Advanced Synthesis of Marizomib Key Intermediates via L-Serine Derivatives for Commercial Scale-up
Advanced Synthesis of Marizomib Key Intermediates via L-Serine Derivatives for Commercial Scale-up
The pharmaceutical industry continuously seeks robust and scalable pathways for complex oncology agents, and the synthesis of Marizomib (Salinosporamide A) represents a pinnacle of synthetic challenge due to its densely functionalized gamma-lactam backbone and five contiguous asymmetric centers. Patent CN111269257A introduces a groundbreaking preparation method that fundamentally shifts the paradigm from traditional total synthesis towards a more efficient, chiral-pool-based approach. This innovation utilizes natural L-serine as the primary starting material, connecting a functionally protected serine fragment with a 1,3-dicarbonyl cyclopropyl compound to construct the core skeleton. This strategic pivot not only enhances preparation efficiency and overall yield but also aligns with modern green chemistry principles by minimizing the use of hazardous reagents. For R&D directors and supply chain leaders, this patent signals a viable route for securing high-purity pharmaceutical intermediates with improved safety profiles and reduced environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of Marizomib, such as the classic route established by Corey et al., has been revered for its chemical elegance but criticized for its industrial impracticality. As illustrated in the foundational synthesis overview, these conventional pathways often rely on lengthy linear sequences involving expensive chiral auxiliaries or transition metal catalysts that are difficult to remove to ppm levels required for API production. 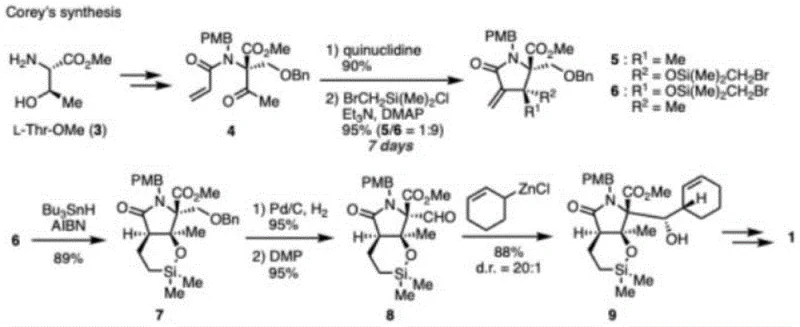 The reliance on highly toxic reagents and solvents with poor water solubility creates significant bottlenecks in waste management and operator safety. Furthermore, the purification and recovery processes in these legacy routes often suffer from substantial product loss, leading to low recovery rates and limiting the availability of the target product for clinical trials. The instability of certain intermediates in aqueous environments further complicates the workup procedures, necessitating stringent anhydrous conditions that drive up operational costs and energy consumption in a manufacturing setting.
The reliance on highly toxic reagents and solvents with poor water solubility creates significant bottlenecks in waste management and operator safety. Furthermore, the purification and recovery processes in these legacy routes often suffer from substantial product loss, leading to low recovery rates and limiting the availability of the target product for clinical trials. The instability of certain intermediates in aqueous environments further complicates the workup procedures, necessitating stringent anhydrous conditions that drive up operational costs and energy consumption in a manufacturing setting.
The Novel Approach
In stark contrast, the novel methodology detailed in the patent leverages the inherent chirality of abundant natural amino acids to bypass many of these stereochemical hurdles. By selecting L-serine as the starting raw material, the synthesis effectively installs critical stereocenters early in the sequence, drastically reducing the need for downstream resolution. The connection of the obtained serine fragment, protected by robust functional groups like PMB and TBS, with the 1,3-dicarbonyl cyclopropyl compound creates the Marizomib key intermediate framework with high atom economy. This approach is not only safer and greener but also demonstrates superior stability during the synthesis process. The use of low-toxicity solvents, particularly ethyl acetate for washing and extraction steps, avoids the complications associated with high-toxicity reagents, ensuring a higher recovery rate of the target product and better overall process stability suitable for GMP manufacturing environments.
