Scalable Production of Optically Active Synthon A via Advanced Asymmetric Hydrogenation Technology
Introduction to Advanced Synthon A Manufacturing
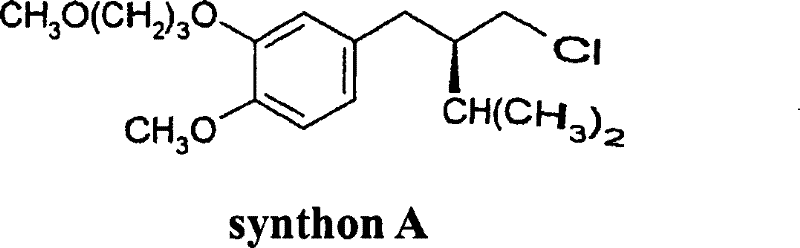
The pharmaceutical industry's demand for high-purity chiral intermediates has driven significant innovation in asymmetric synthesis, particularly for critical antihypertensive agents. Patent CN101142164B introduces a groundbreaking method for the preparation of optically active 3-phenylpropionic acid derivatives, specifically targeting the synthesis of "Synthon A," a vital precursor for the renin inhibitor Aliskiren. This technology addresses long-standing inefficiencies in chiral pool synthesis by enabling the direct utilization of cis/trans isomer mixtures without prior rigorous separation. By leveraging advanced planar chiral diphosphine ligands, the process achieves exceptional stereocontrol and operational simplicity. For global supply chain leaders, this represents a pivotal shift towards more robust and economically viable manufacturing routes for complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (R)-2-alkyl-3-phenylpropionic acids relied heavily on the asymmetric hydrogenation of pure trans-3-phenylacrylic acid derivatives. As illustrated in prior art such as WO 02/02500, this conventional approach necessitates the isolation of the trans-isomer through repeated extractions and crystallizations, which is technically demanding and yield-loss prone. Furthermore, the catalysts employed in these legacy methods, often based on phenylferrocenyl phosphine ligands, suffer from low substrate-to-catalyst ratios (s/c) of approximately 5700:1. This limitation forces manufacturers to use relatively large quantities of expensive precious metal catalysts to achieve modest enantiomeric excess values around 95% ee, rendering the process economically inefficient for large-scale operations.

The Novel Approach
In stark contrast, the methodology disclosed in CN101142164B revolutionizes the workflow by accepting crude cis/trans isomer mixtures directly from upstream elimination reactions. The novel process tolerates significant amounts of trans-isomers (at least 1% to 10% by weight) within the feedstock, eliminating the need for complex purification steps before hydrogenation. This flexibility allows for the use of industrial purity grade mixtures containing at least 80% combined cis and trans isomers. By removing the bottleneck of isomer separation, the new approach drastically streamlines the production timeline and reduces solvent consumption, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing while maintaining rigorous quality standards.
Mechanistic Insights into Planar Chiral Diphosphine Catalysis
The core of this technological breakthrough lies in the utilization of transition metal complexes featuring planar chiral diphosphine ligands with a ring-aromatic backbone, such as Phanephos derivatives. These ligands create a highly specific chiral environment around the rhodium center, facilitating the enantioselective hydrogenation of the olefinic bond even in the presence of geometric isomer impurities. The steric and electronic properties of ligands like (R)-Phanephos or its xylyl-substituted analogs ensure that the catalyst preferentially binds and reduces the substrate to the desired (R)-configuration. This mechanistic robustness allows the reaction to proceed efficiently at remarkably high substrate-to-catalyst ratios, exceeding 10,000:1 and potentially reaching 30,000:1, which is a substantial improvement over traditional ferrocenyl-based systems.
Beyond the catalytic step, the process incorporates a sophisticated enantiomeric enrichment mechanism via crystallization. Even if the initial hydrogenation yields an enantiomeric excess of roughly 80-86%, the subsequent addition of basic salt formers induces selective crystallization of the desired enantiomer. Surprisingly, achiral bases such as ammonia or lithium hydroxide are effective in this role, forming salts that precipitate with extremely high optical purity (≥98% ee). This dual-stage purification strategy—combining high-performance catalysis with thermodynamic crystallization—ensures that the final product meets the stringent purity specifications required for active pharmaceutical ingredient (API) synthesis without the need for costly chiral chromatography.
