Advanced Synthesis of Cyclic Phosphate Compounds for High-Performance Battery Electrolytes
Advanced Synthesis of Cyclic Phosphate Compounds for High-Performance Battery Electrolytes
The rapid evolution of the lithium-ion battery industry demands electrolyte additives that can withstand high voltage and high-temperature operating conditions without compromising safety or cycle life. Patent CN114989219B introduces a groundbreaking preparation method for cyclic phosphate compounds that addresses these critical needs through a novel three-step synthetic route. This technology leverages a mild cyclization reaction between diol compounds and phosphorus trichloride, followed by a controlled substitution and a unique light-assisted oxidation process. By eliminating the generation of corrosive hydrogen chloride gas during the final oxidation stage, this method significantly enhances product stability and overall yield. For R&D directors and procurement specialists in the energy storage sector, this patent represents a viable pathway to sourcing high-purity battery & energy storage materials that ensure long-term cell reliability and performance consistency in next-generation electric vehicles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for vinyl phosphate and cyclic phosphate additives often suffer from violent reaction conditions that are difficult to control on a commercial scale. Conventional oxidation methods frequently generate hydrogen chloride gas as a byproduct, which poses a severe risk of causing ring-opening reactions in the sensitive phosphate structure. This side reaction not only drastically reduces the final yield of the target compound but also introduces difficult-to-remove impurities that can degrade battery performance. Furthermore, the harsh conditions required in older methodologies often necessitate expensive corrosion-resistant equipment and complex waste gas treatment systems, driving up the overall cost of manufacturing. These technical bottlenecks have historically limited the availability of high-quality electrolyte additives, creating supply chain vulnerabilities for battery manufacturers seeking to scale production of high-energy-density cells.
The Novel Approach
The methodology disclosed in CN114989219B overcomes these historical challenges by implementing a温和 (mild) reaction sequence that prioritizes structural integrity and process safety. The core innovation lies in the final oxidation step, which utilizes oxygen or air under specific light irradiation conditions rather than harsh chemical oxidants that produce acidic byproducts. This approach ensures that the cyclic phosphate ring remains intact throughout the synthesis, leading to substantially higher yields and easier purification. The use of common organic solvents such as dichloromethane, toluene, and tetrahydrofuran further simplifies the process infrastructure, making it highly adaptable for existing chemical manufacturing facilities. By shifting the reaction paradigm from aggressive chemical oxidation to controlled photo-oxidation, this technology offers a robust solution for the cost reduction in battery & energy storage materials manufacturing while maintaining exceptional product quality.
Mechanistic Insights into Photo-Oxidized Cyclization
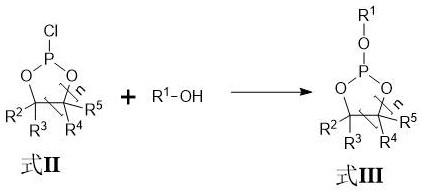
The synthesis begins with a precise cyclization reaction where a diol compound reacts with phosphorus trichloride to form a chlorophosphate intermediate. This step is carefully controlled at temperatures between -10°C and 50°C to manage the exothermic nature of the reaction and ensure complete conversion of the starting materials. The resulting intermediate serves as a stable scaffold for the subsequent introduction of functional alkoxy groups, which are critical for the additive's performance in the electrolyte. The ability to fine-tune the molar ratio of phosphorus trichloride to diol allows manufacturers to optimize the reaction kinetics, minimizing the formation of oligomeric byproducts that could complicate downstream purification. This foundational step sets the stage for a high-efficiency process that maximizes atom economy and reduces raw material waste.
Following the substitution reaction, the critical oxidation step converts the trivalent phosphorus intermediate into the final pentavalent cyclic phosphate compound. Unlike traditional methods, this process employs light irradiation with an intensity of 100 W to 400 W in the presence of oxygen, which facilitates the oxidation without generating hydrogen chloride gas. 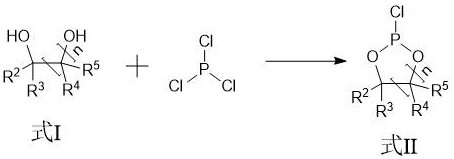
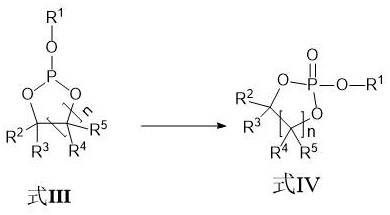 This mechanism effectively prevents the acid-catalyzed ring-opening degradation that plagues conventional synthesis, thereby preserving the structural integrity of the additive. The absence of corrosive gas byproducts also simplifies the engineering requirements for the reactor system, reducing capital expenditure and maintenance costs. The final product is purified through vacuum distillation, achieving purity levels exceeding 99% with metal ion content controlled to less than 1 ppm, ensuring compatibility with sensitive battery electrode materials.
