Advanced Synthesis of N-Substituted 2-Hydroxy-Morpholin-3-One for Commercial Aprepitant Production
Advanced Synthesis of N-Substituted 2-Hydroxy-Morpholin-3-One for Commercial Aprepitant Production
The pharmaceutical industry constantly seeks robust and cost-effective pathways for synthesizing critical intermediates, particularly for high-value antiemetic drugs like aprepitant. Patent CN102153522B introduces a transformative methodology for preparing N-substituted 2-hydroxy-morpholin-3-one compounds, which serve as pivotal building blocks in this therapeutic class. This innovation addresses long-standing challenges in yield optimization and operational simplicity, offering a distinct advantage over traditional synthetic routes. By leveraging a novel hydrolysis strategy, manufacturers can achieve superior purity profiles essential for downstream API synthesis. The structural versatility of these compounds allows for various substitutions, making them adaptable for diverse medicinal chemistry campaigns. 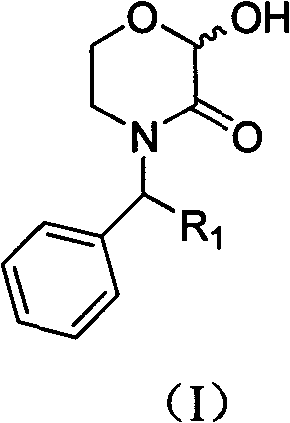 .
.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-benzyl or N-phenylethyl morpholinones has relied on cumbersome multi-step sequences that hinder efficient commercialization. One prevalent prior art method involves the reaction of N-substituted ethanolamines with diethyl oxalate, followed by a reduction step using borohydrides. This approach is fraught with inefficiencies, including low overall yields and the generation of significant chemical waste associated with boron byproducts. Another existing route attempts direct condensation with glyoxylic acid, but this often suffers from poor conversion rates and difficult purification protocols. These legacy methods impose substantial burdens on procurement teams due to the high cost of specialized reducing agents and the complexity of waste management. Furthermore, the operational hazards associated with handling reactive hydride species on a large scale present unnecessary safety risks in a manufacturing environment.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a streamlined acylation-hydrolysis sequence that dramatically simplifies the production landscape. The core innovation lies in the conversion of a dichloroacetamide precursor into the target morpholinone ring system through a base-mediated hydrolysis. 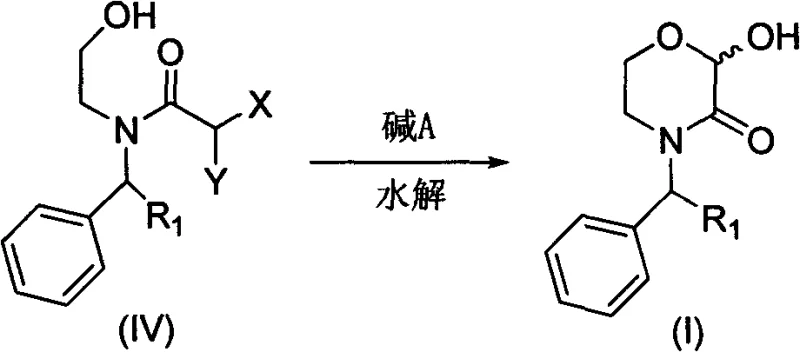 . This transformation proceeds under relatively mild thermal conditions, typically between 20°C and 100°C, utilizing inexpensive inorganic or organic bases such as triethylamine or sodium carbonate. The elimination of stoichiometric reducing agents not only reduces raw material expenses but also simplifies the workup procedure to standard aqueous extractions. This shift represents a paradigm change in process chemistry, moving away from sensitive redox manipulations toward robust nucleophilic substitutions that are inherently safer and more predictable for scale-up operations.
. This transformation proceeds under relatively mild thermal conditions, typically between 20°C and 100°C, utilizing inexpensive inorganic or organic bases such as triethylamine or sodium carbonate. The elimination of stoichiometric reducing agents not only reduces raw material expenses but also simplifies the workup procedure to standard aqueous extractions. This shift represents a paradigm change in process chemistry, moving away from sensitive redox manipulations toward robust nucleophilic substitutions that are inherently safer and more predictable for scale-up operations.
Mechanistic Insights into Base-Catalyzed Cyclization
The chemical elegance of this process is rooted in the intramolecular nucleophilic substitution that drives ring closure. Upon exposure to aqueous base, the dichloroacetamide moiety undergoes hydrolysis, generating a reactive intermediate that facilitates the attack of the pendant hydroxyl group onto the carbonyl carbon. This cyclization event is thermodynamically favored and proceeds with high fidelity, preserving the stereochemical integrity of the chiral centers present in the starting material. For instance, when using chiral N-(1-phenylethyl) ethanolamine, the reaction conditions are sufficiently gentle to prevent racemization, ensuring the production of enantiomerically pure intermediates crucial for biological activity. The use of dichloroacetyl chloride as the acylating agent introduces the necessary leaving groups that activate the system for subsequent ring formation without requiring external activation. 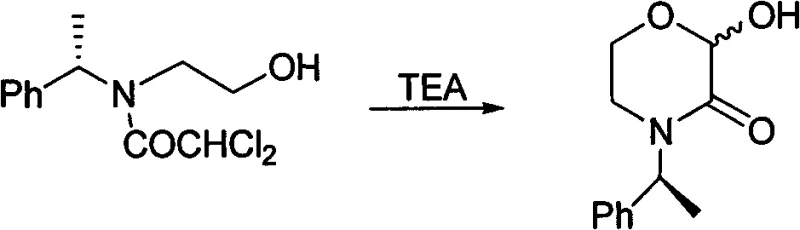 .
