Revolutionizing Beta-Damascone Production: A Safer, Scalable Synthetic Route for Global Fragrance Supply Chains
Revolutionizing Beta-Damascone Production: A Safer, Scalable Synthetic Route for Global Fragrance Supply Chains
The global demand for high-quality terpenoid fragrances continues to surge, driven by the premiumization of personal care and fine fragrance markets. At the heart of this sector lies beta-damascone, a molecule renowned for its intense, elegant rose-like aroma that serves as a cornerstone for countless luxury compositions. However, the historical reliance on hazardous and complex synthetic pathways has often constrained supply reliability and inflated costs for procurement teams worldwide. A groundbreaking technical disclosure found in patent CN111440055B offers a transformative solution, detailing a streamlined three-step synthesis that bypasses traditional bottlenecks. This report analyzes the technical merits of this novel approach, providing critical insights for R&D directors seeking purity, procurement managers targeting cost reduction in fragrance intermediate manufacturing, and supply chain leaders focused on continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of beta-damascone has been plagued by significant operational challenges that impact both safety and economics. As illustrated in prior art such as US4250332A and other legacy routes, conventional methodologies frequently depend on the use of highly reactive organometallic reagents like Grignard reagents or metal hydrides. These reagents are not only prohibitively expensive due to the cost of magnesium and specialized handling requirements but also introduce severe safety liabilities. They are notoriously pyrophoric and extremely sensitive to moisture, necessitating rigorous anhydrous conditions that complicate reactor design and increase energy consumption for drying solvents. Furthermore, the workup procedures for quenching these reactive species generate substantial volumes of hazardous waste, creating environmental compliance burdens and disposal costs that erode profit margins.
The Novel Approach
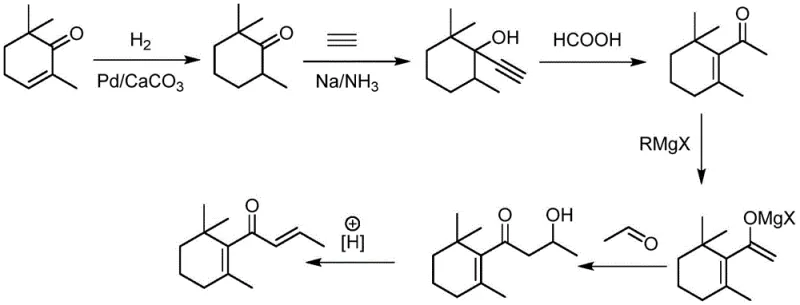
In stark contrast, the methodology disclosed in CN111440055B introduces a paradigm shift by utilizing readily available, stable reagents to construct the carbon skeleton efficiently. The core innovation lies in the direct nucleophilic addition of acetylene to 2,6,6-trimethylcyclohexanone under basic catalysis in liquid ammonia, followed by a telescoped condensation with acetaldehyde. This strategy completely eliminates the need for transition metal catalysts in the carbon-carbon bond-forming steps and avoids pyrophoric reagents entirely. By leveraging simple alkali metal hydroxides or amides, the process achieves remarkable conversion rates exceeding 98% while maintaining selectivity above 97%. This simplification of the reaction matrix not only enhances the safety profile dramatically but also facilitates a much cleaner reaction mixture, thereby reducing the complexity of downstream purification and significantly lowering the total cost of ownership for manufacturers.
