Advanced Asymmetric Synthesis of Chloramphenicol Intermediates for Scalable Pharmaceutical Manufacturing
Advanced Asymmetric Synthesis of Chloramphenicol Intermediates for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective routes for critical antibiotic precursors. Patent CN115745823A introduces a groundbreaking preparation method for N-[(1R,2S)-2-hydroxy-1-hydroxymethyl-2-(4-nitrophenyl)-ethyl]-acetamide, a pivotal intermediate in the synthesis of chloramphenicol. This technology addresses long-standing inefficiencies in chiral synthesis by replacing traditional resolution methods with a streamlined, catalytic asymmetric approach. The disclosed methodology leverages a recyclable copper-based catalyst system that delivers superior enantioselectivity and reaction yields compared to historical benchmarks. For global supply chain leaders, this represents a significant opportunity to optimize the manufacturing of high-purity pharmaceutical intermediates while adhering to green chemistry principles. The following analysis details the technical merits and commercial viability of this innovative synthetic pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the enantioselective synthesis of N-[(1R,2S)-2-hydroxy-1-hydroxymethyl-2-(4-nitrophenyl)-ethyl]-acetamide has relied heavily on chiral resolution using tartaric acid. This conventional approach is inherently flawed due to its theoretical maximum yield of 50%, necessitating the recycling or disposal of the unwanted enantiomer. Furthermore, the resolution process often requires multiple crystallization steps, leading to excessive solvent consumption and prolonged production cycles. The reliance on stoichiometric amounts of chiral resolving agents significantly inflates raw material costs and generates substantial chemical waste, conflicting with modern sustainability mandates. Additionally, achieving high optical purity through resolution can be unpredictable, often resulting in batch-to-batch variability that complicates quality control protocols for regulatory compliance.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a catalytic asymmetric strategy that fundamentally transforms the production economics. By employing a chiral Cu-BOX complex, the synthesis achieves direct construction of the chiral center with high fidelity, bypassing the 50% yield ceiling of resolution methods. The process operates under mild conditions, utilizing readily available reagents such as formaldehyde and trimethylchlorosilane, which simplifies procurement logistics. The catalyst system is not only effective but also recyclable, offering a sustainable advantage that reduces the environmental footprint of the manufacturing process. This shift from stoichiometric resolution to catalytic asymmetry ensures a more consistent supply of high-purity intermediates, directly addressing the reliability concerns of procurement managers in the antibiotic sector.
Mechanistic Insights into Cu-BOX Catalyzed Asymmetric Aldol Reaction
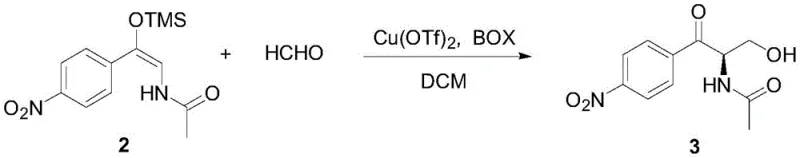
The core of this technological breakthrough lies in the second step: the asymmetric Aldol reaction mediated by a copper-bisoxazoline (Cu-BOX) complex. In this mechanism, the copper center coordinates with the chiral BOX ligand to create a rigid, sterically defined environment around the reactive site. When the silyl enol ether substrate interacts with the activated formaldehyde within this chiral pocket, the transition state is strictly controlled, favoring the formation of the desired (1R) configuration. The use of additives like trifluoroethanol further enhances the reaction rate and selectivity by stabilizing the transition state through hydrogen bonding interactions. This precise molecular recognition ensures that the carbon-carbon bond formation occurs with exceptional stereocontrol, yielding products with enantiomeric excess values reaching up to 94% ee.
From an impurity control perspective, this catalytic mechanism offers distinct advantages over non-catalytic methods. The high enantioselectivity inherently minimizes the formation of the undesired diastereomer, drastically reducing the burden on downstream purification processes. In traditional synthesis, removing trace chiral impurities often requires expensive preparative chromatography or repeated recrystallizations, which erode profit margins. By front-loading the stereochemical integrity in the Aldol step, the process ensures that the subsequent hydrogenation step proceeds with a clean substrate. This results in a final product profile that meets stringent pharmacopeial standards for chiral purity with minimal additional processing, thereby enhancing the overall efficiency of the production line.
