Scalable Synthesis of 2-Chloro-4,4,4-Trifluoroacetyl Ethyl Acetate for Industrial Fungicide Production
Scalable Synthesis of 2-Chloro-4,4,4-Trifluoroacetyl Ethyl Acetate for Industrial Fungicide Production
The global demand for high-efficiency, low-toxicity fungicides continues to drive innovation in the agrochemical sector, specifically for succinate dehydrogenase inhibitors like thifluzamide. At the heart of this value chain lies the critical intermediate, 2-chloro-4,4,4-trifluoroacetyl ethyl acetate, whose efficient production is paramount for downstream drug synthesis. Patent CN103570533A introduces a robust preparation method that addresses longstanding challenges in fluorinated intermediate manufacturing by utilizing direct contact reactions between trifluoroacetyl ethyl acetate and chlorinating agents such as sulfuryl chloride or chlorine gas. This technological breakthrough offers a streamlined pathway that eliminates the need for organic solvents, thereby simplifying the purification process and enhancing overall process safety. For R&D directors and process engineers, understanding the nuances of this solvent-free protocol is essential for optimizing yield and purity profiles in industrial settings. The method demonstrates exceptional selectivity, ensuring that the reactive alpha-position is chlorinated without compromising the integrity of the trifluoromethyl group or the ester functionality. Furthermore, the ability to conduct this reaction under relatively mild thermal conditions represents a significant advancement over traditional halogenation techniques that often require harsh catalysts or extreme temperatures. As we delve deeper into the technical specifics, it becomes clear that this patent provides a foundational blueprint for reliable agrochemical intermediate supplier networks aiming to secure stable supply chains for next-generation crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-chlorinated beta-keto esters has been plagued by issues related to poor selectivity and excessive waste generation. Traditional methods often rely on the use of volatile organic solvents to dissipate the heat of reaction and dissolve reagents, which subsequently necessitates energy-intensive distillation steps for solvent recovery. In many legacy processes, the use of molecular chlorine in the presence of Lewis acid catalysts can lead to over-chlorination, resulting in a complex mixture of mono- and di-chlorinated impurities that are difficult to separate. This not only diminishes the overall yield of the desired 2-chloro-4,4,4-trifluoroacetyl ethyl acetate but also complicates the waste treatment protocols due to the presence of hazardous halogenated byproducts. Moreover, the handling of gaseous chlorine in open or semi-open systems poses significant safety risks to plant personnel and requires specialized containment infrastructure. The economic burden of these conventional approaches is further exacerbated by the low atom economy associated with stoichiometric reagents that do not fully convert, leading to higher raw material consumption per kilogram of final product. Consequently, manufacturers have long sought a more direct, cleaner, and cost-effective route that minimizes downstream processing requirements while maximizing throughput.
The Novel Approach
The methodology disclosed in the patent data presents a paradigm shift by employing a solvent-free system that leverages the intrinsic reactivity of trifluoroacetyl ethyl acetate. By directly contacting the substrate with sulfuryl chloride or chlorine gas, the process achieves high conversion rates without the dilution effects of a solvent medium. This concentrated reaction environment enhances the collision frequency between reactant molecules, thereby accelerating the reaction kinetics and allowing for shorter cycle times. Specifically, the use of sulfuryl chloride offers the dual benefit of acting as both a chlorinating agent and a source of sulfur dioxide, which evolves as a gas and drives the equilibrium forward. Alternatively, the direct chlorination route using chlorine gas is noted for its rapid reaction profile, capable of reaching completion within a mere 3 to 4 hours under controlled conditions. The absence of solvent not only reduces the physical volume of the reaction mixture, allowing for larger batch sizes in existing reactors, but also simplifies the workup procedure to a straightforward vacuum distillation. This streamlined approach effectively removes residual reagents and acidic byproducts, yielding a product with high purity suitable for immediate use in subsequent synthetic steps. 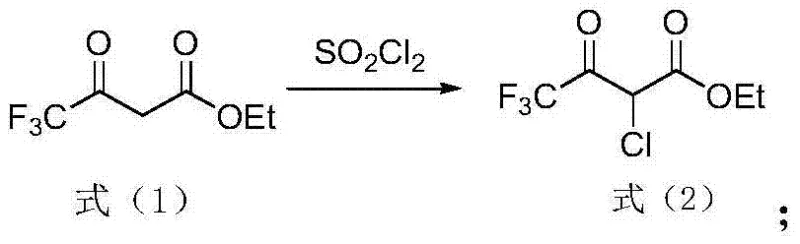
