Scalable Synthesis of 9-(2-Hydroxyethyl)Carbazole for Advanced Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries continuously demand high-purity intermediates that can be manufactured reliably at scale. A pivotal advancement in this domain is detailed in patent CN102633708A, which outlines a novel synthesizing and purifying method for the critical intermediate 9-(2-hydroxyethyl)carbazole. This compound serves as a foundational building block for various optoelectronic materials and bioactive pharmaceutical ingredients, where structural integrity and purity are paramount. Traditional synthesis routes often struggle with selectivity issues, leading to complex impurity profiles that complicate downstream processing. The disclosed technology addresses these challenges by introducing a phase transfer catalytic system that operates under significantly milder conditions than conventional strong-base methods. By optimizing reaction parameters and solvent systems, this approach not only enhances yield but also simplifies the purification workflow, making it an attractive option for manufacturers seeking a reliable pharmaceutical intermediate supplier. The strategic implementation of this chemistry allows for better control over the molecular architecture, ensuring that the final product meets the stringent specifications required for high-value applications in drug discovery and material science.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 9-(2-hydroxyethyl)carbazole has been plagued by significant technical hurdles that impact both cost and safety. Conventional methods frequently rely on hazardous reagents such as ethylene oxide, which is a gaseous substance prone to explosion, necessitating specialized high-pressure equipment and rigorous safety protocols that drive up capital expenditure. Alternatively, processes using chloroethanol or bromoethanol often require strong bases like sodium hydride to deprotonate the weakly acidic nitrogen atom on the carbazole ring. These harsh conditions can lead to thermal instability and make solvent recovery difficult, particularly when high-boiling aprotic solvents like DMF are employed. Furthermore, a critical chemical challenge arises from the similar acidity of the starting material's N-H bond and the product's hydroxyl O-H bond. This similarity often triggers uncontrolled cascade reactions, resulting in the formation of polymeric by-products and oligomers that are structurally similar to the target molecule.
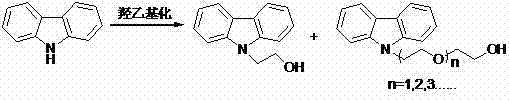
As illustrated in the reaction pathway above, the competition between the desired N-alkylation and unwanted O-alkylation leads to a complex mixture that is notoriously difficult to separate. Standard purification techniques such as repeated recrystallization or column chromatography are often required to achieve acceptable purity levels, which drastically reduces overall yield and increases waste generation. These inefficiencies render many traditional methods unsuitable for cost reduction in pharmaceutical intermediate manufacturing, as the loss of raw materials and the energy intensity of purification erode profit margins. Additionally, the use of moisture-sensitive reagents demands anhydrous conditions, adding another layer of operational complexity and expense. For supply chain managers, these factors translate into longer lead times and higher vulnerability to production disruptions, highlighting the urgent need for a more robust and selective synthetic strategy.
The Novel Approach
The innovative method described in the patent data overcomes these legacy issues by employing a phase transfer catalysis (PTC) system combined with inexpensive inorganic bases. Instead of relying on dangerous gases or pyrophoric hydrides, this process utilizes halogenated ethanol and catalysts such as quaternary ammonium salts or polyethylene glycols to facilitate the reaction across phase boundaries. This shift allows the reaction to proceed at moderate temperatures ranging from 40°C to 100°C, eliminating the need for extreme thermal inputs or cryogenic cooling. The core breakthrough lies in the ability of the PTC system to differentiate between the reactivity of the carbazole nitrogen and the product hydroxyl group, effectively suppressing the formation of the polymeric impurities seen in older methods. By carefully selecting the solvent system, operators can maximize the solubility of the product while keeping unreacted starting materials in a state that allows for easy physical separation. This results in a cleaner crude product that requires less intensive purification, thereby streamlining the entire manufacturing workflow. For procurement teams, this translates to a more predictable production schedule and reduced dependency on specialty chemicals that are subject to volatile market pricing.
