Scalable Chiral Synthesis of Ivosidenib via Protonic Acid Catalysis for Global Pharma Supply Chains
Scalable Chiral Synthesis of Ivosidenib via Protonic Acid Catalysis for Global Pharma Supply Chains
The pharmaceutical industry is constantly seeking robust, scalable, and safe synthetic routes for complex oncology therapeutics like Ivosidenib, a targeted mutant inhibitor of isocitrate dehydrogenase-1 (IDH1). A significant technological breakthrough in this domain is detailed in patent CN111349081B, which discloses a novel chiral synthesis method that fundamentally alters the manufacturing landscape for this critical API intermediate. Unlike conventional approaches that rely on racemic synthesis followed by difficult separation, this invention utilizes a chiral protonic acid catalyst to drive an asymmetric four-component Ugi reaction. This strategic shift not only enhances the stereochemical purity of the final product but also addresses critical supply chain bottlenecks associated with traditional resolution techniques. For procurement and R&D leaders, understanding this transition from resolution-dependent workflows to direct asymmetric catalysis is vital for securing long-term supply stability and cost efficiency in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ivosidenib has been plagued by inefficiencies inherent in racemic pathways. As illustrated in the prior art, the standard process involves a four-component Ugi reaction using achiral reagents in trifluoroethanol, followed by a Buchwald-Hartwig coupling, ultimately yielding a racemic mixture that requires chiral column resolution. 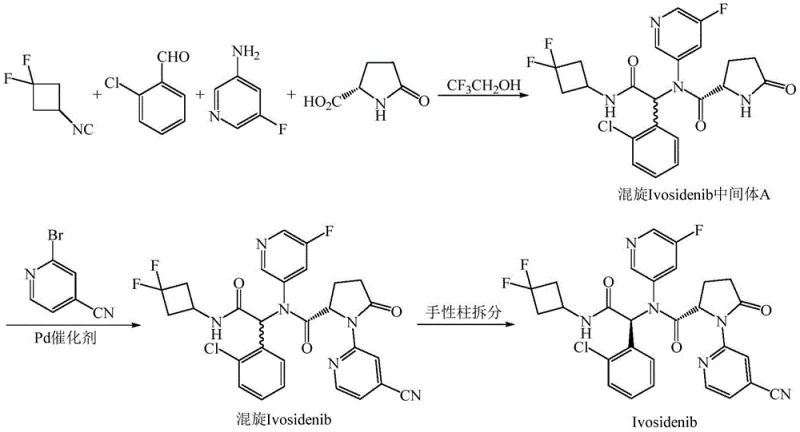 This reliance on chiral chromatography presents a severe bottleneck for commercial scale-up, as preparative chiral columns have limited loading capacities and require expensive stationary phases, drastically increasing the cost of goods sold (COGS). Furthermore, the use of trifluoroethanol as a solvent poses significant regulatory and safety challenges; classified as an ICH Class 4 solvent, it lacks sufficient toxicological data for widespread pharmaceutical use, necessitating rigorous removal steps and complicating waste management protocols. These factors combined create a fragile supply chain vulnerable to delays and quality inconsistencies, making the conventional route suboptimal for meeting the growing global demand for IDH1 inhibitors.
This reliance on chiral chromatography presents a severe bottleneck for commercial scale-up, as preparative chiral columns have limited loading capacities and require expensive stationary phases, drastically increasing the cost of goods sold (COGS). Furthermore, the use of trifluoroethanol as a solvent poses significant regulatory and safety challenges; classified as an ICH Class 4 solvent, it lacks sufficient toxicological data for widespread pharmaceutical use, necessitating rigorous removal steps and complicating waste management protocols. These factors combined create a fragile supply chain vulnerable to delays and quality inconsistencies, making the conventional route suboptimal for meeting the growing global demand for IDH1 inhibitors.
The Novel Approach
In stark contrast, the methodology described in patent CN111349081B introduces a streamlined, catalytic asymmetric approach that bypasses these historical hurdles. By employing a specialized chiral protonic acid catalyst, the synthesis achieves high stereoselectivity directly during the bond-forming Ugi reaction, effectively rendering the chiral column resolution step obsolete. 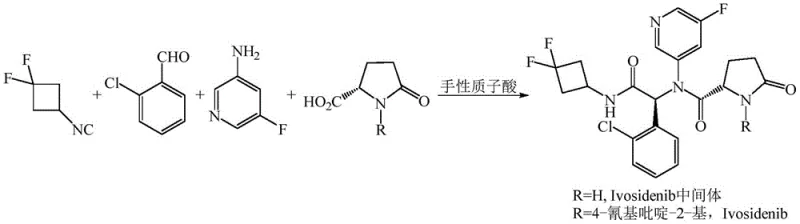 This one-pot transformation integrates the L-pyroglutamic acid derivative, o-chlorobenzaldehyde, 3-amino-5-fluoropyridine, and the isonitrile component under the influence of the chiral catalyst, establishing the critical stereocenter with high fidelity. Moreover, the process allows for the substitution of hazardous trifluoroethanol with safer, more industrially friendly solvents such as dichloromethane, dichloroethane, or toluene. This dual improvement—eliminating the resolution bottleneck and upgrading the solvent system—results in a manufacturing process that is not only chemically superior but also operationally simpler, offering a clear pathway for reliable Ivosidenib intermediate supplier partnerships focused on quality and scalability.
