Advanced Metal-Free Synthesis of Furyl O-Diketone Derivatives for Commercial Scale-Up
Advanced Metal-Free Synthesis of Furyl O-Diketone Derivatives for Commercial Scale-Up
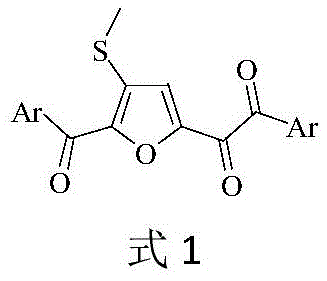
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing complex heterocyclic scaffolds, which serve as critical building blocks for bioactive molecules. Patent CN108358877B introduces a groundbreaking approach to synthesizing furyl o-diketone derivatives, a novel class of drug intermediates characterized by a unique furyl o-diketone parent structure modifiable with aryl and thioether groups. This technology represents a significant departure from traditional transition-metal-catalyzed routes, utilizing a metal-free oxidative cyclization strategy that leverages inexpensive aryl acetone compounds and dimethyl sulfoxide (DMSO). By employing a simple iodine and persulfate catalytic system under air atmosphere, this invention addresses long-standing challenges in heterocyclic synthesis, offering a pathway that is not only chemically efficient but also economically superior for industrial applications.
The core innovation lies in the ability to construct the polysubstituted furan ring directly from readily available ketone precursors without the necessity for pre-functionalized alkynes or expensive noble metal catalysts. This method provides a versatile platform for generating diverse libraries of furyl o-diketone derivatives, which are valuable intermediates in the development of new therapeutic agents. The process is designed with industrial scalability in mind, utilizing common chemical reagents and avoiding the stringent operational conditions often associated with organometallic chemistry. As a reliable pharmaceutical intermediate supplier, understanding such technological shifts is crucial for maintaining competitiveness in the global supply chain, particularly when aiming for cost reduction in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of furan rings has relied heavily on transition metal catalysis, particularly involving palladium, copper, gold, or silver complexes. Seminal work by researchers such as Utimoto and Huang utilized palladium-catalyzed cyclization of alkynyl ketones, which, while effective, suffers from significant drawbacks including the high cost of noble metals and the requirement for strictly anhydrous and oxygen-free conditions. Furthermore, many of these conventional methods exhibit limited substrate scope, often failing to accommodate non-terminal alkynes or sensitive functional groups without extensive protection-deprotection sequences. The reliance on stoichiometric amounts of oxidants like silver carbonate or copper salts further exacerbates the economic and environmental burden, generating substantial heavy metal waste that requires complex and costly removal processes to meet pharmaceutical purity standards. These factors collectively hinder the commercial viability of such routes for large-scale production of high-purity OLED material or drug intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN108358877B utilizes a metal-free catalytic system driven by elemental iodine and potassium persulfate. This approach fundamentally shifts the paradigm by using DMSO not merely as a solvent but as a dual-purpose reagent that provides both the oxygen atom for ring closure and the methylthio substituent. The reaction proceeds efficiently under an air atmosphere at moderate temperatures (120°C), eliminating the need for expensive inert gas protection and specialized equipment. This novel route demonstrates exceptional tolerance for a wide range of substituents on the aryl ring, including halogens, trifluoromethyl groups, and alkyl chains, achieving isolated yields between 63% and 92%. By circumventing the use of heavy metals, this process inherently simplifies the purification workflow, thereby facilitating the commercial scale-up of complex polymer additives and pharmaceutical intermediates with reduced environmental impact and lower overall production costs.
Mechanistic Insights into Iodine-Persulfate Mediated Oxidative Cyclization
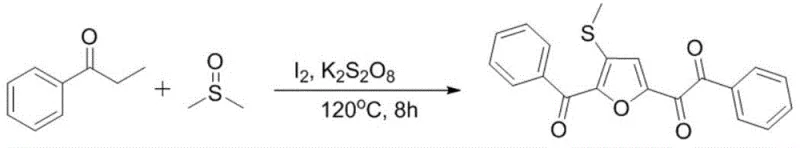
The reaction mechanism proposed for this transformation involves a sophisticated radical cascade initiated by the interaction of iodine and persulfate. Experimental evidence, including radical trapping studies using TEMPO and BHT which completely inhibited the reaction, confirms that the process proceeds via a free-radical pathway. Initially, the aryl acetone undergoes alpha-iodination or oxidation to form an intermediate species, while DMSO participates actively in the cycle. The persulfate oxidant generates sulfate radical anions which abstract hydrogen atoms, propagating the radical chain. Crucially, the DMSO molecule is activated to serve as the oxygen source for the furan ring formation, while simultaneously introducing the methylthio group at the 3-position through a radical coupling event. This intricate interplay between the iodine catalyst, the persulfate oxidant, and the DMSO reactant allows for the simultaneous construction of the heterocyclic core and the installation of functional handles in a single pot.