Mechanistic Insights into Solvent-Free Alpha-Chlorination
From a mechanistic perspective, the chlorination of trifluoroacetyl ethyl acetate involves an electrophilic substitution at the active methylene carbon situated between the two carbonyl groups. The presence of the strongly electron-withdrawing trifluoromethyl group significantly increases the acidity of the alpha-protons, facilitating the formation of the enol or enolate tautomer which acts as the nucleophile. In the absence of a solvent, the interaction between the enolic form of the substrate and the electrophilic chlorine species (derived from SO2Cl2 or Cl2) is highly efficient. The reaction proceeds through a transition state where the chlorine atom is transferred to the alpha-carbon, accompanied by the elimination of hydrogen chloride. One of the critical challenges in this transformation is preventing poly-chlorination, as the introduction of the first chlorine atom further activates the remaining alpha-proton towards subsequent substitution. However, the patent data indicates that by strictly controlling the molar ratio of the chlorinating agent to the substrate—specifically maintaining a ratio between 1:1 and 1:1.5—the reaction can be arrested at the mono-chlorinated stage with high selectivity. This precise stoichiometric control is vital for maintaining the quality of the intermediate and avoiding the formation of difficult-to-remove dichloro impurities. Additionally, the temperature range of 0-50°C provides a kinetic window that balances reaction rate with selectivity, ensuring that the thermal energy is sufficient to overcome the activation barrier without promoting side reactions or decomposition of the sensitive fluorinated backbone.
Impurity control in this system is largely governed by the efficient removal of gaseous byproducts. As the reaction progresses, hydrogen chloride and, in the case of sulfuryl chloride, sulfur dioxide are generated. In a solvent-free environment, these gases can escape the reaction matrix more readily, especially under reduced pressure or with mild agitation, preventing them from participating in reverse reactions or catalyzing degradation pathways. The final purification step involves vacuum distillation at temperatures between 20-50°C, which is sufficiently low to prevent thermal degradation of the product while effectively stripping away any unreacted starting material or volatile chlorinating agents. This gentle isolation technique preserves the structural integrity of the ester and ketone functionalities, which is crucial for the subsequent coupling reactions required to synthesize thifluzamide. The result is a product stream with purity levels reaching up to 98%, demonstrating the efficacy of this mechanistic approach in producing high-specification chemical building blocks. 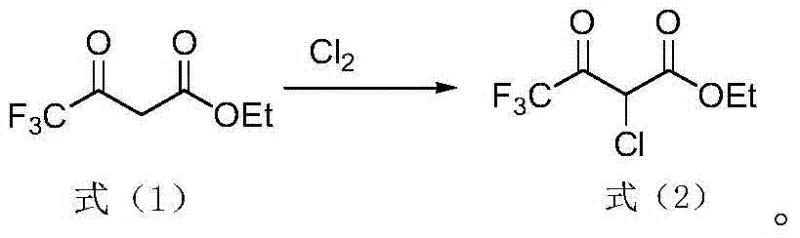
How to Synthesize 2-Chloro-4,4,4-Trifluoroacetyl Ethyl Acetate Efficiently
The synthesis of this critical intermediate requires precise adherence to the operational parameters outlined in the patent to ensure reproducibility and safety on a commercial scale. The process begins with the charging of high-purity trifluoroacetyl ethyl acetate into a reactor equipped with efficient cooling and gas scrubbing capabilities. Depending on the chosen chlorinating agent, the temperature must be rigorously maintained; for sulfuryl chloride, a range of 25-35°C is optimal, whereas chlorine gas introduction requires colder conditions around 0-5°C to manage the exotherm. The addition of the chlorinating agent should be performed dropwise or via controlled sparging to prevent local hotspots that could trigger runaway reactions. Following the reaction period, which may extend up to 24 hours for the sulfuryl chloride route or as little as 3 hours for the chlorine route, the mixture is subjected to reduced pressure distillation.
