Advanced Nickel-Catalyzed Synthesis of Chiral Alpha-Aryl Phosphates for Commercial Scale-Up
Advanced Nickel-Catalyzed Synthesis of Chiral Alpha-Aryl Phosphates for Commercial Scale-Up
The landscape of asymmetric synthesis is constantly evolving, driven by the demand for more efficient and sustainable pathways to access complex chiral building blocks. A significant breakthrough in this domain is documented in Chinese Patent CN114315894B, which discloses a novel methodology for the construction of chiral alpha-aryl phosphates and chiral alpha-aryl phosphine compounds. These structural motifs are pivotal in the development of new ligands and pharmaceutically active molecules, yet their synthesis has historically been fraught with challenges regarding stereocontrol and reaction efficiency. The patent introduces a robust dual catalytic system merging nickel catalysis with photoredox chemistry, enabling the direct cross-coupling of aryl halides with alpha-bromophosphates. This innovation not only fills a critical gap in the literature regarding the efficient construction of these scaffolds but also provides a versatile platform for generating diverse libraries of chiral organophosphorus compounds essential for modern drug discovery.
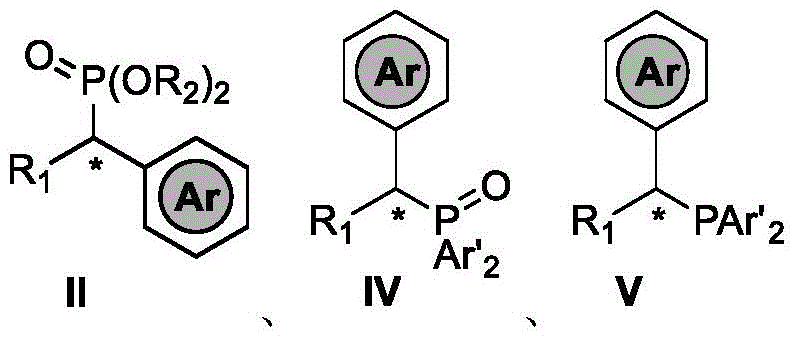
For R&D directors and process chemists, the ability to access these structures reliably is paramount. The disclosed compounds, represented by general formulas II, IV, and V, encompass a wide range of substituents where R1 and R2 can be alkyl or aryl groups, and Ar represents various aryl moieties. This structural flexibility allows for the fine-tuning of electronic and steric properties, which is crucial when designing ligands for asymmetric catalysis or optimizing the pharmacokinetic profiles of API candidates. By leveraging this patented technology, manufacturers can overcome the synthetic bottlenecks that have previously limited the availability of high-purity chiral phosphates in the supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral organophosphorus compounds has relied heavily on stoichiometric chiral auxiliaries or the resolution of racemic mixtures, both of which suffer from inherent inefficiencies. Classical methods often require harsh reaction conditions, such as elevated temperatures and strong bases, which can lead to the decomposition of sensitive functional groups and poor atom economy. Furthermore, many existing protocols depend on expensive precious metal catalysts like palladium or iridium, which not only drive up the raw material costs but also introduce stringent requirements for residual metal removal in pharmaceutical applications. The lack of a general, efficient method for constructing the chiral carbon-phosphorus bond directly from simple precursors has been a long-standing obstacle, limiting the exploration of this chemical space in medicinal chemistry and materials science.
The Novel Approach
The methodology outlined in patent CN114315894B represents a paradigm shift by employing a synergistic nickel/photoredox catalytic system. This approach facilitates the reductive cross-coupling of readily available aryl halides and alpha-bromophosphates under remarkably mild conditions. Unlike traditional thermal processes, this reaction is driven by visible light irradiation, typically using blue LEDs, which activates the photocatalyst to mediate single-electron transfer events. This strategy avoids the need for extreme temperatures and allows for the preservation of delicate functional groups that would otherwise be compromised. The reaction scheme below illustrates the core transformation, highlighting the convergence of the alpha-bromophosphate and the aryl iodide to form the desired chiral product with high stereochemical fidelity.
