Advanced Synthesis of N-Boc-trans-1,4-cyclohexanediamine for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for chiral building blocks that balance high purity with economic feasibility. Patent CN112898178A introduces a transformative preparation method for N-Boc-trans-1,4-cyclohexanediamine, a critical intermediate in the synthesis of kinase inhibitors and poly ADP ribosylation protein inhibitors. Unlike conventional approaches that rely on the wasteful excess of expensive starting materials, this innovation employs a strategic benzophenone protection sequence. By temporarily masking one amino group as an imine, the process ensures precise mono-Boc substitution without requiring molar excesses of the diamine backbone. This technical breakthrough not only optimizes atom economy but also establishes a foundation for more sustainable and cost-effective pharmaceutical intermediates manufacturing, addressing key pain points for both R&D and procurement teams globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of mono-protected cyclohexanediamines has been plagued by inefficiency and high raw material costs. Standard literature methods typically depend on using a significant molar excess of trans-1,4-cyclohexanediamine to statistically favor mono-substitution over di-substitution when reacting with Boc anhydride. This approach creates a substantial economic burden because the diamine is a high-value chiral starting material. Furthermore, separating the desired mono-protected product from unreacted diamine and di-protected byproducts often requires complex purification steps, which lowers overall yield and increases solvent consumption. For reliable pharmaceutical intermediates supplier networks, these inefficiencies translate into volatile pricing and inconsistent supply continuity, making it difficult to forecast production costs accurately for downstream API manufacturing.
The Novel Approach
The methodology disclosed in CN112898178A circumvents these statistical limitations through a deterministic protection strategy. Instead of relying on excess diamine, the process first reacts the diamine with benzophenone to form a mono-imine intermediate, effectively blocking one reactive site. This allows the subsequent Boc protection to occur selectively on the remaining free amine with high precision. Finally, the benzophenone group is cleaved under mild Lewis acid conditions to reveal the target molecule. This sequence ensures that every mole of the expensive diamine starting material is utilized efficiently, drastically reducing waste. 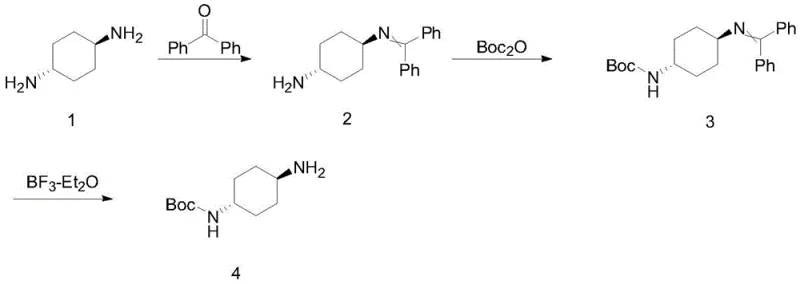 This streamlined pathway facilitates cost reduction in pharmaceutical intermediates manufacturing by minimizing raw material input while maintaining high reaction selectivity and ease of isolation.
This streamlined pathway facilitates cost reduction in pharmaceutical intermediates manufacturing by minimizing raw material input while maintaining high reaction selectivity and ease of isolation.
Mechanistic Insights into Benzophenone-Mediated Selective Protection
The core of this synthetic innovation lies in the reversible formation of the benzophenone imine. In the first step, trans-1,4-cyclohexanediamine condenses with benzophenone in the presence of a catalyst like p-toluenesulfonic acid and a water scavenger such as trimethyl orthoformate. This reaction proceeds under reflux in toluene, driving the equilibrium towards the imine intermediate (Compound 2) by removing water azeotropically. The steric bulk of the benzophenone group effectively shields one nitrogen atom, preventing it from participating in subsequent acylation reactions. This mechanistic control is crucial for achieving high regioselectivity without the need for cryogenic conditions or exotic reagents, making the process highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Following the Boc protection of the free amine, the final deprotection step utilizes boron trifluoride diethyl etherate (BF3·Et2O) as a Lewis acid catalyst. This reagent coordinates with the nitrogen lone pair of the imine, increasing its electrophilicity and facilitating hydrolysis or alcoholysis in solvents like methanol or ethanol. 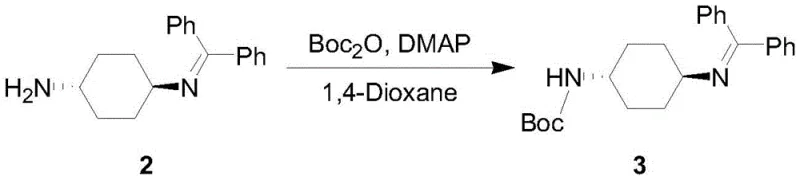 This mild cleavage condition is particularly advantageous because it avoids harsh acidic or basic environments that could compromise the stability of the Boc group or induce racemization. The result is a high-purity product with minimal impurity profiles, satisfying the rigorous quality standards required for high-purity pharmaceutical intermediates used in sensitive drug synthesis pathways.
