Advanced Fluorination Technology for High-Purity 1,7,8-Trifluoro-2-Naphthol Commercialization
Advanced Fluorination Technology for High-Purity 1,7,8-Trifluoro-2-Naphthol Commercialization
The landscape of electronic chemical manufacturing is constantly evolving, driven by the demand for higher purity intermediates and more sustainable production methodologies. Patent CN101550071B introduces a groundbreaking approach to synthesizing 1,7,8-trifluoro-2-naphthol, a pivotal intermediate for next-generation liquid crystal displays. This technology shifts away from traditional, costly fluorination reagents towards a direct fluorine gas methodology that operates under controlled low-temperature conditions. By leveraging the unique solubility characteristics of the product in protic polar solvents, this invention achieves high selectivity in a single operational step. For industry leaders, this represents a significant opportunity to optimize supply chains for display materials while ensuring the rigorous purity standards required for high-performance optoelectronic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoronaphthol derivatives relied heavily on specialized electrophilic fluorinating agents, such as fluoropyridinium salts. While effective in introducing fluorine atoms, these reagents present substantial economic and technical barriers for large-scale manufacturing. The primary drawback lies in the tendency of these aggressive fluorinating agents to cause over-fluorination, leading to the formation of unwanted byproducts like 1,1,7,8-tetrafluoro-1,2-dihydronaphthalen-2-one. Remedying this issue necessitates a subsequent reduction step to convert the ketone byproduct back to the desired naphthol structure, thereby complicating the process flow. Furthermore, the decomposition of these expensive salts and the potential for side reactions with chlorinated solvents create significant impurity burdens that challenge downstream purification efforts.
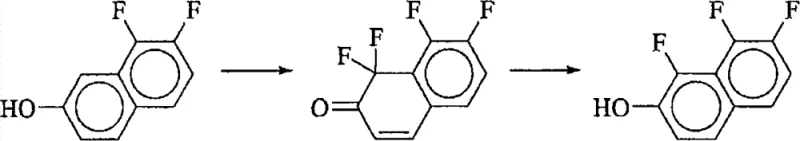
The Novel Approach
In stark contrast to legacy methods, the patented process utilizes elemental fluorine gas diluted in an inert carrier, reacting directly with 7,8-difluoro-2-naphthol. This method capitalizes on the high reactivity of fluorine gas but tames it through precise temperature control ranging from -100°C to -20°C. The brilliance of this approach lies in the choice of solvent; by using protic polar solvents like methanol, the system exploits the lower solubility of the trifluorinated product compared to the starting material. As the reaction proceeds, the desired 1,7,8-trifluoro-2-naphthol precipitates out of the solution, physically removing it from the reactive environment and preventing further fluorination. This self-limiting mechanism ensures high purity without the need for complex protective group strategies or multi-step reductions.
Mechanistic Insights into Low-Temperature Electrophilic Fluorination
The core of this technological advancement is the precise management of reaction kinetics and thermodynamics through solvent engineering and thermal control. At the molecular level, the introduction of fluorine gas into a cold methanol solution creates a highly selective environment for electrophilic aromatic substitution. The low temperature suppresses the activation energy required for secondary fluorination events, while the protic nature of the solvent stabilizes the transition states involved in the initial substitution. Crucially, the immediate precipitation of the product acts as a kinetic trap; once the molecule is fluorinated at the 1-position, it crystallizes out of the liquid phase. This physical separation is far more effective than relying solely on chemical selectivity, as it removes the product from contact with the fluorinating agent before over-reaction can occur.
For R&D directors focused on impurity profiles, this mechanism offers a distinct advantage in controlling the spectral purity of the final intermediate. Traditional methods often struggle with regio-isomers and over-fluorinated species that share similar physical properties with the target molecule, making chromatographic separation difficult and yield-loss heavy. By preventing the formation of the tetrafluoro-ketone byproduct at the source, this process drastically simplifies the workup procedure. The resulting crude material requires minimal purification, often needing only a simple wash and recrystallization to meet the stringent specifications demanded by the liquid crystal industry. This level of control is essential for maintaining the performance characteristics of the final display materials.
How to Synthesize 1,7,8-Trifluoro-2-Naphthol Efficiently
The implementation of this synthesis route requires careful attention to safety protocols regarding fluorine gas handling, but the operational steps are remarkably streamlined compared to prior art. The process begins with the dissolution of the difluoro-naphthol precursor in a suitable alcohol solvent, followed by cooling to cryogenic temperatures using liquid nitrogen or equivalent refrigeration. Once the thermal equilibrium is established, a diluted stream of fluorine gas is introduced under constant agitation. The visual indication of product formation is often marked by the onset of precipitation, signaling the successful conversion. Detailed standard operating procedures regarding gas flow rates, stoichiometric ratios, and quenching protocols are critical for reproducibility and safety.