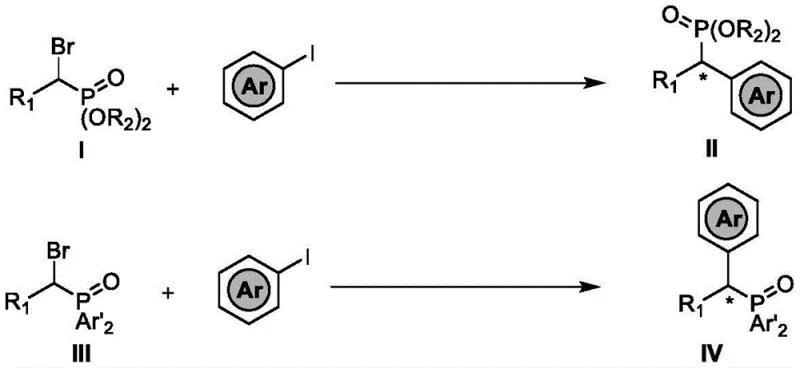
By utilizing this novel approach, manufacturers can achieve cost reduction in pharmaceutical intermediate manufacturing through several mechanisms. The use of nickel, an earth-abundant metal, significantly lowers catalyst costs compared to noble metals. Additionally, the mild reaction conditions reduce energy consumption and minimize the formation of by-products, thereby simplifying downstream purification processes. This efficiency translates directly into a more reliable supply chain for high-value chiral intermediates, ensuring consistent quality and availability for downstream applications.
Mechanistic Insights into Ni/Photo-Redox Dual Catalysis
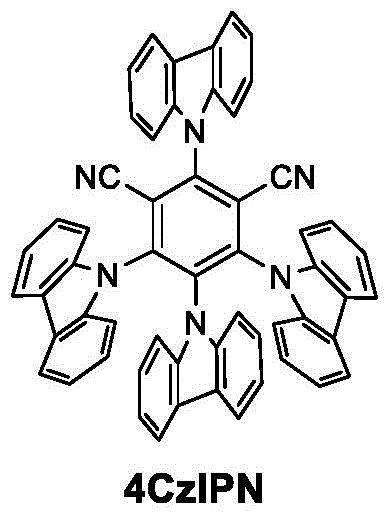
The success of this transformation lies in the intricate interplay between the nickel catalyst and the organic photosensitizer. The cycle initiates with the excitation of the photosensitizer, such as 4CzIPN (2,4,5,6-tetra(9-carbazolyl)-isophthalonitrile), upon irradiation with blue light. This excited state species acts as a potent reductant or oxidant, facilitating the generation of radical intermediates from the substrates. Specifically, the photosensitizer mediates the reduction of the nickel center, cycling it between different oxidation states (Ni(0), Ni(I), Ni(II), Ni(III)) necessary for the oxidative addition of the aryl halide and the subsequent capture of the radical generated from the alpha-bromophosphate.
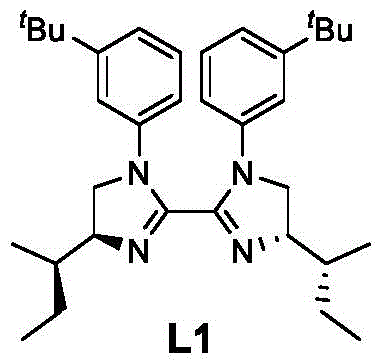
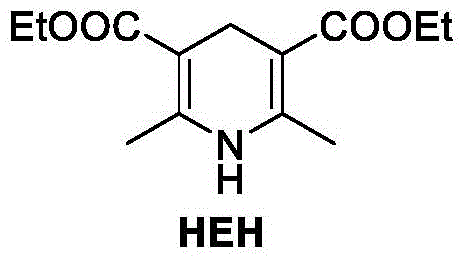
Crucial to the enantioselectivity of the reaction is the chiral ligand, such as the bisoxazole derivative L1 shown below. This ligand creates a chiral environment around the nickel center, effectively discriminating between the prochiral faces of the intermediate species during the bond-forming step. The precise spatial arrangement enforced by the ligand ensures that the coupling occurs with high stereocontrol, yielding products with excellent enantiomeric excess (ee) values, often exceeding 90%. The reducing agent, typically diethyl 2,6-dimethyl-1,4-dihydro-3,5-pyridinedicarboxylate (HEH), plays a vital role in regenerating the active catalytic species and maintaining the redox balance of the system.
