Advanced Fermentation Technology For High-Purity Nosiheptide Production And Commercial Scale-Up
Advanced Fermentation Technology For High-Purity Nosiheptide Production And Commercial Scale-Up
The global demand for efficient, non-absorbable feed additives has driven significant innovation in the biosynthesis of thiopeptide antibiotics. Patent CN1840684A introduces a breakthrough methodology utilizing a novel strain, Streptomyces glaucogriseus HSC-SN-1-52 (CCTCC No.M 205133), to achieve superior nosiheptide titers. This technology represents a pivotal shift from static fermentation protocols to dynamic, fed-batch processes that optimize precursor availability. For R&D directors and procurement specialists, understanding this pathway is crucial for securing a reliable nosiheptide supplier capable of meeting stringent quality and volume requirements. The patent details a comprehensive approach involving specific medium formulations, precise pH control, and strategic supplementation of sulfur sources and amino acids, resulting in yields exceeding 1500mg/L in tank fermentation. This level of productivity underscores the viability of the process for commercial scale-up of complex polypeptide antibiotics, ensuring a stable supply chain for the agrochemical and veterinary sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of nosiheptide relied on strains such as Streptomyces actuosus or Streptomyces antibioticus, which often exhibited metabolic limitations under standard fermentation conditions. Traditional methods typically employed static media compositions that failed to account for the dynamic nutrient consumption rates of the microorganism during different growth phases. This often led to premature depletion of critical precursors, particularly sulfur-containing compounds necessary for the formation of the characteristic thiazole rings found in the nosiheptide structure. Furthermore, conventional downstream processing frequently utilized less efficient solvent extraction techniques that resulted in lower recovery rates and higher impurity profiles. The lack of precise control over feeding strategies meant that potential yields were left unrealized, driving up the cost per kilogram and creating supply bottlenecks for manufacturers seeking cost reduction in feed additive manufacturing. These inefficiencies also complicated the purification steps, requiring more rigorous and costly refinement to meet regulatory standards for animal health products.
The Novel Approach
The methodology outlined in CN1840684A addresses these historical inefficiencies through a sophisticated fed-batch fermentation strategy. By isolating and utilizing the high-producing Streptomyces glaucogriseus HSC-SN-1-52 strain, the process establishes a robust biological foundation for enhanced synthesis. The core innovation lies in the intermittent supplementation of potassium sulfate and specific amino acids like silk fibroin peptide at critical time points (72, 96, and 120 hours). This targeted feeding ensures that the biosynthetic machinery of the bacteria remains saturated with necessary building blocks throughout the production phase, preventing metabolic stalling. Additionally, the integration of countercurrent chromatography for purification offers a distinct advantage over traditional column chromatography, providing higher resolution separation with reduced solvent consumption. This holistic approach not only maximizes the titer but also streamlines the downstream workflow, offering a compelling value proposition for partners looking for high-purity nosiheptide with improved process economics.
Mechanistic Insights into Sulfur-Enhanced Polypeptide Biosynthesis
Nosiheptide is a complex sulfur-containing polypeptide characterized by a central pyridine ring substituted with multiple thiazole rings and an indole moiety. The biosynthesis of such a intricate molecule is a non-ribosomal process that demands a precise supply of specific amino acid precursors, including serine, cysteine, tryptophan, and threonine. The structural complexity requires the enzymatic cyclization of these linear precursors into heterocyclic units, a process heavily dependent on the availability of sulfur. In the context of the patented fermentation process, the addition of exogenous sulfur sources directly supports the formation of these thiazole rings, which are critical for the antibiotic activity and structural integrity of the final molecule.
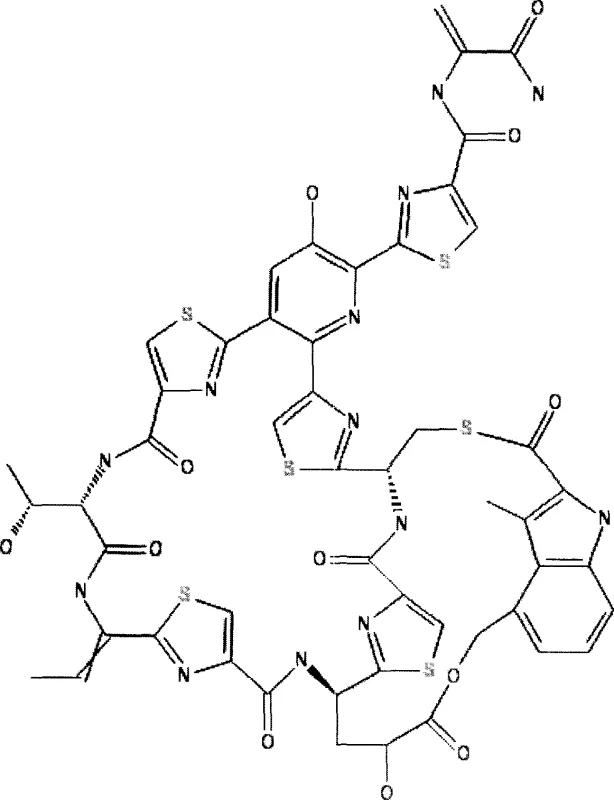
The mechanism involves the activation of amino acids by specific synthetases, followed by condensation and cyclization reactions. The patent highlights that serine serves as a precursor for both the pyridine core and dehydroalanine residues, while cysteine contributes to the thiazole rings. By maintaining an optimal concentration of sulfur ions through intermittent feeding, the fermentation environment mimics an ideal metabolic state where the rate-limiting steps associated with sulfur assimilation are bypassed. This mechanistic understanding allows for better control over the impurity profile, as imbalanced precursor ratios often lead to the accumulation of incomplete peptide fragments or analogues. Consequently, this process ensures a cleaner crude product, facilitating easier purification and higher final purity specifications essential for regulatory compliance in veterinary applications.
