Scalable Synthesis of 3-(Methyl Alkoxy Phosphoryl) Propionate for High-Volume Agrochemical Production
Scalable Synthesis of 3-(Methyl Alkoxy Phosphoryl) Propionate for High-Volume Agrochemical Production
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of critical organophosphorus intermediates. Patent CN103342718A introduces a transformative methodology for preparing 3-(methyl alkoxy phosphoryl) propionate compounds, which serve as essential precursors for broad-spectrum herbicides like Glufosinate-ammonium. This technical disclosure represents a significant departure from traditional synthetic routes by eliminating the need for harsh anhydrous and anaerobic conditions while simultaneously enabling the flexible synthesis of mixed alkoxy derivatives. For R&D directors and procurement strategists, this patent offers a compelling value proposition: a robust, high-yielding process that simplifies supply chain logistics and reduces operational complexity. By leveraging anhydrous protonic solvents that function dually as reaction media and reactant sources, the method achieves product purities exceeding 98% and yields over 95%, setting a new benchmark for efficiency in agrochemical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phosphonate esters has been plagued by operational inefficiencies and stringent safety requirements that inflate production costs. One prominent prior art method, documented in J. Org. Chem. (1991), utilizes sodium ethoxide as a catalyst to react ethyl acrylate with methyl ethoxy phosphinic acid. While functional, this approach suffers from a yield ceiling of approximately 81.4% and necessitates complex post-reaction processing to remove the alkaline catalyst and separate by-products. 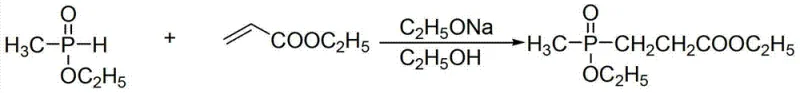 Furthermore, alternative pathways such as those described in CN101665514A rely on vinyl acids and demand strictly anhydrous and oxygen-free environments. These conditions require specialized reactor setups, inert gas purging systems, and rigorous moisture control, all of which contribute to substantial capital expenditure and increased lead times. The inability of these legacy methods to easily produce compounds with non-identical alkoxy groups further limits their utility in developing diverse pesticide libraries, creating a bottleneck for innovation in crop protection chemistry.
Furthermore, alternative pathways such as those described in CN101665514A rely on vinyl acids and demand strictly anhydrous and oxygen-free environments. These conditions require specialized reactor setups, inert gas purging systems, and rigorous moisture control, all of which contribute to substantial capital expenditure and increased lead times. The inability of these legacy methods to easily produce compounds with non-identical alkoxy groups further limits their utility in developing diverse pesticide libraries, creating a bottleneck for innovation in crop protection chemistry.
The Novel Approach
In stark contrast, the methodology disclosed in CN103342718A revolutionizes the production landscape by utilizing dialkyl methylphosphonates and acrylates in anhydrous protonic solvents such as alcohols or carboxylic acids. This innovative strategy eliminates the need for external catalysts like sodium ethoxide and removes the burden of maintaining anaerobic conditions. The solvent itself plays a dual role, acting not only as the medium but also participating in the reaction to generate the corresponding ether or ester groups, thereby allowing for the precise engineering of mixed alkoxy structures where R1 and R2 can be different. 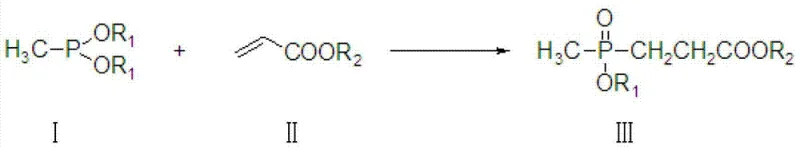 This flexibility is paramount for custom synthesis, enabling manufacturers to tailor intermediates for specific downstream applications without redesigning the entire process. The reaction proceeds smoothly at mild temperatures between 0°C and 50°C, drastically reducing energy consumption and safety risks associated with exothermic runaways, while consistently delivering yields superior to 95%.
This flexibility is paramount for custom synthesis, enabling manufacturers to tailor intermediates for specific downstream applications without redesigning the entire process. The reaction proceeds smoothly at mild temperatures between 0°C and 50°C, drastically reducing energy consumption and safety risks associated with exothermic runaways, while consistently delivering yields superior to 95%.
Mechanistic Insights into Protonic Solvent-Mediated Michael Addition
The core of this technological advancement lies in the unique interaction between the phosphonate substrate and the protonic solvent during the Michael addition process. Mechanistically, the reaction initiates with the formation of a reactive species from the dialkyl methylphosphonate, which then undergoes nucleophilic attack on the electron-deficient double bond of the acrylate. Unlike traditional base-catalyzed routes that generate stoichiometric salt waste, this system leverages the acidic protons of the solvent (e.g., ethanol or acetic acid) to facilitate the transformation without generating corrosive by-products. The solvent molecules actively participate in the stabilization of transition states and can engage in transesterification-like processes with alkyl anions generated in situ. This dynamic equilibrium ensures that the alkoxy groups on the final phosphonate product are derived from both the starting phosphonate and the solvent/acrylate components, providing a versatile handle for structural diversification. Such a mechanism inherently minimizes side reactions, leading to a cleaner crude profile that simplifies downstream purification.