Impurity control is inherently managed by the specificity of the catalytic cycle. The mild conditions prevent thermal degradation pathways, while the rapid turnover of the catalyst minimizes the residence time of reactive intermediates that could lead to side reactions. The compatibility of the system with various bases, such as cesium carbonate, and solvents like 1,4-dioxane/DMA mixtures, further optimizes the reaction profile, ensuring that the final product meets the stringent purity specifications required for high-purity pharmaceutical intermediates.
How to Synthesize Chiral Alpha-Aryl Phosphate Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the preparation of the catalytic mixture and the control of reaction parameters. The process begins with the assembly of the catalyst system under an inert atmosphere to prevent oxidation of the sensitive nickel species. Once the catalytic cocktail is prepared, the substrates are introduced, and the reaction is driven by light energy rather than thermal energy. This distinct feature allows for precise control over the reaction kinetics. For a detailed breakdown of the operational parameters, including specific molar ratios and workup procedures, please refer to the standardized synthesis guide provided below.
- Prepare the catalytic system by mixing photosensitizer (e.g., 4CzIPN), nickel catalyst (e.g., NiBr2·DME), chiral ligand (L1), base, and reducing agent in an organic solvent under inert atmosphere.
- Add alpha-bromophosphate and aryl iodide substrates to the reaction mixture and irradiate with blue light (450-455 nm) at 15-30°C for 8-12 hours to facilitate cross-coupling.
- Quench the reaction with water, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify the crude product via column chromatography to obtain the high-purity chiral phosphate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this nickel-catalyzed photoredox methodology offers substantial strategic benefits. The shift away from precious metal catalysts to nickel-based systems fundamentally alters the cost structure of the synthesis. Nickel salts are significantly more abundant and less expensive than palladium or iridium complexes, leading to a drastic reduction in raw material expenditures. Furthermore, the elimination of expensive chiral ligands often associated with noble metal catalysis, in favor of more accessible chiral diamines or bisoxazoles, contributes to overall cost optimization. This economic efficiency makes the production of complex chiral phosphates commercially viable on a larger scale.
- Cost Reduction in Manufacturing: The utilization of earth-abundant nickel catalysts and organic photosensitizers eliminates the dependency on volatile precious metal markets. This stability in raw material pricing allows for more accurate long-term budgeting and reduces the risk of supply disruptions caused by geopolitical factors affecting rare metal mining. Additionally, the high atom economy of the cross-coupling reaction minimizes waste generation, lowering the costs associated with waste disposal and environmental compliance.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining supply continuity. The tolerance of the method to various functional groups means that a wider range of starting materials can be sourced without requiring extensive protection-deprotection sequences. This flexibility simplifies the sourcing strategy and reduces the lead time for acquiring specialized precursors, thereby enhancing the overall agility of the supply chain.
- Scalability and Environmental Compliance: The mild reaction temperatures (15-30°C) and the use of visible light make this process highly amenable to scale-up. Unlike exothermic thermal reactions that require complex cooling systems at scale, this photochemical process can be managed with standard LED arrays, reducing energy consumption. The reduced need for hazardous reagents and the generation of fewer toxic by-products align with green chemistry principles, facilitating easier regulatory approval and minimizing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial aspects of this technology, we have compiled the following answers based on the detailed disclosures in patent CN114315894B. These insights are intended to clarify the operational feasibility and the specific advantages of this synthetic route for potential partners and stakeholders evaluating its integration into their production workflows.
Q: What are the primary advantages of this nickel-catalyzed method over traditional synthesis?
A: This method utilizes abundant nickel instead of expensive precious metals, operates under mild room temperature conditions with visible light, and demonstrates excellent functional group compatibility and high enantioselectivity (up to 96% ee).
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the protocol uses simple reagents and standard equipment (Schlenk tubes, blue LEDs). The mild conditions and high atom economy make it highly scalable for commercial production of complex chiral intermediates.
Q: What types of substrates are compatible with this cross-coupling reaction?
A: The method shows broad substrate universality, tolerating various functional groups such as esters, aldehydes, trifluoromethyl groups, and heterocycles like thiophene and indole derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alpha-Aryl Phosphate Supplier
The technological advancements described in patent CN114315894B represent a significant opportunity for the pharmaceutical and fine chemical industries to access high-value chiral intermediates more efficiently. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative academic and patent discoveries into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of chiral alpha-aryl phosphate meets the highest international standards.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific project needs. Whether you require custom synthesis services or bulk supply of these specialized intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain with reliable, high-quality chiral building blocks.