How to Synthesize Nosiheptide Efficiently
The synthesis of nosiheptide via this optimized fermentation route requires strict adherence to sterile techniques and precise environmental controls to maximize the genetic potential of the Streptomyces glaucogriseus strain. The process begins with the activation of the strain on slant media, followed by a seed train expansion to ensure a vigorous inoculum. The fermentation stage is the critical control point, where parameters such as dissolved oxygen, agitation speed, and pH must be meticulously monitored. The strategic addition of nutrients is not arbitrary but is timed to coincide with the secondary metabolite production phase of the bacteria. Detailed standardized synthetic steps for implementing this protocol are provided in the guide below.
- Activate the Streptomyces glaucogriseus HSC-SN-1-52 strain on slant media at 28°C for 3-5 days, followed by seed culture expansion in shake flasks containing starch and soybean powder.
- Inoculate the fermentation tank with a 5-10% seed load into a medium comprising corn starch, glucose, soybean powder, and calcium carbonate, maintaining pH between 7.2 and 7.6.
- Implement intermittent feeding of potassium sulfate and silk fibroin peptide at 72, 96, and 120 hours to boost titers, followed by ethanol extraction and countercurrent chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this advanced fermentation technology translates into tangible operational benefits beyond simple yield metrics. The primary advantage lies in the significant enhancement of process efficiency, which directly impacts the cost structure of the final active ingredient. By achieving higher titers per batch, the fixed costs associated with fermentation tank usage, sterilization, and labor are amortized over a larger output volume. This efficiency gain is critical for maintaining competitiveness in the global market for feed additives, where margin pressures are constant. Furthermore, the robustness of the Streptomyces glaucogriseus strain reduces the risk of batch failures, ensuring greater supply continuity and reliability for long-term contracts.
- Cost Reduction in Manufacturing: The implementation of intermittent feeding strategies eliminates the need for excessively rich initial media, which can be cost-prohibitive and lead to osmotic stress on the cells. By adding expensive precursors like specific amino acids only when needed, the process minimizes waste and raw material costs. Additionally, the high recovery rate achieved through countercurrent chromatography reduces the loss of product during purification, further driving down the effective cost of goods sold. This lean manufacturing approach allows for substantial cost savings without compromising on the quality or potency of the nosiheptide produced.
- Enhanced Supply Chain Reliability: The scalability of this fermentation process from shake flasks to 10-liter tanks and beyond demonstrates its readiness for industrial deployment. The use of readily available raw materials such as corn starch, soybean powder, and common inorganic salts ensures that the supply chain is not vulnerable to the volatility of exotic reagent markets. This accessibility of inputs guarantees that production schedules can be maintained consistently, reducing lead time for high-purity nosiheptide batches. For supply chain planners, this means a more predictable inventory flow and the ability to respond swiftly to fluctuations in market demand for animal health products.
- Scalability and Environmental Compliance: The process design inherently supports environmental sustainability by optimizing resource utilization. The high conversion efficiency means less organic waste is generated per unit of product, simplifying wastewater treatment requirements. Moreover, the use of countercurrent chromatography typically involves lower solvent volumes compared to traditional flash chromatography, reducing the environmental footprint of the purification stage. These factors contribute to a more sustainable manufacturing profile, aligning with the increasing regulatory and corporate social responsibility demands placed on chemical manufacturers today.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of nosiheptide based on the patented technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this material into their product portfolios.
Q: What distinguishes the Streptomyces glaucogriseus HSC-SN-1-52 strain from conventional nosiheptide producers?
A: This specific strain, deposited as CCTCC No.M 205133, demonstrates superior biosynthetic capability when combined with a specialized feeding strategy. Unlike older strains that may suffer from metabolic bottlenecks, this variant supports higher titers up to 1700mg/L in shake flask cultures through optimized precursor uptake.
Q: How does the intermittent supplementation of sulfur sources impact the fermentation yield?
A: Nosiheptide is a sulfur-rich polypeptide containing multiple thiazole rings. The patent data indicates that adding potassium sulfate at critical growth phases (72h, 96h, 120h) prevents sulfur limitation, directly correlating to increased antibiotic synthesis and overall process efficiency without requiring excessive initial nutrient loads.
Q: Is the purification process scalable for industrial manufacturing of feed additives?
A: Yes, the process utilizes countercurrent chromatography (CCC) following ethanol extraction. This method offers high recovery rates (over 78%) and effectively removes impurities, making it suitable for large-scale production where consistent purity and cost-efficiency are paramount for regulatory compliance in animal nutrition.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nosiheptide Supplier
The technological advancements detailed in patent CN1840684A highlight the immense potential of optimized microbial fermentation for producing complex thiopeptide antibiotics. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of nosiheptide meets the exacting standards required for veterinary and agrochemical applications. We understand the critical nature of supply chain stability and are committed to delivering consistent quality.
We invite you to collaborate with us to optimize your supply chain for this vital feed additive. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and formulation needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, high-quality source of nosiheptide that supports your commitment to animal health and productivity.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