Mechanistic Insights into Phase Transfer Catalyzed N-Alkylation
Understanding the mechanistic underpinnings of this synthesis is crucial for R&D directors evaluating the feasibility of technology transfer. The reaction mechanism relies on the transport of hydroxide ions from the aqueous or solid phase into the organic phase where the carbazole resides. The phase transfer catalyst forms a lipophilic ion pair with the hydroxide anion, which then deprotonates the carbazole nitrogen to generate a nucleophilic carbazolyl anion. This anion attacks the halogenated ethanol to form the desired 9-(2-hydroxyethyl)carbazole. Crucially, the reaction conditions are tuned to stop the process before the hydroxyl group on the newly formed product can compete for alkylation. The presence of inorganic iodides further enhances the reaction rate through halogen exchange, generating more reactive iodo-intermediates in situ. This catalytic cycle ensures high turnover and selectivity, minimizing the consumption of base and reducing the generation of inorganic salt waste.
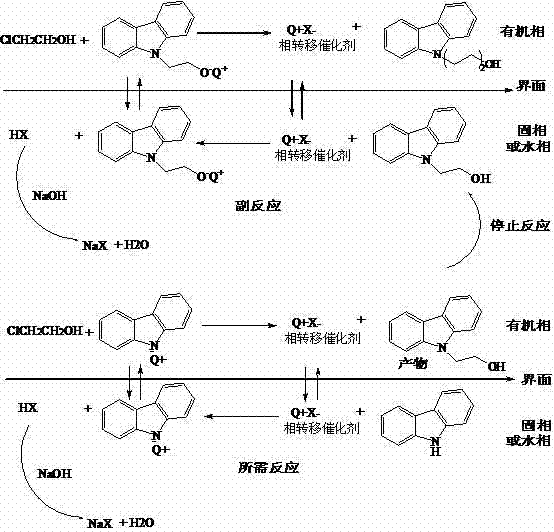
The diagram above highlights the divergence between the desired reaction pathway and the side reactions that occur in the absence of proper control. In the upper section, uncontrolled conditions lead to O-alkylation and subsequent polymerization, creating a distribution of chain lengths that are hard to isolate. In contrast, the lower section demonstrates how the phase transfer catalyst directs the reaction specifically towards N-alkylation, terminating the process at the mono-substituted stage. This level of control is essential for maintaining a narrow impurity profile, which simplifies the analytical validation required for regulatory compliance. By inhibiting the O-alkylation pathway, the process avoids the formation of ethers and esters that would otherwise co-elute with the product during chromatography. This mechanistic precision ensures that the commercial scale-up of complex pharmaceutical intermediates remains viable without compromising on quality standards. It provides a scientific basis for the observed improvements in yield and purity, offering confidence to technical stakeholders regarding the robustness of the chemistry.
How to Synthesize 9-(2-Hydroxyethyl)Carbazole Efficiently
Implementing this synthesis route requires careful attention to solvent selection and stoichiometry to replicate the high efficiency reported in the patent literature. The process begins with the dissolution of carbazole in a suitable organic solvent, followed by the addition of the inorganic base and the phase transfer catalyst. Maintaining vigorous stirring is essential to ensure effective mass transfer between the phases, which drives the reaction kinetics. Once the reaction is complete, the workup involves a strategic filtration step that leverages the solubility differences between the product and the starting material. This allows for the direct recovery of unreacted carbazole, which can be recycled back into the process, further enhancing the economic viability of the method. The detailed standardized synthesis steps see the guide below for specific parameters regarding temperature, timing, and reagent ratios.
- Dissolve carbazole in a selected organic solvent system and add inorganic base, halogenated ethanol, phase transfer catalyst, and inorganic iodide.
- Heat the mixture to 40-100°C under vigorous stirring until the reaction reaches completion, then filter to remove insolubles.