.
Impurity control is another critical aspect where this mechanism excels. Traditional reduction pathways often generate alcohol byproducts or over-reduced species that are structurally similar to the target, complicating purification. In this hydrolysis-driven route, the primary byproducts are simple salts and water-soluble species that are easily removed during the aqueous workup phase. The selectivity of the base-catalyzed cyclization minimizes the formation of oligomeric side products, leading to a crude reaction mixture with a significantly higher assay of the desired morpholinone. This inherent cleanliness reduces the load on downstream purification units, such as crystallization or chromatography columns, thereby enhancing the overall throughput of the manufacturing line. The ability to tune the base strength and solvent polarity allows process chemists to further optimize the impurity profile for specific regulatory requirements.
How to Synthesize N-Substituted 2-Hydroxy-Morpholin-3-One Efficiently
Implementing this synthesis requires careful attention to reagent quality and thermal management to maximize yield and optical purity. The process begins with the acylation of the amino alcohol, followed by the critical hydrolysis step that forms the heterocyclic core. Detailed standard operating procedures regarding stoichiometry, addition rates, and temperature ramps are essential for reproducible results. The following guide outlines the generalized workflow derived from the patent examples, providing a roadmap for laboratory and pilot plant execution.
- Acylation of N-substituted ethanolamine with dichloroacetyl chloride in the presence of an organic base like triethylamine.
- Hydrolysis of the resulting dichloroacetamide intermediate using aqueous base at elevated temperatures.
- Purification of the final morpholinone product via recrystallization from mixed solvents such as ethyl acetate and n-hexane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The transition from reduction-based chemistry to hydrolysis-based chemistry fundamentally alters the cost structure of the intermediate. By removing the dependency on expensive and volatile reducing agents, the variable cost of goods sold is significantly reduced. Additionally, the use of commodity chemicals like triethylamine and dichloromethane ensures a stable supply chain不受 geopolitical fluctuations affecting specialized reagents. The simplified workup procedure also translates to shorter cycle times, allowing manufacturing facilities to increase asset utilization and respond more rapidly to market demand spikes.
- Cost Reduction in Manufacturing: The elimination of borohydride reagents removes a major cost driver from the bill of materials, while the simplified aqueous workup reduces solvent consumption and waste disposal fees. This leaner process architecture allows for substantial margin improvement without compromising product quality. The avoidance of complex purification steps further lowers energy consumption and labor costs associated with extended processing times.
- Enhanced Supply Chain Reliability: Relying on widely available bulk chemicals rather than niche reducing agents mitigates the risk of supply disruptions. The robustness of the reaction conditions means that production can be maintained even with minor variations in raw material specifications, ensuring consistent output. This resilience is critical for maintaining continuous supply to downstream API manufacturers who operate on tight just-in-time schedules.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional methods, aligning with increasingly stringent environmental regulations. The absence of heavy metal catalysts or toxic reducing agents simplifies effluent treatment and reduces the environmental footprint of the facility. This green chemistry profile enhances the corporate sustainability rating and facilitates easier regulatory approval for new drug filings.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into existing portfolios. The following questions address common concerns regarding scalability, purity, and regulatory compliance. These answers are derived directly from the experimental data and claims presented in the patent documentation.
Q: What are the advantages of this hydrolysis method over borohydride reduction?
A: This novel hydrolysis method eliminates the need for expensive and hazardous borohydride reducing agents, simplifying the workflow and significantly lowering raw material costs while maintaining high stereochemical integrity.
Q: Is this process suitable for large-scale manufacturing of aprepitant intermediates?
A: Yes, the process utilizes common reagents like triethylamine and water, operates at manageable temperatures, and avoids complex chromatographic purifications, making it highly scalable for industrial production.
Q: How is chirality preserved during the synthesis?
A: The reaction conditions are mild enough to prevent racemization of the chiral center on the phenylethyl group, ensuring the production of optically pure intermediates required for active pharmaceutical ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted 2-Hydroxy-Morpholin-3-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to full-scale manufacturing. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical applications. Our commitment to process excellence means we can deliver the cost and efficiency benefits of this novel hydrolysis method to your supply chain immediately.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized synthesis of N-substituted 2-hydroxy-morpholin-3-one can enhance your competitive position in the global market.