How to Synthesize Synthon A Efficiently
The synthesis of Synthon A via this patented route involves a sequence of well-defined chemical transformations starting from aromatic aldehydes. The process begins with an aldol-type condensation to form the acrylic acid derivative, followed by the critical asymmetric hydrogenation step described above. The resulting acid is then subjected to salt formation and crystallization to boost optical purity, followed by protonation to recover the free acid. For detailed operational parameters including temperature profiles, pressure settings, and specific reagent quantities, please refer to the standardized synthesis guide below which outlines the critical control points for ensuring batch-to-batch consistency.
- Prepare a mixture of cis/trans isomers of 3-phenylacrylic acid derivatives, preferably containing at least 40% cis-isomer, without requiring complex purification of the trans-isomer.
- Perform enantioselective hydrogenation using a chiral rhodium catalyst with planar chiral diphosphine ligands (e.g., Phanephos derivatives) at high substrate-to-catalyst ratios.
- Enrich the optical purity of the hydrogenated product by crystallizing the salt formed with a basic agent like ammonia or lithium hydroxide, achieving >98% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical feasibility. The ability to bypass the isolation of pure trans-isomers significantly reduces the number of unit operations, thereby lowering capital expenditure requirements and minimizing waste generation. This streamlined workflow translates directly into a more resilient supply chain, as fewer processing steps mean fewer potential points of failure or delay. Additionally, the high efficiency of the catalyst system reduces the dependency on scarce precious metals, mitigating risks associated with raw material price volatility and ensuring long-term cost stability for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive separation processes for trans-isomers and the ability to operate at high substrate-to-catalyst ratios significantly lower the overall cost of goods sold. By reducing the catalyst loading from typical industry standards to levels where s/c ratios exceed 10,000:1, the consumption of expensive rhodium complexes is minimized. Furthermore, the use of simple, achiral bases like ammonia for crystallization avoids the cost of specialized chiral resolving agents, driving down material costs substantially while maintaining high yield efficiency throughout the production cycle.
- Enhanced Supply Chain Reliability: The robustness of the process against feedstock variations ensures consistent output quality even when starting material purity fluctuates. Since the method tolerates cis/trans mixtures directly from elimination reactions, suppliers can secure raw materials more easily without demanding ultra-high purity specifications from upstream vendors. This flexibility shortens lead times for high-purity pharmaceutical intermediates and enhances the reliability of delivery schedules, allowing manufacturing partners to maintain steady inventory levels without the risk of production stoppages due to raw material bottlenecks.
- Scalability and Environmental Compliance: The patent demonstrates successful scale-up from laboratory gram-scale to multi-hundred kilogram batches, proving the technology's readiness for commercial scale-up of complex pharmaceutical intermediates. The process supports continuous hydrogenation using cascade reactor systems, which improves heat management and safety profiles. Moreover, the reduction in solvent usage and waste streams associated with fewer purification steps aligns with modern green chemistry principles, simplifying environmental compliance and reducing the burden of waste treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy for R&D and technical procurement teams evaluating this route for potential licensing or contract manufacturing opportunities.
Q: What is the advantage of using cis/trans isomer mixtures in this synthesis?
A: The patented method eliminates the need for difficult and costly separation of trans-isomers. It allows the direct use of crude isomer mixtures containing significant amounts of trans-isomers (up to 30% or more), simplifying the upstream synthesis and reducing overall production costs.
Q: How is high optical purity (ee) achieved if the hydrogenation yields only ~80-86% ee?
A: The process employs a subsequent crystallization step using basic salt formers such as ammonia or lithium hydroxide. This step dramatically enriches the enantiomeric excess from the initial hydrogenation levels (e.g., 83-86%) to greater than 98%, often reaching 99.2% or higher in the final isolated solid.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the patent explicitly describes successful scale-up from gram-scale laboratory experiments to multi-hundred kilogram batches (e.g., 300 kg scale). It also supports continuous hydrogenation processes using cascade reactor systems, ensuring viability for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Synthon A Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient chiral synthesis in the development of next-generation cardiovascular therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels that meet or exceed global pharmacopeial standards for API intermediates.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in asymmetric catalysis can optimize your production costs and secure your supply of high-quality Synthon A.