This mild cleavage condition is particularly advantageous because it avoids harsh acidic or basic environments that could compromise the stability of the Boc group or induce racemization. The result is a high-purity product with minimal impurity profiles, satisfying the rigorous quality standards required for high-purity pharmaceutical intermediates used in sensitive drug synthesis pathways.
How to Synthesize N-Boc-trans-1,4-cyclohexanediamine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. It begins with the condensation of diamine and benzophenone, followed by Boc protection in dichloromethane with aqueous base, and concludes with the Lewis acid-mediated deprotection. Each step is optimized for yield and purity, with specific attention paid to temperature control and stoichiometry to prevent side reactions. For detailed operational parameters, reagent grades, and workup procedures necessary for replication, please refer to the standardized guide below.
- Condense trans-1,4-cyclohexanediamine with benzophenone using trimethyl orthoformate and p-toluenesulfonic acid in toluene to form the mono-protected imine intermediate.
- React the imine intermediate with di-tert-butyl dicarbonate (Boc2O) in dichloromethane under alkaline conditions to install the Boc protecting group on the free amine.
- Perform selective deprotection of the benzophenone imine moiety using boron trifluoride diethyl etherate in an alcohol solvent to yield the final N-Boc-trans-1,4-cyclohexanediamine.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this benzophenone protection strategy offers significant logistical and financial benefits. By eliminating the need for excess diamine, manufacturers can stabilize their raw material inventory requirements and reduce the capital tied up in unused starting materials. The simplified purification process also means shorter batch cycles and reduced solvent waste disposal costs, contributing to a leaner manufacturing operation. These efficiencies directly support reducing lead time for high-purity pharmaceutical intermediates, allowing suppliers to respond more agilely to market demand fluctuations without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the stoichiometric utilization of trans-1,4-cyclohexanediamine. In traditional processes, the cost of recovering or discarding excess diamine significantly inflates the unit price. By switching to a masking strategy, the material cost per kilogram of the final product is substantially lowered. Additionally, the use of common solvents like toluene, DCM, and methanol ensures that procurement teams can source materials easily without relying on specialized or hazardous reagents, further optimizing the overall cost structure.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to higher batch success rates and consistent output. Traditional methods that rely on precise control of excess reagents can be sensitive to mixing efficiency and addition rates, leading to batch-to-batch variability. This new method's reliance on thermodynamic control via imine formation makes the process more forgiving and reproducible. For supply chain heads, this translates to greater predictability in delivery schedules and a reduced risk of production delays caused by failed batches or extensive rework.
- Scalability and Environmental Compliance: The process operates at moderate temperatures and avoids the use of heavy metal catalysts or highly toxic reagents. The workup involves standard aqueous washes and crystallization or pulping steps, which are easily scalable from kilograms to metric tons. Furthermore, the improved atom economy means less chemical waste is generated per unit of product, aligning with increasingly strict environmental regulations. This sustainability profile enhances the long-term viability of the supply chain and reduces the regulatory burden associated with waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this intermediate. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring accuracy and relevance for technical decision-makers evaluating this synthesis route for potential integration into their supply chains.
Q: How does this method improve atom economy compared to traditional synthesis?
A: Traditional methods often require a large excess of the expensive trans-1,4-cyclohexanediamine to control mono-substitution. This patented approach uses a benzophenone masking strategy, allowing for stoichiometric usage of the diamine, thereby significantly reducing raw material waste and cost.
Q: What is the achieved purity level of the final product?
A: The described process consistently yields N-Boc-trans-1,4-cyclohexanediamine with an HPLC purity of up to 99.4%, meeting stringent requirements for pharmaceutical intermediate applications without extensive recrystallization.
Q: Is the deprotection step compatible with large-scale production?
A: Yes, the deprotection utilizes boron trifluoride diethyl etherate in common alcohol solvents like methanol or ethanol at moderate temperatures (40-60°C), ensuring safe and scalable operation suitable for industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Boc-trans-1,4-cyclohexanediamine Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from patent theory to commercial reality requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of N-Boc-trans-1,4-cyclohexanediamine meets the exacting standards required for global pharmaceutical applications. We are committed to delivering not just a product, but a reliable supply solution that supports your drug development timelines.
We invite you to collaborate with us to leverage this advanced synthesis technology for your projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing capabilities can enhance the efficiency and profitability of your supply chain.