Mechanistic Insights into Alkynylation and Meyer-Schuster Rearrangement
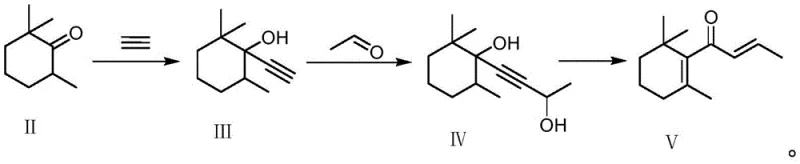
From a mechanistic perspective, the success of this route hinges on the precise control of the alkynylation step and the subsequent acid-catalyzed rearrangement. The initial reaction involves the generation of an acetylide anion in situ within a liquid ammonia solvent system, which acts as both a solvent and a reactant medium. The use of alkali metal catalysts such as potassium hydroxide or sodium amide allows for the deprotonation of acetylene under moderate pressure (0.5-3.0 MPa) and temperature (0-60°C). This generates a highly nucleophilic species that attacks the carbonyl carbon of the cyclohexanone derivative with high regioselectivity. Crucially, the patent details a clever catalyst modification strategy where a weak acidic aqueous solution is introduced directly into the reaction system. This subtle pH adjustment neutralizes excess base just enough to prevent the decomposition of the sensitive propargylic alcohol intermediate (Compound III) while retaining sufficient catalytic activity for the subsequent condensation with acetaldehyde, showcasing a sophisticated understanding of reaction kinetics.
The final transformation relies on the Meyer-Schuster rearrangement, a powerful tool for converting propargylic alcohols into alpha,beta-unsaturated ketones. In this specific application, the use of inorganic or organic acids like phosphoric acid or formic acid promotes the migration of the triple bond and hydration to form the conjugated enone system characteristic of beta-damascone. The patent specifies reaction temperatures between 40°C and 120°C, which are mild enough to prevent polymerization of the sensitive diene-one system yet energetic enough to drive the equilibrium towards the thermodynamic product. Impurity control is inherently built into this mechanism; by avoiding harsh oxidizing agents or radical initiators common in older routes, the formation of over-oxidized byproducts or ring-opened degradation products is minimized. This results in a crude product profile that is exceptionally clean, allowing for high-purity beta-damascone to be isolated via standard distillation techniques without the need for complex chromatographic separations.
How to Synthesize Beta-Damascone Efficiently
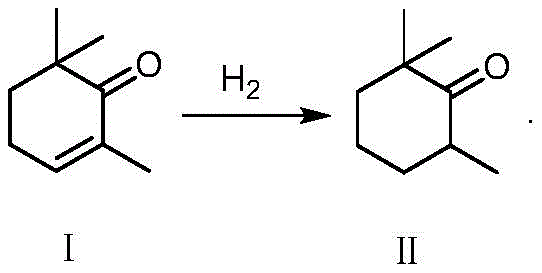
Implementing this synthesis requires careful attention to the preparation of the starting material, 2,6,6-trimethylcyclohexanone (Compound II), which serves as the foundational building block for the entire sequence. The patent outlines a robust hydrogenation protocol where 2,6,6-trimethylcyclohexenone is reduced using hydrogen gas over a Lindlar-type palladium catalyst supported on calcium carbonate. This step is critical because it ensures the saturation of the ring double bond without affecting the carbonyl group, achieving conversion rates above 99% and selectivity exceeding 98%. Following this precursor preparation, the main synthetic sequence proceeds through the acetylene addition and rearrangement steps described previously. The detailed standardized synthesis steps below outline the specific operational parameters, including pressure ranges, molar ratios, and workup procedures necessary to replicate the high yields reported in the patent data, serving as a practical guide for process chemists aiming to adopt this technology.
- Perform nucleophilic addition of acetylene to 2,6,6-trimethylcyclohexanone (Compound II) using alkali metal hydroxide or amide catalyst in liquid ammonia at 0-60°C and 0.5-3.0 MPa to generate Compound III.
- Modify the catalyst system by adding a weak acidic aqueous solution, then react Compound III with acetaldehyde at 0-60°C to form the propargylic alcohol intermediate (Compound IV).