- Purify the crude product by dissolving in a polar solvent to recover unreacted carbazole, followed by decolorization and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this manufacturing process offers substantial benefits that align with the strategic goals of cost efficiency and supply chain resilience. The elimination of hazardous reagents like ethylene oxide removes the need for specialized storage and handling infrastructure, significantly lowering the barrier to entry for production facilities. This simplification of safety requirements reduces insurance costs and minimizes the risk of regulatory shutdowns due to compliance issues. Furthermore, the ability to recover and reuse unreacted raw materials directly impacts the cost of goods sold, providing a buffer against fluctuations in raw material pricing. The mild reaction conditions also extend the lifespan of reactor vessels and reduce energy consumption associated with heating and cooling, contributing to a more sustainable manufacturing footprint. These factors collectively enhance the reliability of supply, ensuring that downstream customers receive consistent quality without unexpected delays.
- Cost Reduction in Manufacturing: The transition to inorganic bases and phase transfer catalysts replaces expensive and hazardous reagents, leading to significant cost savings in raw material procurement. By avoiding the use of sodium hydride or high-pressure ethylene oxide systems, manufacturers can operate with standard glass-lined or stainless steel reactors, reducing capital depreciation costs. The simplified purification process reduces solvent consumption and waste disposal fees, which are often hidden costs in traditional synthetic routes. Additionally, the recovery of unreacted carbazole means that less fresh material is needed per batch, optimizing the overall material balance. These efficiencies compound over large production volumes, resulting in a more competitive pricing structure for the final intermediate without sacrificing margin.
- Enhanced Supply Chain Reliability: Sourcing hazardous gases like ethylene oxide can be a bottleneck due to strict transportation regulations and limited supplier availability. By switching to liquid halogenated ethanols and solid inorganic salts, the supply chain becomes more flexible and less prone to disruption. The robustness of the reaction conditions means that production is less sensitive to minor variations in utility supply, such as cooling water temperature fluctuations. This stability allows for more accurate forecasting and inventory planning, reducing the need for excessive safety stock. For global buyers, this translates to shorter lead times for high-purity pharmaceutical intermediates and a lower risk of allocation during market shortages. The ability to source reagents from multiple vendors further mitigates single-source risk, ensuring continuity of supply.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, avoiding exothermic runaways that are common with strong hydride bases. This thermal safety profile allows for larger batch sizes without proportional increases in risk, facilitating the transition from pilot plant to full commercial production. The use of recoverable solvents and the generation of less hazardous waste streams align with increasingly strict environmental regulations globally. Reduced solvent usage and the ability to recycle aqueous phases minimize the environmental footprint of the manufacturing site. This compliance advantage is critical for multinational corporations that audit their suppliers on sustainability metrics. It ensures that the production of these key intermediates supports broader corporate social responsibility goals while maintaining operational excellence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this intermediate. They are derived from the specific advantages and operational parameters detailed in the underlying patent documentation. Understanding these aspects helps stakeholders make informed decisions about integrating this material into their supply chains. The answers reflect the consensus on best practices for handling and sourcing this specific chemical entity.
Q: How does this method prevent polymerization side reactions?
A: By utilizing phase transfer catalysis, the process exploits the subtle acidity differences between the carbazole N-H bond and the product hydroxyl O-H bond, effectively inhibiting O-alkylation and subsequent chain growth.
Q: Can unreacted raw materials be recovered?
A: Yes, the purification strategy leverages solubility differences in polar solvents to filter out unreacted carbazole, allowing for significant raw material recovery and cost optimization.
Q: Is this process suitable for large-scale production?
A: Absolutely. The method operates under mild temperatures without requiring anhydrous or anaerobic conditions, using standard equipment that facilitates safe and efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-(2-Hydroxyethyl)Carbazole Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We adhere to stringent purity specifications and utilize rigorous QC labs to verify every batch against the highest industry standards. Our commitment to quality assurance means that you receive a product that is ready for immediate use in sensitive downstream applications, minimizing your internal testing burden. We view ourselves not just as a vendor, but as a strategic extension of your R&D and supply chain teams.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how we can support your project goals. Request a Customized Cost-Saving Analysis to understand how our optimized manufacturing processes can improve your bottom line. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver consistent quality. Let us help you secure a stable supply of high-performance intermediates for your next breakthrough product.