Understanding the impurity profile is vital for R&D directors focused on purity and杂质谱 (impurity spectrum). The mechanism suggests that side reactions could potentially arise from over-oxidation of the ketone or competitive radical pathways; however, the optimized conditions (specifically the 2.0 equivalents of persulfate and 50 mol% iodine) effectively channel the reaction towards the desired cyclization. The formation of the cationic intermediate followed by intramolecular cyclization and deprotonation ensures high regioselectivity. The absence of transition metals means that metal-induced side reactions, such as homocoupling of aryl groups often seen in palladium chemistry, are virtually eliminated. This results in a cleaner crude reaction mixture, reducing the burden on downstream purification and ensuring that the final high-purity furyl o-diketone derivatives meet stringent quality specifications required for subsequent drug synthesis steps.
How to Synthesize Furyl O-Diketone Derivatives Efficiently
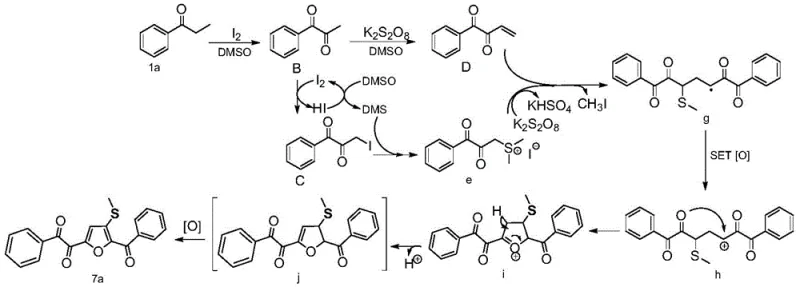
The synthesis protocol outlined in the patent provides a straightforward, one-pot procedure that is highly amenable to standard laboratory and pilot plant operations. The process begins with the precise weighing of aryl acetone substrates, elemental iodine, and potassium persulfate, which are then combined in a reaction vessel with DMSO serving as the solvent. The simplicity of the setup, requiring only heating under air, makes it accessible for facilities without specialized inert atmosphere gloveboxes. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the optimal conditions identified through extensive screening.
- Combine aryl acetone (0.5 mmol), elemental iodine (50 mol%), and potassium persulfate (2.0 equivalents) in a reaction vessel.
- Add dimethyl sulfoxide (DMSO) as both solvent and reactant (2.0 mL) and stir the mixture under an air atmosphere.
- Heat the reaction mixture to 120°C for 8 hours, then cool, extract with ethyl acetate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers transformative benefits regarding cost structure and logistical reliability. The elimination of noble metal catalysts such as palladium and platinum removes a major variable cost driver, as these metals are subject to significant market volatility and supply constraints. Furthermore, the avoidance of heavy metals simplifies the regulatory compliance landscape, as there is no need for expensive and time-consuming heavy metal scavenging steps or rigorous testing for residual metal content in the final API intermediate. This streamlining of the manufacturing process translates directly into substantial cost savings and a more predictable production timeline, enhancing the overall competitiveness of the supply chain.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with commodity chemicals like iodine and potassium persulfate drastically lowers the raw material cost per kilogram of product. Additionally, the use of DMSO as both solvent and reactant improves atom economy and reduces the volume of organic solvents required, leading to significant savings in solvent procurement and waste disposal costs. The simplified workup procedure, which avoids complex metal removal protocols, further reduces labor and processing time, contributing to a leaner and more cost-efficient manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable starting materials such as aryl acetones and DMSO ensures a robust supply chain that is less susceptible to disruptions compared to routes dependent on specialized alkynes or sensitive organometallic reagents. The ability to run the reaction under air atmosphere eliminates the dependency on bulk nitrogen or argon supplies, reducing logistical complexity and infrastructure costs. This resilience is critical for maintaining continuous production schedules and meeting delivery commitments for high-purity drug intermediates in a dynamic global market.
- Scalability and Environmental Compliance: The one-pot nature of the reaction and the use of relatively mild temperatures (120°C) make this process highly scalable from gram to multi-ton quantities without significant re-engineering. The absence of toxic heavy metal waste aligns with increasingly stringent environmental regulations and corporate sustainability goals, minimizing the environmental footprint of the manufacturing process. This green chemistry approach not only mitigates regulatory risks but also enhances the brand reputation of the manufacturer as a responsible and sustainable partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity on substrate scope, reaction conditions, and product quality. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of this iodine-catalyzed method over traditional palladium catalysis?
A: This method eliminates the need for expensive noble metal catalysts like palladium or platinum, significantly reducing raw material costs and simplifying downstream purification by removing heavy metal residues. It also operates under air atmosphere rather than requiring inert gas protection.
Q: What is the role of dimethyl sulfoxide (DMSO) in this reaction?
A: DMSO serves a dual function as both the reaction solvent and a critical reactant, providing the oxygen atom for the furan ring construction and the methylthio group for the substitution at the 3-position of the furan ring.
Q: Does this synthesis method tolerate various substituents on the aryl ring?
A: Yes, the process demonstrates broad substrate compatibility, successfully tolerating electron-withdrawing groups like halogens (F, Cl, Br) and trifluoromethyl groups, as well as electron-donating alkyl and alkoxy groups, with yields ranging from 63% to 92%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Furyl O-Diketone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of innovative synthetic methodologies like the one described in CN108358877B for advancing drug discovery and development pipelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into reliable industrial supply. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities, guaranteeing that every batch of furyl o-diketone derivative meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced metal-free technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget without compromising quality. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate your path to market with high-quality, cost-effective chemical solutions.