- Execute the Meyer-Schuster rearrangement of Compound IV using inorganic or organic acids (e.g., phosphoric acid, formic acid) at 40-120°C to yield high-purity Beta-Damascone (Compound V).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling strategic advantages that extend far beyond simple yield improvements. The primary value driver is the drastic simplification of the raw material portfolio. By replacing expensive, specialty organometallic reagents with commodity chemicals like acetylene, acetaldehyde, ammonia, and caustic soda, the direct material costs are significantly reduced. Furthermore, the stability of these reagents allows for bulk purchasing and long-term storage without the degradation issues associated with moisture-sensitive Grignard solutions. This shift transforms the supply chain from a fragile, just-in-time model dependent on specialized vendors into a robust, inventory-friendly system capable of weathering market volatility. The elimination of hazardous reagent handling also translates to lower insurance premiums and reduced regulatory compliance overhead, contributing to substantial overall cost savings in fragrance intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from multiple vectors, primarily the substitution of high-cost catalysts with inexpensive alkali bases. Traditional routes often require stoichiometric amounts of magnesium or lithium reagents, whereas this method uses catalytic quantities of potassium hydroxide or sodium amide. Additionally, the simplified workup procedure, which involves straightforward extraction and distillation rather than complex quenching and filtration of metal salts, reduces labor hours and utility consumption. The high selectivity of the reaction minimizes the loss of valuable starting materials into waste streams, effectively maximizing the atom economy of the process and driving down the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any reliable beta-damascone supplier, and this technology directly addresses common disruption points. The reagents employed are globally available commodity chemicals with mature supply chains, reducing the risk of shortages that often plague specialty reagent markets. Moreover, the process operates under moderate conditions that are compatible with standard multipurpose chemical reactors, meaning production does not require dedicated, exotic equipment that could become a single point of failure. This flexibility allows manufacturers to easily shift production between different facilities or scale up capacity rapidly in response to surging market demand without lengthy lead times for equipment fabrication.
- Scalability and Environmental Compliance: As the industry moves towards greener manufacturing practices, this route offers a distinct advantage in terms of environmental footprint. The absence of heavy metal catalysts in the key bond-forming steps eliminates the need for expensive and difficult heavy metal scavenging processes, ensuring the final product meets stringent purity specifications for food and cosmetic applications. The waste stream is predominantly composed of benign salts and aqueous ammonia, which are easier to treat and dispose of compared to the toxic sludge generated by organometallic quenches. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the brand value of the end-product for eco-conscious consumers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-damascone synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the underlying patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the competitive positioning of the resulting product in the global marketplace.
Q: How does this new synthesis method improve safety compared to traditional Grignard routes?
A: Traditional methods rely on pyrophoric Grignard reagents or metal hydrides which pose significant fire and explosion risks. This patented route utilizes stable alkali metal hydroxides or amides in liquid ammonia, drastically reducing operational hazards and eliminating the need for stringent moisture-free environments required for organometallics.
Q: What are the expected conversion rates and selectivity for the key intermediates?
A: The process demonstrates exceptional efficiency. The initial acetylene addition step achieves over 98% conversion of the ketone with greater than 97% selectivity for the alkynyl alcohol. Subsequent condensation and rearrangement steps maintain high conversion rates above 97% and 98% respectively, ensuring minimal waste and high overall yield.
Q: Can this process be scaled for industrial production of fragrance intermediates?
A: Yes, the protocol is designed for scalability. It operates at moderate temperatures (0-120°C) and pressures (0.5-3.0 MPa) compatible with standard stainless steel reactors. The elimination of sensitive reagents allows for robust continuous or batch processing, making it ideal for commercial scale-up of complex terpene intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Damascone Supplier
The technical potential of this synthesis route represents a significant opportunity for the fine chemical industry, yet translating patent claims into commercial reality requires deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this translation, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team of expert process chemists is adept at optimizing reaction parameters such as temperature gradients and mixing efficiencies to ensure that the high selectivity observed in the lab is maintained at the plant scale. We operate state-of-the-art rigorous QC labs equipped with advanced analytical instrumentation to monitor every batch, ensuring that our output consistently meets stringent purity specifications required by top-tier fragrance houses and flavor manufacturers globally.
We invite procurement leaders and R&D directors to engage with us to explore how this advanced synthesis method can optimize your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data for our beta-damascone batches and to discuss route feasibility assessments for your broader portfolio. Let us demonstrate how our commitment to innovation and quality can become a cornerstone of your long-term sourcing strategy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
