Advanced Solid Methionine Hydroxy Analogue Calcium Salt for Efficient Animal Nutrition
Advanced Solid Methionine Hydroxy Analogue Calcium Salt for Efficient Animal Nutrition
The global animal nutrition sector is constantly seeking innovative solutions to balance feed efficiency with operational safety and cost management. A significant breakthrough in this domain is documented in patent CN113068769A, which discloses a novel solid methionine hydroxy analogue calcium salt compound possessing unique antibacterial and acidifying properties. This invention addresses the longstanding dichotomy between the functional benefits of liquid methionine and the handling convenience of solid powders. By engineering a composite material that integrates calcium 2-hydroxy-4-methylthiobutyrate with residual liquid methionine and water, the technology delivers a product that mimics the biological efficacy of liquid forms while eliminating the logistical nightmares associated with corrosive liquids. For procurement leaders and R&D teams, this represents a pivotal shift towards safer, more efficient feed additive sourcing strategies that do not compromise on nutritional performance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied heavily on liquid methionine or fully neutralized calcium salts, both of which present distinct operational challenges. Liquid methionine, while biologically potent, is a dark brown viscous liquid with strong corrosivity and a pungent odor, necessitating expensive specialized storage tanks and automated spraying systems that increase capital expenditure significantly. Furthermore, its high acidity can trigger unwanted reactions with other feed components, and its viscosity leads to poor mixing uniformity, especially in low-temperature environments. On the other hand, traditional solid calcium salts produced via existing patents often undergo complete neutralization, resulting in a pH near 7. While easier to handle, these fully neutralized salts lose the critical acidifying and antibacterial functions inherent to the free acid form, thereby limiting their utility as a comprehensive feed solution and failing to replace the need for separate acidifiers in feed formulations.
The Novel Approach
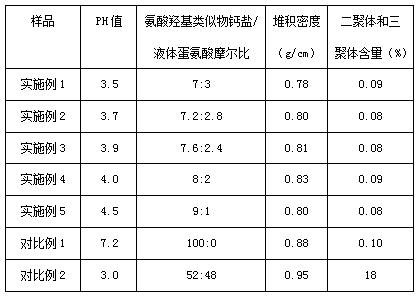
The methodology outlined in CN113068769A revolutionizes this landscape by introducing a controlled partial neutralization process followed by spray drying. Instead of driving the reaction to completion, the process meticulously maintains a molar ratio of calcium salt to free acid between 3.5:0.5 and 4.5:1.5. This precise stoichiometric control ensures the final solid powder retains a pH range of 3.5 to 4.5, effectively locking in the antibacterial and acidifying capabilities of liquid methionine within a free-flowing solid matrix. The result is a loose, non-caking powder with a bulk density of approximately 0.80 g/cm³ that dissolves readily and mixes uniformly without the need for specialized liquid dosing infrastructure. This dual-functionality approach allows feed manufacturers to simplify their supply chains by sourcing a single ingredient that fulfills multiple nutritional and preservative roles simultaneously.
Mechanistic Insights into Controlled Neutralization and Polymer Suppression
The core chemical innovation lies in the suppression of oligomer formation during the synthesis. Liquid methionine naturally contains dimers and trimers which exhibit lower bioavailability and contribute to product instability and caking. The patented process utilizes a specific neutralization temperature range of 80-100°C and a rapid spray drying step at 100-140°C to kinetically trap the desired monomeric and salt forms while minimizing polymerization. By keeping the total content of dimers and trimers below 0.1%, the process ensures that the methionine source is highly bioavailable for animal metabolism. This is critical because, as illustrated in the biological pathway, the compound must be efficiently converted into L-methionine within the animal body to participate in protein synthesis and energy metabolism. High levels of polymers would otherwise hinder this conversion, reducing the overall feed conversion ratio and wasting valuable raw materials.
Furthermore, the reaction mechanism involves the direct interaction of liquid methionine aqueous solutions with calcium oxide or hydroxide under mild conditions. Unlike older methods that require hazardous precursors like hydrocyanic acid or complex multi-step hydrolysis and crystallization processes, this direct neutralization is thermodynamically favorable and kinetically fast. The process avoids the formation of sticky filter cakes common in crystallization methods by transitioning directly from a transparent liquid reaction mixture to a dry powder via atomization. This eliminates the need for energy-intensive recrystallization steps and solvent recovery systems, thereby streamlining the production workflow. The resulting microstructure of the powder particles prevents moisture absorption and agglomeration, solving the stability issues that have plagued previous generations of solid methionine analogues in humid storage environments.
