Advanced Manufacturing of Nitalapram Intermediates Using Novel Grignard Cyclization Technology
Introduction to Next-Generation Nitalapram Synthesis
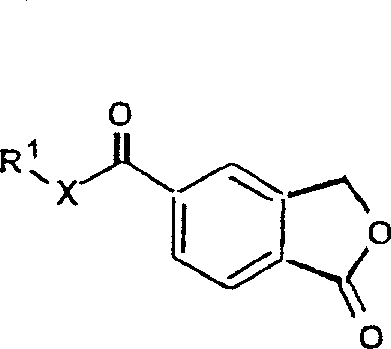
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the production of critical antidepressant agents like Nitalapram (Citalopram). Patent CN1268129A discloses a groundbreaking preparation method that fundamentally shifts the synthetic paradigm away from hazardous heavy metal reagents toward a safer, more efficient Grignard-based protocol. This technology leverages stable 5-substituted-2-benzo[c]furanone derivatives—specifically esters or amides—as the foundational building blocks, reacting them sequentially with organomagnesium reagents to construct the complex molecular architecture. By replacing the traditional cupric cyanide exchange or harsh sulfuric acid cyclization steps with a controlled dehydration of ester/amide intermediates, this process offers a compelling value proposition for manufacturers aiming to optimize their supply chains while adhering to stricter environmental regulations.
The strategic importance of this methodology lies in its ability to generate high-purity intermediates through a sequence that is inherently more forgiving and scalable than prior art. The core innovation involves the formation of a 1,3-dihydroisobenzofuran skeleton via a double Grignard addition followed by a ring-closure event that preserves the delicate functional groups required for biological activity. For R&D directors and process chemists, this represents a significant opportunity to redefine the impurity profile of the final API, as the avoidance of copper residues simplifies downstream purification. Furthermore, the versatility of the starting materials allows for the tuning of reaction kinetics, ensuring that the process remains viable from kilogram-scale development up to multi-ton commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Nitalapram has been plagued by significant safety and efficiency bottlenecks associated with the introduction of the cyano group and the formation of the isobenzofuran ring. Traditional routes often rely on the reaction of 5-bromo derivatives with cupric cyanide, a process that introduces severe toxicity risks due to the generation of cyanide ions and copper waste, necessitating complex and costly wastewater treatment protocols. Alternatively, methods involving the direct cyclization of 5-cyano-2-benzo[c]furanone derivatives using concentrated sulfuric acid pose challenges regarding corrosion control and the potential for hydrolyzing the sensitive nitrile group under such aggressive acidic conditions. These legacy processes not only increase the operational expenditure related to hazard management but also limit the flexibility of the manufacturing line due to the stringent containment requirements for toxic reagents.
Moreover, the reliance on specific halogenated precursors in older methodologies can lead to supply chain vulnerabilities, as the availability of high-purity 5-bromo-phthalides fluctuates with market demand. The multi-step nature of these conventional routes, often requiring isolation of unstable intermediates, results in cumulative yield losses and increased solvent consumption, which directly impacts the cost of goods sold (COGS). For procurement managers, the hidden costs of disposing of heavy metal sludge and managing corrosive acid waste streams are substantial, often overshadowing the raw material costs themselves. Consequently, there is a pressing industry need for a synthetic route that decouples the production of this vital antidepressant from these hazardous and inefficient chemical transformations.