- Charge trifluoroacetyl ethyl acetate into a reactor and cool to 0-50°C depending on the chlorinating agent selected.
- Slowly add sulfuryl chloride or introduce chlorine gas while maintaining strict temperature control to manage exothermicity.
- Upon completion, remove residual gases and unreacted reagents under reduced pressure to isolate the pure chlorinated product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free synthesis route offers compelling economic and logistical benefits that directly impact the bottom line. The elimination of organic solvents translates to a drastic reduction in raw material procurement costs, as there is no longer a need to purchase, store, and recover large volumes of volatile organic compounds. This simplification of the bill of materials also reduces the complexity of inventory management and lowers the risk associated with the storage of flammable liquids. Furthermore, the use of commodity chemicals like sulfuryl chloride and chlorine gas ensures a stable and reliable supply of reagents, mitigating the risk of production stoppages due to specialty chemical shortages. The high atom economy of the reaction means that less raw material is wasted, leading to significant cost reduction in fungicide intermediate manufacturing without compromising on output quality. From a logistics perspective, the simplified workup procedure reduces the turnaround time for each batch, allowing facilities to increase their throughput and respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The solvent-free nature of this process eliminates the capital and operational expenditures associated with solvent recovery units and waste incineration. By removing the need for distillation columns dedicated solely to solvent recycling, manufacturers can redirect energy resources towards the primary product isolation, thereby lowering the utility cost per kilogram of product. Additionally, the high yield reported in the patent examples implies that less starting material is required to produce the same amount of final product, further driving down the variable cost of goods sold. The reduction in waste volume also lowers the fees associated with hazardous waste disposal, contributing to a leaner and more profitable operational model.
- Enhanced Supply Chain Reliability: Relying on widely available industrial gases and liquids like chlorine and sulfuryl chloride insulates the supply chain from the volatility often seen in the market for specialized fine chemical reagents. This accessibility ensures that production schedules can be maintained consistently, reducing lead time for high-purity agrochemical building blocks. The robustness of the reaction conditions, which tolerate slight variations in temperature without catastrophic failure, adds a layer of resilience to the manufacturing process, ensuring that delivery commitments to downstream pharmaceutical or agrochemical clients are met reliably. This stability is crucial for maintaining long-term contracts and fostering trust with global partners who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The simplicity of the reactor setup, which does not require complex solvent handling systems, makes this process highly amenable to commercial scale-up of complex fluorinated intermediates. Facilities can easily transition from pilot plant scales to multi-ton production runs with minimal equipment modification. Environmentally, the process aligns with green chemistry principles by minimizing solvent emissions and reducing the overall E-factor (mass of waste per mass of product). The efficient scrubbing of acidic off-gases ensures compliance with stringent environmental regulations, protecting the manufacturer from potential fines and reputational damage while promoting a sustainable corporate image.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-chloro-4,4,4-trifluoroacetyl ethyl acetate. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering a transparent view of the process capabilities. Understanding these details helps stakeholders make informed decisions regarding process integration and quality assurance protocols.
Q: What are the primary advantages of the solvent-free chlorination method described in CN103570533A?
A: The solvent-free approach significantly reduces waste generation and eliminates the need for solvent recovery systems, leading to lower operational costs and a smaller environmental footprint compared to traditional solution-phase chlorinations.
Q: How does this process ensure high selectivity for the mono-chlorinated product?
A: By carefully controlling the molar ratio of the chlorinating agent (1:1 to 1:1.5) and maintaining moderate temperatures (0-50°C), the reaction favors mono-substitution at the active methylene position, minimizing di-chlorinated byproducts.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the use of commodity chemicals like sulfuryl chloride or chlorine gas, combined with simple distillation workup, makes this route highly scalable and economically viable for multi-ton production of agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-4,4,4-Trifluoroacetyl Ethyl Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the synthesis of advanced agrochemicals like thifluzamide. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our capability to implement the solvent-free chlorination technology described in CN103570533A allows us to offer a competitive product with a superior impurity profile, catering to the exacting standards of the global pharmaceutical and crop protection industries.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our solutions align perfectly with your project milestones and regulatory needs. Together, we can drive efficiency and innovation in the production of essential fluorinated intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
