary advantage of this racemization method over high-pressure techniques?
A: The primary advantage is the operation under normal atmospheric pressure, which eliminates the need for expensive high-pressure autoclaves and significantly reduces safety risks and capital expenditure compared to prior art methods requiring harsh pressure conditions.
Q: How does this process improve the overall yield of R-(+)-Lipoic Acid?
A: By recycling the unwanted S-(-)-enantiomer back into the racemic pool through thermal racemization, the theoretical yield is no longer capped at 50%, allowing for a cumulative yield of approximately 85% or higher through multiple cycles.
Q: Which solvents are suitable for the thermal racemization step?
A: The patent specifies high-boiling organic solvents such as toluene, xylene, trimethylbenzene, DMF, phenetole, diphenyl ether, or phenylcarbinol, which can sustain the required reflux temperatures of 110-220°C without degradation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-(+)-Lipoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the atmospheric racemization technology described in Patent CN102442994A for the efficient production of high-value chiral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients benefit from both laboratory precision and industrial robustness. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of R-(+)-Lipoic Acid delivered meets the highest international standards. We are committed to leveraging advanced process chemistry to drive down costs while maintaining the uncompromising quality required by the global pharmaceutical and nutraceutical sectors.
We invite forward-thinking partners to collaborate with us to explore how this optimized synthesis route can enhance your supply chain resilience and profitability. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your project timelines and secure your position in the competitive market for high-purity pharmaceutical intermediates.