- Dissolve 7,8-difluoro-2-naphthol in a protic polar solvent such as methanol and cool the solution to a temperature range between -100°C and -20°C.
- Introduce a diluted mixture of fluorine gas and inert gas (e.g., nitrogen) into the reaction vessel while maintaining vigorous stirring and low temperature control.
- Allow the resulting 1,7,8-trifluoro-2-naphthol product to precipitate out of the solution due to lower solubility, then isolate and purify the crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift from specialized fluorinating salts to elemental fluorine gas represents a fundamental restructuring of the cost base for this intermediate. Fluorine gas is a commodity chemical with a well-established global supply chain, whereas fluoropyridinium salts are niche, high-margin reagents produced by a limited number of suppliers. By eliminating the dependency on these expensive proprietary reagents, manufacturers can achieve substantial cost reductions in electronic chemical manufacturing. Additionally, the removal of the reduction step (required to fix over-fluorination in old methods) means fewer unit operations, less energy consumption, and reduced labor hours per batch. This efficiency translates directly into improved margin structures and more competitive pricing for downstream liquid crystal producers.
- Cost Reduction in Manufacturing: The elimination of expensive fluoropyridinium salts removes a significant variable cost component from the bill of materials. Furthermore, the single-step nature of the reaction avoids the capital and operational expenditures associated with the secondary reduction reactor and purification columns needed in conventional routes. This streamlined workflow reduces solvent usage and waste disposal costs, contributing to a leaner and more economically resilient production model that can better withstand fluctuations in raw material markets.
- Enhanced Supply Chain Reliability: Relying on commodity gases like fluorine and nitrogen, along with common solvents like methanol, mitigates the risk of supply disruptions often associated with specialty fine chemicals. The robustness of the supply chain is further enhanced by the simplicity of the process equipment, which does not require exotic materials of construction beyond standard fluorination-compatible alloys. This accessibility ensures that production can be scaled or shifted between facilities with greater flexibility, securing continuity of supply for critical display panel manufacturing lines.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of continuous or semi-continuous gas addition, which allows for precise heat management even in larger reactors. From an environmental standpoint, the avoidance of chlorinated solvents and the reduction of heavy metal catalysts (often used in reduction steps) aligns with increasingly strict global environmental regulations. The simpler waste stream, primarily consisting of aqueous washes and recovered solvents, facilitates easier treatment and compliance, reducing the regulatory burden on manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,7,8-trifluoro-2-naphthol. These insights are derived directly from the technical disclosures within the patent literature, focusing on the practical implications for industrial adoption. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this intermediate into their existing supply chains or product development pipelines.
Q: Why is direct fluorine gas preferred over fluoropyridinium salts for this synthesis?
A: Direct fluorine gas eliminates the need for expensive, specialized fluorinating agents like fluoropyridinium salts. It avoids the formation of difficult-to-remove tetrafluoro-byproducts that require additional reduction steps, thereby simplifying the workflow and reducing raw material costs significantly.
Q: How does the new method control selectivity to prevent over-fluorination?
A: The process utilizes a specific solubility differential in protic polar solvents at low temperatures. As the 1,7,8-trifluoro-2-naphthol forms, it precipitates out of the solution immediately. This physical removal from the reaction medium prevents further exposure to fluorine gas, effectively halting over-fluorination to tetrafluoro derivatives.
Q: What are the downstream applications of this intermediate?
A: This compound serves as a critical building block for trifluoronaphthalene-based liquid crystal compounds. These materials are essential for advanced display technologies, particularly those requiring negative dielectric anisotropy for VA (Vertical Alignment) and IPS (In-Plane Switching) modes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,7,8-Trifluoro-2-Naphthol Supplier
As the demand for high-performance liquid crystal materials continues to surge, the need for reliable sources of key intermediates like 1,7,8-trifluoro-2-naphthol has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch meets the exacting standards required for optoelectronic applications. We understand that consistency is key in the display industry, and our advanced manufacturing capabilities are designed to deliver that reliability consistently.
We invite potential partners to engage with our technical procurement team to discuss how this innovative fluorination technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to validate the suitability of our materials for your next-generation display projects. Let us collaborate to drive efficiency and innovation in the electronic materials sector together.