How to Synthesize Methionine Hydroxy Analogue Calcium Salt Efficiently
The synthesis protocol described in the patent offers a robust framework for industrial implementation, focusing on simplicity and reproducibility. The process begins with the dilution of commercial-grade liquid methionine to a specific concentration, followed by the controlled addition of a calcium source. The reaction parameters, including temperature and pH, are tightly regulated to ensure the correct ratio of salt to free acid is achieved before the mixture enters the drying stage. This straightforward approach minimizes the risk of operator error and equipment fouling, making it an ideal candidate for continuous manufacturing setups. For detailed technical specifications and standardized operating procedures required for pilot or commercial scale-up, please refer to the structured guide below.
- Prepare an aqueous solution of liquid methionine (20-70 wt%) and heat to approximately 40°C with stirring.
- Add calcium oxide or calcium hydroxide powder gradually to the solution, maintaining a neutralization temperature between 80-100°C to achieve a pH of 3-6.
- Subject the resulting transparent liquid product to spray drying at 100-140°C to obtain the final powdery solid compound with high dispersibility.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this solid methionine hydroxy analogue calcium salt offers profound advantages for supply chain resilience and cost structure optimization. The transition from a corrosive liquid to a stable solid powder fundamentally alters the logistics profile of the raw material. It eliminates the need for dedicated corrosion-resistant tankers and specialized pumping systems, allowing the product to be transported using standard bulk powder handling equipment. This flexibility significantly reduces the barrier to entry for smaller feed mills that previously could not justify the capital investment for liquid methionine infrastructure. Moreover, the solid form mitigates the risks associated with leakage and spillage during transit, enhancing workplace safety and reducing environmental compliance costs related to hazardous liquid handling.
- Cost Reduction in Manufacturing: The streamlined synthesis route eliminates the need for expensive catalysts, organic solvents, and complex purification steps like recrystallization found in prior art. By utilizing a direct neutralization and spray drying sequence, the process achieves yields exceeding 99%, drastically minimizing raw material waste. The removal of solvent recovery units and the reduction in energy consumption associated with drying wet filter cakes translate into substantial operational expenditure savings. Additionally, the dual functionality of the product means feed formulators can potentially reduce the dosage of separate acidifiers and antibacterial agents, further lowering the total cost of goods sold for the final feed product.
- Enhanced Supply Chain Reliability: Solid powders inherently possess superior shelf-life characteristics compared to viscous liquids that may degrade or separate over time. The low hygroscopicity and resistance to caking ensure that the product remains free-flowing even after extended storage periods, reducing the incidence of inventory write-offs due to spoilage. This stability allows procurement managers to optimize inventory levels and negotiate better terms with suppliers, knowing that the material can be stored safely without degradation of its acidifying potency. The use of widely available raw materials like calcium oxide and commercial liquid methionine also insulates the supply chain from the volatility associated with niche chemical intermediates.
- Scalability and Environmental Compliance: The process is designed for seamless scalability, moving from laboratory benchtop to multi-ton production without significant re-engineering. The absence of hazardous byproducts like ammonia gas or salt-laden wastewater, which are common in alternative synthesis routes involving nitriles or ammonium salts, simplifies waste treatment protocols. This aligns with increasingly stringent environmental regulations, reducing the permitting burden and potential fines associated with effluent discharge. The high atom economy of the neutralization reaction ensures that nearly all input mass is converted into saleable product, supporting sustainability goals and improving the overall carbon footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel methionine source. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the product's performance metrics and operational benefits. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this compound into existing feed formulations or production lines.
Q: What distinguishes this solid methionine compound from traditional calcium salts?
A: Unlike traditional calcium salts which are fully neutralized (pH ~7) and lose acidifying properties, this novel compound retains a specific ratio of free acid (pH 3.5-4.5), preserving the antibacterial and acidifying functions of liquid methionine while offering solid handling benefits.
Q: How does the new process improve product stability and flowability?
A: By strictly controlling the dimer and trimer content to below 0.1% and utilizing spray drying, the process prevents the hygroscopicity and caking issues common in high-polymer liquid methionine products, ensuring excellent fluidity and long-term storage stability.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method utilizes simple neutralization and standard spray drying equipment, avoiding complex multi-step syntheses or hazardous reagents like hydrocyanic acid, making it highly suitable for large-scale commercial manufacturing with yields exceeding 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methionine Hydroxy Analogue Calcium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the solid methionine hydroxy analogue calcium salt technology in modernizing animal nutrition supply chains. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to this advanced feed additive is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical control of dimer and trimer content to guarantee optimal bioavailability. We are committed to delivering high-purity methionine sources that meet the exacting standards of global feed manufacturers while maintaining the highest levels of safety and consistency.
We invite you to collaborate with our technical procurement team to explore how this innovation can drive value for your organization. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your operation. We encourage you to reach out for specific COA data and route feasibility assessments to validate the compatibility of this compound with your current formulation matrix. Let us help you engineer a more resilient and cost-effective supply chain for your animal nutrition products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