The Novel Approach
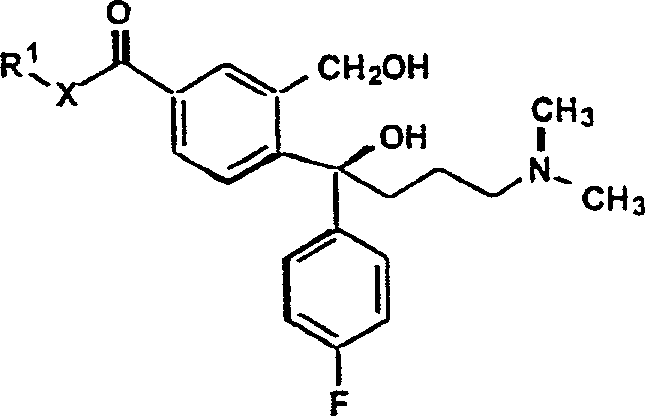
The methodology outlined in CN1268129A presents a transformative solution by utilizing 5-ester or 5-amide substituted phthalides as the primary substrates, which are reacted with 4-halogen-fluorophenyl Grignard reagents followed by 3-halo-N,N-dimethyl-propylamine Grignard reagents. This sequential organometallic strategy allows for the precise construction of the quaternary carbon center at the 1-position of the isobenzofuran ring without the immediate need for a nitrile group, thereby avoiding the compatibility issues associated with cyanide chemistry during the Grignard addition. The resulting intermediate, a 1,3-dihydroisobenzofuran derivative bearing an ester or amide moiety, is subsequently cyclized and dehydrated under mild acidic or basic conditions to yield the target nitrile. This approach effectively bypasses the need for cupric cyanide entirely, replacing a toxic substitution reaction with a cleaner dehydration step using reagents like thionyl chloride or phosphorus pentachloride.
From a process engineering perspective, this novel route offers superior controllability, as the ester-to-nitrile conversion can be finely tuned to minimize side reactions such as over-hydrolysis or polymerization. The ability to telescope the two Grignard additions without isolating the ketone intermediate (Formula IVa) significantly streamlines the workflow, reducing the number of reactor charges and thermal cycles required. This consolidation of steps not only enhances the overall throughput of the manufacturing facility but also improves the atom economy of the process. By shifting the complexity from hazardous waste management to controlled organic synthesis, this method aligns perfectly with modern green chemistry principles, offering a sustainable pathway for the reliable supply of high-purity Nitalapram intermediates to the global market.
Mechanistic Insights into Sequential Grignard Addition and Cyclization
The heart of this synthetic innovation lies in the dual nucleophilic attack on the lactone carbonyl of the 5-substituted-2-benzo[c]furanone. Initially, the 4-halogen-fluorophenyl magnesium halide acts as a potent nucleophile, opening the lactone ring to form a keto-acid salt intermediate, which upon workup or in situ transformation yields the ketone species (Formula IVa). This step is critical as it establishes the stereochemical and structural foundation for the subsequent addition. The second Grignard reagent, derived from 3-halo-N,N-dimethyl-propylamine, then attacks the newly formed ketone carbonyl. This second addition creates a tertiary alcohol intermediate which is primed for intramolecular cyclization. The presence of the dimethylamino group requires careful handling to prevent coordination with the magnesium species, yet the patent demonstrates that using magnesium bromide reagents effectively mitigates these coordination issues, ensuring high conversion rates.
Following the double addition, the cyclization to form the 1,3-dihydroisobenzofuran core (Formula V) is achieved through acid or base catalysis. In the acidic pathway, the tertiary hydroxyl group is protonated and eliminated as water, generating a carbocation that is immediately trapped by the adjacent oxygen atom of the original lactone framework, closing the five-membered ring. Alternatively, under basic conditions, the intermediate can be activated via sulfonate ester formation (e.g., using mesyl chloride) to create a better leaving group, facilitating the ring closure via an SN1-like mechanism. This mechanistic flexibility allows manufacturers to select conditions that best suit their existing equipment and safety protocols. Finally, the conversion of the pendant ester or amide group to the nitrile (Formula VI) proceeds via standard dehydration mechanisms, where reagents like thionyl chloride activate the carbonyl oxygen, leading to the elimination of water or alcohol and the formation of the carbon-nitrogen triple bond essential for the drug's pharmacological activity.
How to Synthesize Nitalapram Efficiently
The execution of this synthesis requires precise control over stoichiometry and temperature to manage the exothermic nature of Grignard reactions while ensuring complete conversion of the starting phthalide. The process begins with the preparation of the 5-substituted-2-benzo[c]furanone, which can be derived from commercially available 5-carboxyl-2-benzo[c]furanone via simple esterification or amidation. Detailed standardized operating procedures for the sequential addition of Grignard reagents, the specific workup protocols to isolate the cyclized intermediate, and the final dehydration conditions are critical for reproducibility. To ensure successful implementation in a GMP environment, operators must adhere to strict anhydrous conditions during the organometallic steps and utilize appropriate quenching strategies to handle the reactive magnesium salts safely.
