Revolutionizing Beta-Lactam Production: Advanced Enzymatic Acylation for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient pathways for the production of critical beta-lactam antibiotics, and patent CN1087350C presents a transformative approach to enzymatic synthesis. This specific intellectual property details a novel process for producing penicillins and cephalosporins by reacting a beta-lactam nucleus with an amide, catalyzed by penicillin acylase in the presence of enzyme inhibitors. Traditionally, the presence of inhibitors such as phenylacetic acid or p-hydroxyphenylacetic acid in the reaction mixture has been viewed as a detrimental factor that necessitates rigorous purification of intermediates like 6-aminopenicillanic acid (6-APA) or 7-amino-deacetoxycephalosporanic acid (7-ADCA). However, this technology challenges that paradigm by demonstrating that the reaction can proceed effectively even when these inhibitors are present, thereby streamlining the manufacturing workflow. For R&D directors and process engineers, this represents a significant opportunity to simplify synthetic routes and reduce the environmental footprint of antibiotic production. The ability to bypass complex purification steps not only accelerates the timeline from raw material to active pharmaceutical ingredient but also aligns with the growing global demand for greener, more sustainable chemical manufacturing practices within the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional methods for the enzymatic acylation of beta-lactam nuclei have long been plagued by the stringent requirement for high-purity starting materials. In traditional processes, intermediates such as 6-APA and 7-ADCA must be meticulously purified to remove trace amounts of enzyme inhibitors like phenylacetic acid before they can be subjected to acylation. This purification typically involves extraction with organic solvents, which introduces significant operational costs, safety hazards, and environmental burdens associated with solvent recovery and waste disposal. Furthermore, to drive the equilibrium towards the desired product in the absence of optimized conditions, conventional methods often require a large molar excess of the amino acid derivative, sometimes up to 4 to 6 moles per mole of the beta-lactam nucleus. This excessive use of expensive chiral amino acid derivatives not only inflates raw material costs but also complicates the downstream isolation of the final product, as separating the unreacted starting materials and by-products becomes increasingly difficult. The cumulative effect of these inefficiencies is a manufacturing process that is costly, time-consuming, and less adaptable to the fluctuating quality of bulk raw materials available in the global supply chain.
The Novel Approach
The novel approach disclosed in this patent fundamentally alters the reaction dynamics by embracing the presence of inhibitors rather than fighting against them. By carefully controlling the concentration of specific inhibitors within the reaction mixture, the process allows for the direct use of impure intermediates that would otherwise be rejected or require expensive reprocessing. This method operates effectively within a temperature range of -5°C to +35°C and utilizes immobilized penicillin acylase, which offers superior stability and reusability compared to free enzymes. The strategic addition of inhibitors or the tolerance of their presence prevents the hydrolysis of the formed product and the acid amide reactant, thereby improving the overall molar yield without the need for massive excesses of reagents. This shift in methodology means that manufacturers can source lower-grade, more cost-effective raw materials and still achieve high-purity final products through a simplified workflow. For a reliable pharmaceutical intermediates supplier, adopting this technology translates into a more robust supply chain capable of withstanding raw material variability while maintaining consistent output quality and significantly reduced operational expenditures.
Mechanistic Insights into Penicillin Acylase-Catalyzed Acylation
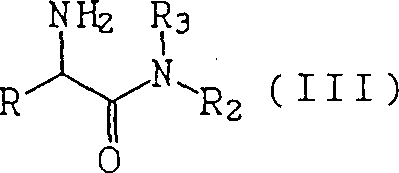
The core of this technological advancement lies in the nuanced understanding of penicillin acylase (E.C.3.5.1.11) kinetics and its interaction with specific molecular structures. The enzyme, typically derived from microbial sources such as Escherichia coli or Kluyvera, facilitates the nucleophilic attack of the beta-lactam nucleus on the acyl donor. In the context of this patent, the reactant is an alpha-substituted amino acid amide, represented structurally in the provided data. The presence of inhibitors, which are structurally similar to the side chains cleaved during hydrolysis, traditionally competes for the active site, leading to enzyme deactivation. However, the patent reveals that at specific volumetric molar concentrations, these inhibitors do not completely halt catalysis but rather modulate the reaction environment to favor acylation over hydrolysis. This delicate balance prevents the enzymatic degradation of the newly formed beta-lactam bond, a common side reaction that plagues conventional enzymatic syntheses. By maintaining the reaction in an aqueous medium with controlled pH and temperature, the process ensures that the enzyme remains active long enough to achieve high conversion rates, often exceeding 90% under optimal conditions, without the need for organic co-solvents that might denature the biocatalyst.
Impurity control is another critical aspect of this mechanism, particularly concerning the stereochemistry and side-reaction products. The use of specific amide derivatives, such as D-phenylglycinamide, ensures that the resulting antibiotic possesses the correct chirality required for biological activity. In conventional processes, the hydrolysis of the amide donor by the enzyme itself generates free amino acids, which can act as impurities and complicate purification. The inhibitor-tolerant mechanism described here suppresses this hydrolytic activity, ensuring that the majority of the amide donor is consumed in the desired acylation pathway. This results in a cleaner reaction profile with fewer by-products, simplifying the crystallization and isolation steps. For R&D teams focused on purity and impurity profiles, this means a more predictable process with reduced risk of genotoxic impurities or difficult-to-remove organic residues. The ability to manage the reaction pathway through inhibitor modulation rather than brute-force purification represents a sophisticated level of process control that enhances the overall quality of the high-purity beta-lactam antibiotics produced.