Mechanistic Insights into Amide Coupling and Intramolecular Cyclization
The core of this synthetic strategy lies in the precise construction of the amide bond between the cyclopropyl acid chloride and the protected serine amine, followed by a cascade of cyclization events. The process begins with the generation of the beta-keto acid chloride fragment, which acts as the electrophilic partner. This fragment is coupled with the N-PMB-O-TBS-L-serine methyl ester under controlled conditions to form the linear precursor. The mechanistic elegance is further revealed in the subsequent intramolecular ring-closure reaction, where the latent nucleophile attacks the carbonyl center to form the characteristic gamma-lactam ring. This cyclization is critical as it establishes the rigid bicyclic core essential for proteasome inhibition. The use of specific reagents like Dess-Martin periodinane for oxidation and organozinc species for the introduction of the cyclohexenyl ring ensures high diastereoselectivity. 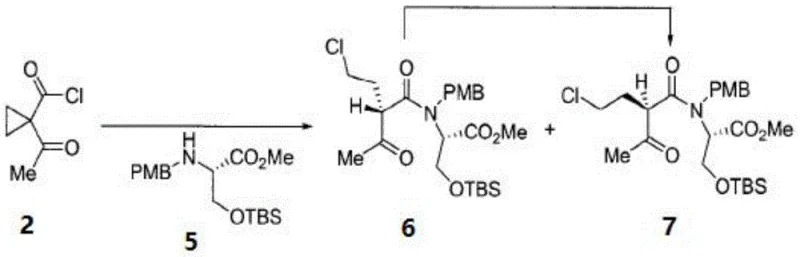 The careful control of reaction temperatures, such as maintaining -78°C during the organozinc addition, is paramount to preventing epimerization and ensuring the correct spatial arrangement of the substituents. This level of mechanistic control allows for the production of intermediates with defined impurity profiles, which is a key requirement for regulatory approval.
The careful control of reaction temperatures, such as maintaining -78°C during the organozinc addition, is paramount to preventing epimerization and ensuring the correct spatial arrangement of the substituents. This level of mechanistic control allows for the production of intermediates with defined impurity profiles, which is a key requirement for regulatory approval.
Furthermore, the impurity control mechanism is embedded within the choice of protecting groups and the sequence of deprotection. The PMB (p-methoxybenzyl) and TBS (tert-butyldimethylsilyl) groups provide orthogonal stability, allowing chemists to manipulate specific functional groups without affecting others. For instance, the removal of the TBS group using hydrofluoric acid reveals the hydroxyl necessary for lactone formation later, while the PMB group remains intact until the final stages. This orthogonality minimizes side reactions such as premature cyclization or elimination, which are common pitfalls in polyfunctional molecule synthesis. The final structural modification steps, including the selenium-mediated cyclization to form the beta-lactone, are executed with high precision, ensuring that the final Marizomib molecule possesses the requisite biological activity. 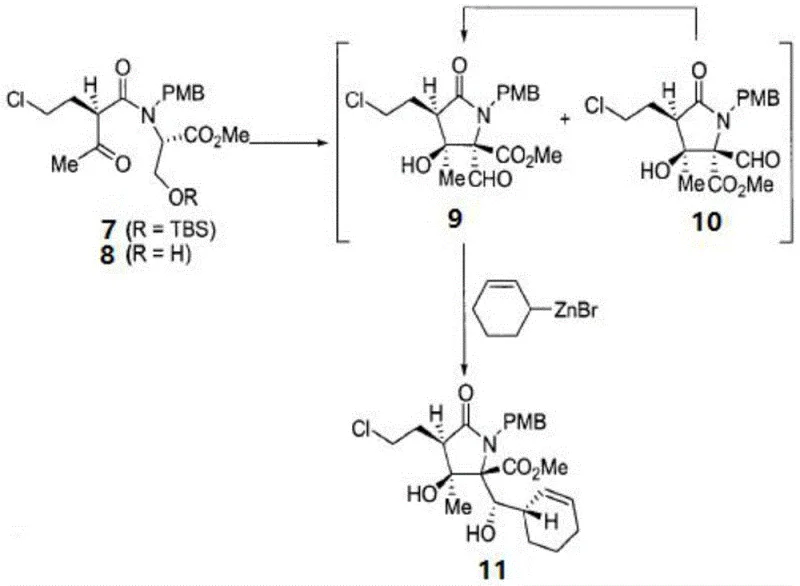 By understanding these mechanistic nuances, process chemists can better optimize reaction parameters to suppress trace impurities that could otherwise complicate the purification of the final API.
By understanding these mechanistic nuances, process chemists can better optimize reaction parameters to suppress trace impurities that could otherwise complicate the purification of the final API.