- React 5-substituted-2-benzo[c]furanone (ester or amide) with 4-halogen-fluorophenyl magnesium halide to form the ketone intermediate.
- Without isolation, react the ketone intermediate with 3-halo-N,N-dimethyl-propylamine magnesium halide to introduce the amine side chain.
- Perform acid or base-catalyzed cyclization to close the isobenzofuran ring, followed by dehydration of the ester/amide group to yield the nitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patent-protected synthesis route translates into tangible operational efficiencies and risk mitigation. The most significant advantage is the complete elimination of cupric cyanide from the supply chain, which removes the regulatory burden and high costs associated with the transport, storage, and disposal of Class A poisons and heavy metal waste. This shift not only lowers the direct cost of raw materials but also drastically reduces the indirect costs related to environmental compliance and hazardous waste treatment facilities. By utilizing stable ester or amide precursors, the process ensures a more reliable supply of starting materials, as these chemicals are less prone to degradation during storage and transport compared to sensitive halogenated intermediates used in older methods.
- Cost Reduction in Manufacturing: The telescoping of the two Grignard reactions into a single pot operation significantly reduces solvent usage and energy consumption by eliminating an intermediate isolation and drying step. Furthermore, the replacement of expensive and toxic copper reagents with more common organomagnesium species leads to substantial savings in reagent costs. The simplified downstream processing, characterized by the absence of copper removal steps (such as chelation or extensive washing), reduces the load on purification units and shortens the batch cycle time, thereby increasing the overall asset utilization of the manufacturing plant.
- Enhanced Supply Chain Reliability: The reliance on robust, non-hazardous intermediates enhances the resilience of the supply chain against regulatory disruptions. Since the process avoids reagents that are subject to strict import/export controls or sudden bans due to environmental concerns, manufacturers can maintain continuous production schedules with greater confidence. The use of widely available reagents like thionyl chloride and common Grignard precursors ensures that the supply of critical inputs remains stable, reducing the risk of production stoppages caused by raw material shortages.
- Scalability and Environmental Compliance: This methodology is inherently scalable, as the exothermicity of the Grignard additions can be managed effectively using standard cooling protocols in large-scale reactors. The reduction in hazardous waste generation aligns with increasingly stringent global environmental standards, facilitating easier permitting for capacity expansion. The cleaner impurity profile resulting from the avoidance of copper contamination simplifies the validation process for regulatory filings, accelerating the time-to-market for generic versions of the drug and securing a competitive advantage in the marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Nitalapram synthesis route. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term viability of the supply partnership.
Q: How does this new method improve safety compared to traditional Cupric Cyanide routes?
A: The traditional method relies on cupric cyanide for cyanation, which generates highly toxic copper-containing waste streams requiring expensive disposal. This novel route utilizes stable ester or amide precursors that are converted to the nitrile via dehydration (e.g., using thionyl chloride), completely eliminating the need for heavy metal cyanide reagents and significantly reducing environmental hazards.
Q: Can this synthesis be telescoped to reduce processing time?
A: Yes, the patent explicitly describes that the two sequential Grignard reactions can be carried out continuously without isolating the intermediate ketone (Formula IVa). This telescoping capability minimizes unit operations, reduces solvent consumption, and shortens the overall cycle time, making it highly attractive for large-scale commercial production.
Q: What are the advantages of using ester/amide starting materials over bromo-precursors?
A: Ester and amide derivatives of 5-carboxyl-2-benzo[c]furanone are generally more stable and easier to handle than their 5-bromo counterparts. Furthermore, the conversion of these groups to the final nitrile function allows for milder reaction conditions compared to the harsh acidic cyclization required when starting directly from the 5-cyano derivative, thereby preserving the integrity of the sensitive isobenzofuran core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitalapram Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN1268129A to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity Nitalapram intermediates that meet stringent purity specifications, leveraging our rigorous QC labs to monitor every step of the Grignard addition and cyclization process. Our state-of-the-art facilities are equipped to handle the specific safety requirements of organometallic chemistry, guaranteeing a consistent and safe supply of this vital antidepressant precursor.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can optimize your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this copper-free route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production volumes. Let us collaborate to drive down costs and enhance the sustainability of your Nitalapram manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