How to Synthesize Cephalexin Efficiently
The synthesis of Cephalexin, a widely used cephalosporin antibiotic, serves as a prime example of the practical application of this patented technology. The process begins with the preparation of an aqueous solution containing 7-amino-deacetoxycephalosporanic acid (7-ADCA) and D-(-)-phenylglycinamide. Unlike traditional methods that demand ultra-pure 7-ADCA, this protocol allows for the use of intermediates containing trace amounts of phenylacetic acid or other inhibitors. The reaction mixture is cooled to a controlled temperature, typically around 4°C, and the pH is meticulously maintained, often near neutrality, using automatic titration systems. Immobilized penicillin acylase is then introduced to the reactor, where it catalyzes the coupling of the side chain to the beta-lactam nucleus. The detailed standardized synthesis steps, including specific reagent quantities, agitation rates, and work-up procedures, are outlined in the guide below for technical reference.
- Prepare an aqueous reaction mixture containing the beta-lactam nucleus (such as 6-APA or 7-ADCA) and the alpha-substituted amino acid amide derivative.
- Adjust the reaction conditions to a temperature range of -5°C to +35°C and maintain a controlled pH level suitable for penicillin acylase activity.
- Introduce the immobilized penicillin acylase catalyst along with a specific enzyme inhibitor to facilitate acylation without prior removal of impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic synthesis method offers profound strategic advantages that extend beyond mere technical feasibility. The primary value proposition lies in the drastic simplification of the upstream supply chain. By eliminating the need for rigorous purification of intermediates to remove enzyme inhibitors, manufacturers can source raw materials from a broader range of suppliers, including those offering lower-cost, technical-grade intermediates. This flexibility reduces dependency on single-source, high-purity vendors and mitigates the risk of supply disruptions caused by quality bottlenecks. Furthermore, the removal of organic solvent extraction steps significantly lowers the consumption of hazardous chemicals, reducing both the direct cost of solvents and the indirect costs associated with environmental compliance, waste treatment, and safety protocols. These operational efficiencies translate into a more resilient and cost-effective manufacturing model that can better absorb market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive purification stages and the reduction in reagent consumption. By avoiding the use of organic solvents for extracting inhibitors, the process saves on solvent purchase, recovery, and disposal costs, which are substantial in large-scale antibiotic production. Additionally, the improved reaction efficiency reduces the molar excess of chiral amino acid derivatives required, directly lowering the bill of materials for every batch produced. This qualitative improvement in process economy allows for significant cost savings in API manufacturing without compromising on the quality or purity of the final pharmaceutical product, making it an attractive option for cost-sensitive generic drug production.
- Enhanced Supply Chain Reliability: Supply chain reliability is significantly bolstered by the process's tolerance for raw material variability. In traditional manufacturing, a slight deviation in the purity of 6-APA or 7-ADCA can lead to batch failures or extensive rework, causing delays in delivery. This new method's ability to function effectively with impure intermediates means that production schedules are less vulnerable to raw material quality fluctuations. This robustness ensures a more consistent output of commercial scale-up of complex enzymatic reactions, allowing supply chain managers to promise tighter delivery windows and maintain higher inventory levels of finished goods. It effectively decouples production stability from the volatility of the upstream intermediate market.
- Scalability and Environmental Compliance: From a scalability perspective, the use of immobilized enzymes in an aqueous medium is inherently suited for large-scale industrial reactors. The process avoids the safety hazards associated with large volumes of flammable organic solvents, simplifying facility requirements and reducing insurance and safety compliance costs. Environmentally, the reduction in solvent waste aligns with increasingly stringent global regulations on pharmaceutical manufacturing emissions. This green chemistry approach not only future-proofs the manufacturing facility against regulatory changes but also enhances the corporate sustainability profile, which is becoming a key criterion for partnerships with major multinational pharmaceutical companies seeking eco-friendly suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this inhibitor-tolerant enzymatic synthesis. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy for decision-makers evaluating this technology for potential adoption or procurement. Understanding these nuances is critical for assessing the feasibility of integrating this process into existing production lines or for qualifying new suppliers who utilize this advanced methodology.
Q: How does the presence of enzyme inhibitors affect the yield of beta-lactam synthesis?
A: Contrary to conventional wisdom, the patented process demonstrates that specific enzyme inhibitors, when present in controlled volumetric molar concentrations, do not halt the catalytic activity of penicillin acylase. This allows for the direct use of impure intermediates containing trace inhibitors, maintaining high molar yields without the need for costly purification steps.
Q: What are the primary cost advantages of this enzymatic acylation method?
A: The primary economic benefit stems from the elimination of organic solvent extraction steps traditionally required to remove inhibitors like phenylacetic acid from intermediates. By tolerating these impurities, the process significantly reduces raw material costs, solvent consumption, and waste treatment expenses associated with intermediate purification.
Q: Is this process scalable for industrial production of cephalosporins?
A: Yes, the method utilizes immobilized penicillin acylase, which is highly suitable for industrial reactor setups. The process operates under mild aqueous conditions and avoids harsh organic solvents, making it environmentally compliant and easily scalable from pilot batches to multi-ton commercial production of antibiotics like Cephalexin.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephalexin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced enzymatic processes like the one described in CN1087350C for the production of high-value beta-lactam antibiotics. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory techniques are successfully translated into robust industrial realities. Our facilities are equipped with state-of-the-art biocatalysis reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of Cephalexin or related intermediate meets the highest international standards. We understand that the transition to greener, more efficient synthesis routes requires a partner with deep technical expertise and a commitment to quality, and we are positioned to support your long-term supply needs with reliability and precision.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of these advanced synthetic routes. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific product portfolio, demonstrating how these process improvements can impact your bottom line. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to a supply chain that is not only cost-effective but also resilient, sustainable, and capable of meeting the demanding requirements of the global pharmaceutical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