This one-pot transformation integrates the L-pyroglutamic acid derivative, o-chlorobenzaldehyde, 3-amino-5-fluoropyridine, and the isonitrile component under the influence of the chiral catalyst, establishing the critical stereocenter with high fidelity. Moreover, the process allows for the substitution of hazardous trifluoroethanol with safer, more industrially friendly solvents such as dichloromethane, dichloroethane, or toluene. This dual improvement—eliminating the resolution bottleneck and upgrading the solvent system—results in a manufacturing process that is not only chemically superior but also operationally simpler, offering a clear pathway for reliable Ivosidenib intermediate supplier partnerships focused on quality and scalability.
Mechanistic Insights into Chiral Protonic Acid Catalyzed Ugi Reaction
The core innovation driving this synthetic advancement lies in the precise molecular recognition and activation facilitated by the chiral protonic acid catalyst. These catalysts, typically derived from chiral phosphoric acid scaffolds such as SPINOL or BINOL backbones substituted with bulky groups like 2,4,6-triisopropylphenyl or 2,4,6-tricyclohexylphenyl, function through a dual hydrogen-bonding activation mechanism. 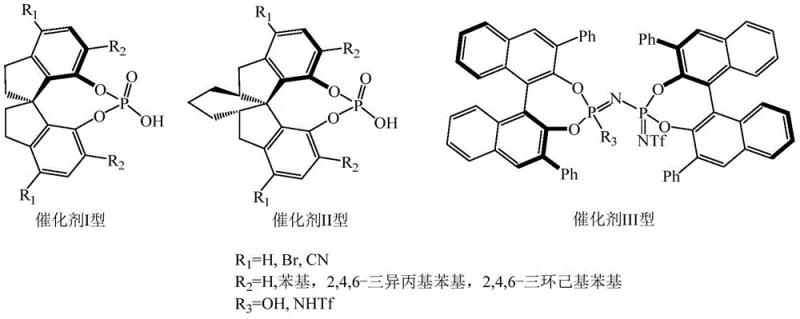 The acidic proton of the catalyst activates the imine intermediate formed in situ from the aldehyde and amine components, while the basic phosphoryl oxygen coordinates with the nucleophilic species, creating a highly organized chiral environment. This rigid transition state restricts the conformational freedom of the reacting species, forcing the attack of the isonitrile and subsequent rearrangement to occur from a specific facial trajectory. Consequently, the reaction yields the desired (S)-configured product with high diastereomeric excess (de value), often exceeding 90% under optimized conditions, thereby minimizing the formation of unwanted stereoisomers that would otherwise complicate downstream purification.
The acidic proton of the catalyst activates the imine intermediate formed in situ from the aldehyde and amine components, while the basic phosphoryl oxygen coordinates with the nucleophilic species, creating a highly organized chiral environment. This rigid transition state restricts the conformational freedom of the reacting species, forcing the attack of the isonitrile and subsequent rearrangement to occur from a specific facial trajectory. Consequently, the reaction yields the desired (S)-configured product with high diastereomeric excess (de value), often exceeding 90% under optimized conditions, thereby minimizing the formation of unwanted stereoisomers that would otherwise complicate downstream purification.
From an impurity control perspective, this catalytic precision is paramount for ensuring the safety and efficacy of the final drug substance. In traditional racemic syntheses, the presence of the opposite enantiomer is a critical quality attribute that must be strictly controlled, often requiring multiple recrystallizations or chromatographic passes which degrade overall yield. The chiral protonic acid approach mitigates this risk at the source by kinetically favoring the formation of the target enantiomer. The specific substituents on the catalyst backbone, such as cyano or bromine groups at the 3,3'-positions, further tune the electronic and steric properties to maximize selectivity. This level of control ensures that the resulting Ivosidenib intermediate meets stringent purity specifications required by global regulatory bodies, reducing the burden on quality control laboratories and accelerating the release of batches for clinical or commercial use.
How to Synthesize Ivosidenib Efficiently
The practical implementation of this chiral synthesis route is designed to be compatible with standard pharmaceutical manufacturing equipment, requiring no specialized high-pressure or cryogenic infrastructure beyond typical cooling capabilities. The process begins with the preparation of the reaction vessel under an inert nitrogen atmosphere to prevent moisture interference, followed by the sequential addition of the L-pyroglutamic acid derivative, aldehyde, amine, and the chiral catalyst in a selected solvent. Detailed standardized synthesis steps see the guide below.
- Combine L-pyroglutamic acid derivative, o-chlorobenzaldehyde, 3-amino-5-fluoropyridine, and a chiral phosphoric acid catalyst in a suitable solvent such as dichloromethane or toluene under inert atmosphere.