This mechanism effectively prevents the acid-catalyzed ring-opening degradation that plagues conventional synthesis, thereby preserving the structural integrity of the additive. The absence of corrosive gas byproducts also simplifies the engineering requirements for the reactor system, reducing capital expenditure and maintenance costs. The final product is purified through vacuum distillation, achieving purity levels exceeding 99% with metal ion content controlled to less than 1 ppm, ensuring compatibility with sensitive battery electrode materials.
How to Synthesize Cyclic Phosphate Compound Efficiently
The standardized production of these high-performance electrolyte additives requires strict adherence to the three-step protocol outlined in the patent data to ensure consistent quality and safety. Operators must maintain precise temperature controls during the addition of phosphorus trichloride and manage the light intensity during the oxidation phase to maximize conversion rates. The process is designed to be scalable, utilizing standard unit operations such as distillation and filtration that are common in fine chemical manufacturing plants. Detailed standard operating procedures regarding solvent selection, reaction times, and workup protocols are essential for replicating the high yields reported in the patent examples. For a comprehensive guide on the specific parameters and safety measures required for implementation, please refer to the technical documentation below.
- Cyclization of diol compounds with phosphorus trichloride at controlled low temperatures to form chlorophosphate intermediates.
- Substitution reaction with hydroxyl compounds to introduce specific alkoxy groups under mild conditions.
- Photo-oxidation using oxygen and light irradiation to convert phosphite to phosphate without generating corrosive HCl gas.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers substantial benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for battery materials. The use of readily available starting materials like diols and phosphorus trichloride, combined with common organic solvents, ensures a stable and resilient supply chain that is less susceptible to raw material shortages. The mild reaction conditions reduce the need for specialized high-pressure or high-temperature equipment, lowering the barrier to entry for contract manufacturing organizations and increasing the number of potential suppliers in the market. This diversification of the supply base enhances supply chain reliability and provides buyers with greater leverage in negotiations. Furthermore, the simplified purification process reduces energy consumption and waste generation, aligning with increasingly stringent environmental regulations and corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous oxidizing agents in favor of oxygen and light significantly lowers the raw material costs associated with the oxidation step. Additionally, the prevention of ring-opening side reactions means that less raw material is wasted on byproducts, directly improving the overall material efficiency of the process. The ability to use standard distillation for purification rather than complex chromatographic techniques further reduces operational expenditures related to solvents and energy. These cumulative efficiencies translate into a more competitive cost structure for the final electrolyte additive, allowing battery manufacturers to achieve cost reduction in battery & energy storage materials manufacturing without sacrificing performance.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard reaction conditions makes this process highly robust against supply chain disruptions. Manufacturers can source the necessary diols and phosphorus reagents from multiple global suppliers, reducing the risk of single-source dependency. The mild nature of the reaction also implies safer transportation and storage requirements for intermediates, facilitating smoother logistics operations. By adopting this technology, companies can secure a more consistent supply of high-purity battery & energy storage materials, ensuring that production schedules for electric vehicle batteries are met without delay.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, with reaction parameters that can be easily translated from laboratory to pilot and full-scale production. The absence of corrosive hydrogen chloride gas during the critical oxidation step simplifies waste gas treatment, reducing the environmental footprint of the manufacturing facility. This compliance with environmental standards minimizes the risk of regulatory fines and operational shutdowns, ensuring long-term business continuity. The high purity of the final product also reduces the burden on battery cell manufacturers to perform additional purification, streamlining the overall value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this cyclic phosphate synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide accurate guidance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this additive into your existing electrolyte formulations. For further technical clarification or custom synthesis requests, our team is available to provide detailed assessments.
Q: How does this synthesis method improve yield compared to traditional oxidation?
A: By utilizing light-assisted oxidation with oxygen, the process avoids the generation of hydrogen chloride gas, which traditionally causes ring-opening reactions and significantly reduces yield.
Q: What purity levels can be achieved with this purification protocol?
A: The patent data indicates that through vacuum distillation and rectification, purity levels exceeding 99% can be consistently achieved, with metal ion content controlled below 1 ppm.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of common organic solvents, mild reaction temperatures ranging from 0°C to 80°C, and standard distillation purification makes the route highly scalable and safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclic Phosphate Compound Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise to translate complex patent methodologies like CN114989219B into commercial reality. Our engineering team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of light-assisted oxidation and distillation is maintained at scale. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of cyclic phosphate compound meets the exacting standards required for lithium-ion battery applications. Our commitment to quality assurance means that you receive a product with consistent impurity profiles, crucial for maintaining the cycle life and safety of your battery cells.
We invite you to collaborate with us to optimize your electrolyte supply chain and leverage the cost advantages of this advanced synthesis route. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your production goals. By partnering with us, you gain access to a reliable supply of high-performance additives that will drive the success of your next-generation energy storage solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