How to Synthesize Marizomib Key Intermediate Efficiently
The synthesis of the Marizomib key intermediate is a multi-step process that requires meticulous attention to reaction conditions and purification techniques to ensure high purity and yield. The patented route simplifies this by breaking it down into manageable fragments that are joined together through robust chemical transformations. The initial preparation of the beta-keto acid chloride involves cyclopropanation of ethyl acetoacetate followed by hydrolysis and chlorination, steps that are scalable and use commodity chemicals. Simultaneously, the L-serine fragment is prepared through esterification and protection, creating a stable nucleophile for the coupling event. The convergence of these two fragments marks the critical juncture of the synthesis, where the stereochemical integrity of the serine is transferred to the final scaffold. Following the coupling, a series of deprotection and cyclization steps finalize the core structure, ready for the final elaboration into Marizomib. This modular approach allows for parallel processing of fragments, significantly reducing the overall manufacturing timeline.
- Synthesize the beta-keto acid chloride fragment from ethyl acetoacetate via cyclopropanation and chlorination.
- Prepare the N-PMB-O-TBS-L-serine methyl ester from natural L-serine through protection and esterification.
- Perform the coupling reaction between the acid chloride and serine derivative, followed by intramolecular cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this L-serine-based synthesis route offers tangible benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike routes that depend on exotic chiral building blocks with long lead times, this method relies on L-serine, a commodity amino acid available in bulk quantities from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency and price volatility. Additionally, the elimination of transition metal catalysts in key steps removes the need for expensive and time-consuming heavy metal scavenging processes, which are often a bottleneck in API manufacturing. The use of ethyl acetate as a primary extraction solvent further reduces the cost of waste disposal and solvent recovery, contributing to a leaner and more cost-effective production model.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the utilization of low-cost starting materials and the avoidance of precious metal catalysts. By leveraging the chiral pool of L-serine, the synthesis bypasses the need for expensive asymmetric catalysis or resolution steps, which typically account for a significant portion of COGS in chiral drug manufacturing. Furthermore, the high recovery rate of the target product mentioned in the patent implies less material waste, directly translating to lower raw material consumption per kilogram of output. The simplified purification protocols, which avoid difficult-to-remove toxic reagents, also reduce the operational expenditure associated with specialized waste treatment and solvent recycling infrastructure.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures a consistent supply of high-quality intermediates. The stability of the intermediates, particularly the protected serine fragments, allows for longer storage times and easier logistics compared to unstable intermediates found in other routes. This stability is crucial for maintaining buffer stocks and ensuring continuity of supply during market fluctuations. Moreover, the use of standard laboratory equipment and common reagents means that the process can be easily transferred between different manufacturing sites without the need for specialized hardware, enhancing the flexibility of the supply network to respond to demand surges.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is significantly superior. The replacement of highly toxic solvents with greener alternatives like ethyl acetate reduces the facility's environmental footprint and simplifies regulatory compliance. The process generates less hazardous waste, lowering the costs associated with disposal and permitting. The scalability is evidenced by the straightforward workup procedures, such as simple filtration and crystallization, which are easily adapted from gram-scale to ton-scale production. This alignment with green chemistry principles not only future-proofs the manufacturing process against tightening environmental regulations but also enhances the corporate sustainability profile of the drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing clarity on the feasibility and advantages of the new route. Understanding these details is essential for stakeholders evaluating the potential for technology transfer or licensing agreements.
Q: How does this new route improve chirality control compared to traditional methods?
A: By utilizing natural L-serine as a starting material, the process leverages the chiral pool strategy, inherently establishing multiple stereocenters without the need for expensive chiral catalysts or resolution steps.
Q: What are the solvent advantages in this patented synthesis method?
A: The method prioritizes the use of ethyl acetate and other low-toxicity solvents for extraction and washing, significantly reducing environmental hazards and operator safety risks compared to routes relying on chlorinated solvents.
Q: Is this synthesis pathway suitable for large-scale industrial production?
A: Yes, the process is designed for scalability with high recovery rates, stable intermediates, and simplified purification steps, making it ideal for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Marizomib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a reliable supply chain for complex oncology intermediates like those required for Marizomib. Our team of expert process chemists has extensively evaluated the route described in CN111269257A and possesses the technical capability to execute this synthesis with precision. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Marizomib intermediate meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to optimize your supply chain and reduce your time to market. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with us, you gain access to our deep expertise in chiral synthesis and process optimization. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can support your Marizomib development goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