- Stir the mixture at ambient temperature to form the imine intermediate, then cool the reaction system to a controlled low temperature ranging from -30°C to 5°C to optimize stereoselectivity.
- Slowly add 1,1-difluoro-3-isocyclobutyronitrile dropwise, allow the reaction to proceed overnight, and subsequently purify the crude product via recrystallization to obtain optically pure Ivosidenib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chiral catalytic technology translates into tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational complexity and enhanced supply reliability. By removing the chiral column resolution step, manufacturers can significantly increase batch sizes and throughput, as the process is no longer limited by the loading capacity of chromatographic columns. This scalability ensures a more consistent supply of high-purity pharmaceutical intermediates, mitigating the risk of stockouts that can disrupt downstream drug formulation and patient access. Furthermore, the elimination of expensive chiral stationary phases and the associated solvent consumption for chromatography leads to substantial cost savings in raw materials and waste disposal, optimizing the overall cost structure of the supply chain.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the removal of the most expensive unit operation in the traditional route: chiral chromatography. Preparative chiral columns represent a significant capital and operational expense due to the high cost of the silica-based stationary phases and the large volumes of solvent required for elution. By achieving high enantioselectivity directly through catalysis, the new process eliminates this cost center entirely. Additionally, the replacement of trifluoroethanol with commodity solvents like dichloromethane or toluene further reduces raw material costs and simplifies solvent recovery systems. These cumulative efficiencies result in a leaner manufacturing process that offers a more competitive pricing structure for bulk procurement without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the robustness of the underlying chemical process. Traditional methods relying on chiral resolution are inherently prone to variability; slight fluctuations in column performance or mobile phase composition can lead to failed batches or off-spec material. The catalytic asymmetric Ugi reaction described in the patent offers a more deterministic outcome, where the stereochemistry is controlled by the molecular structure of the catalyst rather than physical separation dynamics. This predictability allows for tighter production scheduling and more accurate lead time estimations. Moreover, the use of widely available, non-proprietary solvents reduces dependency on niche solvent suppliers, further de-risking the supply chain against market volatility and logistical disruptions.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental footprint of synthetic routes has become a key decision factor. The new method aligns with green chemistry principles by avoiding ICH Class 4 solvents and reducing the total volume of organic waste generated per kilogram of product. The absence of chromatographic purification means less solvent waste and lower energy consumption for solvent evaporation and recovery. This improved environmental profile facilitates easier regulatory approval and compliance with increasingly strict environmental, social, and governance (ESG) mandates. From a scale-up perspective, the reaction conditions are mild and operate within standard temperature ranges, making the transfer from pilot plant to multi-ton commercial production straightforward and technically feasible for established CDMO partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific supply chain needs. Understanding these nuances is essential for R&D teams assessing technology transfer and procurement teams negotiating long-term supply agreements.
Q: How does the chiral protonic acid catalyst improve the Ivosidenib synthesis process compared to traditional methods?
A: The chiral protonic acid catalyst enables a direct asymmetric Ugi four-component reaction, which establishes the chiral center in a single step. This eliminates the need for a post-synthesis chiral column resolution, significantly simplifying the workflow and improving overall yield efficiency.
Q: What are the safety advantages of the solvent system used in this new synthetic route?
A: This novel method replaces trifluoroethanol, an ICH Class 4 solvent with potential toxicity concerns, with safer hydrocarbon or halogenated hydrocarbon solvents like dichloromethane or toluene. This substitution aligns better with green chemistry principles and regulatory safety standards for pharmaceutical manufacturing.
Q: Can this synthesis method be scaled for industrial production without chromatographic purification?
A: Yes, the process is specifically designed for industrial scalability. By achieving high diastereomeric excess (de value) directly through catalysis and utilizing recrystallization for final purification, the method avoids the throughput limitations and high costs associated with preparative chiral chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ivosidenib Supplier
The chiral synthesis route detailed in patent CN111349081B represents a paradigm shift in the manufacturing of IDH1 inhibitors, offering a blend of high stereocontrol and operational simplicity that is ideal for modern pharmaceutical supply chains. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex catalytic processes like this asymmetric Ugi reaction are executed with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including advanced chiral HPLC analysis to guarantee the enantiomeric excess of every batch. We understand that the transition to a new synthetic route requires confidence in both the chemistry and the partner executing it, and our track record in delivering high-purity pharmaceutical intermediates underscores our commitment to excellence.
We invite global pharmaceutical companies and biotech firms to engage with our technical procurement team to explore how this advanced synthesis method can optimize your Ivosidenib supply chain. By leveraging our expertise in process development and scale-up, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to request specific COA data and route feasibility assessments to validate the technical merits of this catalytic approach for your portfolio. Partnering with us ensures access to a reliable Ivosidenib supplier dedicated to driving innovation, reducing costs, and securing the continuity of your critical oncology therapies.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