How to Synthesize N-[(1R,2S)-2-Hydroxy-1-Hydroxymethyl-2-(4-Nitrophenyl)-Ethyl]-Acetamide Efficiently
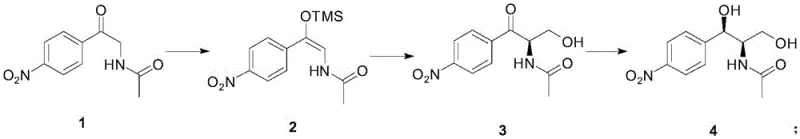
The synthesis is executed via a concise three-step sequence that balances operational simplicity with high performance. The process begins with the protection of the ketone as a silyl enol ether, followed by the critical asymmetric Aldol addition, and concludes with a stereoselective hydrogenation. Each step has been optimized for solvent compatibility and temperature control to maximize yield and safety. The detailed standardized synthesis steps below outline the specific conditions required to replicate the high-efficiency results reported in the patent documentation.
- Perform enol silyletherification of N-[2-(4-nitrophenyl)-2-carbonyl-ethyl]-acetamide using TMSCl and triethylamine in diethyl ether at -30°C to -10°C.
- Conduct asymmetric Aldol reaction with formaldehyde using a Cu(OTf)2 and CH2 iPr-BOX catalyst system in dichloromethane at -78°C to achieve high enantioselectivity.
- Execute asymmetric catalytic hydrogenation of the ketone intermediate using a transition metal catalyst and chiral phosphine ligand in methanol or ethanol under 5 bar hydrogen pressure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain executives, the adoption of this patented methodology translates into tangible strategic benefits beyond mere technical novelty. The elimination of expensive chiral resolving agents and the reduction in solvent-intensive purification steps lead to a streamlined cost structure. Furthermore, the use of robust, recyclable catalysts mitigates the risk of supply disruptions associated with specialty reagents. The process's scalability ensures that manufacturers can respond agilely to market demand fluctuations without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The transition from stoichiometric resolution to catalytic asymmetric synthesis fundamentally alters the cost basis of production. By utilizing a recyclable copper catalyst system, the process eliminates the recurring expense of purchasing large quantities of chiral acids like tartaric acid. Additionally, the higher overall yield means that less raw material is required to produce the same amount of final product, effectively lowering the cost per kilogram. The reduction in solvent usage and waste treatment requirements further contributes to substantial operational savings, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as formaldehyde, triethylamine, and common solvents like dichloromethane and ethanol ensures a stable supply chain. Unlike specialized chiral auxiliaries that may have limited suppliers and long lead times, the reagents for this process are widely available from multiple global sources. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. Moreover, the simplified workflow reduces the number of unit operations, decreasing the potential for equipment bottlenecks and ensuring a smoother, more predictable production schedule.
- Scalability and Environmental Compliance: The mild reaction conditions, particularly the ambient pressure hydrogenation and moderate temperatures in the Aldol step, facilitate easy scale-up from pilot to commercial plants. The process avoids the use of highly toxic or hazardous reagents that would require expensive containment systems, thus lowering capital expenditure for new facilities. From an environmental standpoint, the improved atom economy and catalyst recyclability align with green chemistry principles, helping manufacturers meet increasingly rigorous environmental regulations and sustainability goals without sacrificing productivity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent, providing clarity for technical teams evaluating the feasibility of adoption.
Q: What are the advantages of this new synthesis method over traditional tartaric acid resolution?
A: The new method utilizes recyclable copper-based catalysts and avoids the costly, inefficient, and waste-generating tartaric acid chiral resolution process, significantly improving atom economy and reducing production costs.
Q: What represents the key stereochemical control step in this process?
A: The critical stereochemical control occurs during the asymmetric Aldol reaction (Step 2), where a Cu-BOX complex catalyst directs the formation of the chiral center with high enantiomeric excess (up to 94% ee).
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process features mild reaction conditions, commercially available reagents, and high overall yields (up to 79%), making it highly amenable to commercial scale-up for antibiotic manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-[(1R,2S)-2-Hydroxy-1-Hydroxymethyl-2-(4-Nitrophenyl)-Ethyl]-Acetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving antibiotics like chloramphenicol. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of N-[(1R,2S)-2-hydroxy-1-hydroxymethyl-2-(4-nitrophenyl)-ethyl]-acetamide meets the highest international standards. Our infrastructure is designed to support the complex catalytic processes described in recent patents, delivering consistency and reliability to our global partners.
We invite pharmaceutical manufacturers and procurement leaders to collaborate with us to leverage this advanced technology for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your production efficiency and reduce your overall manufacturing costs.