From an impurity control perspective, the absence of strong alkaline catalysts like sodium ethoxide is a critical quality attribute for pharmaceutical and agrochemical grade intermediates. Traditional methods often leave trace metal residues or require extensive washing steps to neutralize basic impurities, which can degrade sensitive functional groups or complicate waste treatment. In this novel pathway, the reaction environment remains relatively neutral to mildly acidic, preventing the hydrolysis of the ester moieties and preserving the integrity of the phosphonate backbone. The result is a product stream with a content greater than 98%, significantly reducing the burden on quality control laboratories and ensuring that the material meets the stringent specifications required for global regulatory compliance. This high level of purity is achieved without the need for chromatographic separation, relying instead on efficient vacuum distillation, which is highly scalable for industrial operations.
How to Synthesize 3-(Methyl Alkoxy Phosphoryl) Propionate Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing fine chemical manufacturing facilities. The process begins by charging a reactor with an anhydrous protonic solvent, such as dehydrated ethanol or acetic acid, followed by the addition of either the acrylate or the dialkyl methylphosphonate. Temperature control is maintained rigorously between 0°C and 50°C, typically by cooling the mixture to around 5°C before the dropwise addition of the second reactant to manage the exotherm. Once the addition is complete, the reaction mixture is stirred for approximately two hours to ensure full conversion, as monitored by GC analysis showing residual starting materials below 1%. The detailed standardized synthesis steps, including specific molar ratios and distillation parameters, are outlined in the guide below.
- Dissolve acrylate or dialkyl methylphosphonate in an anhydrous protonic solvent such as ethanol or acetic acid.
- Control the reaction temperature between 0°C and 50°C while dropwise adding the second reactant to manage exothermicity.
- Maintain stirring for approximately 2 hours post-addition, then isolate the product via vacuum distillation to achieve >95% yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates directly into tangible operational efficiencies and risk mitigation. The elimination of strict anhydrous and anaerobic requirements removes the need for expensive inert atmosphere equipment and reduces the dependency on high-purity dry solvents, which are often subject to volatile pricing and supply constraints. Furthermore, the simplified workup procedure, which avoids complex catalyst filtration and neutralization steps, significantly shortens the batch cycle time. This acceleration in production throughput allows suppliers to respond more rapidly to market demands, effectively reducing lead times for high-purity agrochemical precursors. The robustness of the process also implies a lower rate of batch failures, ensuring a more reliable supply of critical intermediates for downstream herbicide formulation.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of costly catalytic reagents and the simplification of the purification train. By avoiding the use of sodium ethoxide and the subsequent waste treatment associated with neutralizing strong bases, manufacturers can achieve substantial savings in raw material and disposal costs. Additionally, the high yield exceeding 95% maximizes atom economy, ensuring that a greater proportion of expensive phosphonate starting materials is converted into saleable product rather than waste. The ability to use common solvents like ethanol or acetic acid, which are cheaper and more readily available than specialized aprotic solvents, further drives down the variable cost per kilogram, enhancing overall margin potential for large-scale production campaigns.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of commodity-grade raw materials that are less susceptible to geopolitical disruptions or niche market shortages. The tolerance for mild reaction conditions means that production can be outsourced to a wider network of contract manufacturing organizations (CMOs) without requiring specialized high-containment or ultra-dry facilities. This flexibility allows procurement teams to diversify their supplier base and negotiate more favorable terms. Moreover, the stability of the reaction parameters reduces the risk of unexpected shutdowns due to equipment failure or safety incidents, ensuring a consistent flow of materials to support continuous herbicide manufacturing schedules throughout the growing season.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial tonnage is straightforward due to the absence of hazardous reagents and the manageable thermal profile of the reaction. The mild temperature range of 0°C to 50°C can be easily maintained using standard jacketed reactors and chilled water systems, avoiding the need for cryogenic cooling or high-pressure steam. From an environmental standpoint, the reduction in chemical waste and the avoidance of heavy metal or strong base contaminants simplify wastewater treatment and align with increasingly strict global environmental regulations. This green chemistry profile not only reduces compliance costs but also enhances the corporate sustainability credentials of the final agrochemical product, appealing to environmentally conscious end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the feasibility of adopting this route for your specific supply chain needs. Understanding these nuances is critical for making informed decisions about process validation and vendor qualification.
Q: Does this synthesis require strict anhydrous or anaerobic conditions?
A: No, unlike prior art methods (e.g., CN101665514A), this patented process operates effectively in anhydrous protonic solvents without the need for rigorous oxygen-free environments, significantly simplifying equipment requirements.
Q: Can mixed alkoxy groups be synthesized using this method?
A: Yes, a key advantage of this technology is the ability to produce compounds with different alkoxy groups (R1 ≠ R2) by selecting specific alcohol or carboxylic acid solvents that participate in the reaction.
Q: What is the typical purity and yield of the final product?
A: The process consistently delivers product content greater than 98% and chemical yields exceeding 95%, minimizing waste and downstream purification costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(Methyl Alkoxy Phosphoryl) Propionate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the competitive agrochemical landscape. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of patent CN103342718A are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 3-(methyl alkoxy phosphoryl) propionate meets the highest industry standards. Our commitment to quality assurance ensures that our clients receive materials that are ready for immediate downstream processing, minimizing delays and maximizing overall process efficiency.
We invite you to collaborate with us to optimize your supply chain for next-generation herbicides. By leveraging our expertise in organophosphorus chemistry, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to validate the performance of our materials against your internal benchmarks. Together, we can drive innovation and cost-effectiveness in the global crop protection market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
