Scalable Synthesis of Benzopyran LTB4 Antagonists for Advanced Inflammatory Disease Therapeutics
Scalable Synthesis of Benzopyran LTB4 Antagonists for Advanced Inflammatory Disease Therapeutics
The pharmaceutical industry continuously seeks robust and scalable pathways for producing potent anti-inflammatory agents, particularly those targeting leukotriene B4 (LTB4) receptors. Patent CN1041311C discloses a sophisticated methodology for the preparation of novel benzopyran and other benzo-fused LTB4 antagonists, which hold significant promise for treating conditions such as rheumatoid arthritis, psoriasis, and asthma. This intellectual property outlines a versatile synthetic strategy that allows for the precise installation of diverse substituents on the benzopyran core, enabling the fine-tuning of pharmacological properties. The disclosed compounds feature a specific structural motif where the stereochemistry at the chiral centers is critical for biological activity, necessitating a synthesis capable of high enantiomeric excess. By leveraging advanced organometallic coupling techniques, this patent provides a foundation for developing next-generation therapeutics with improved safety profiles and efficacy.
For procurement managers and supply chain directors, understanding the underlying chemistry is essential for evaluating the reliability of a potential supplier. The ability to consistently produce high-purity intermediates without excessive waste or hazardous byproducts is a key determinant of long-term supply stability. The methods described in this patent utilize widely available starting materials and standard catalytic systems, which mitigates the risk of raw material shortages. Furthermore, the modular nature of the synthesis allows for the rapid adaptation of the process to produce various analogues, ensuring that supply chains remain resilient against shifting market demands for specific drug candidates. This flexibility is crucial for maintaining continuity in the production of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing substituted benzopyran scaffolds often rely on harsh reaction conditions that can compromise product integrity and yield. Classical methods frequently involve multiple protection and deprotection steps, which not only extend the overall processing time but also introduce additional opportunities for impurity formation. In many legacy processes, the introduction of aryl groups onto the heterocyclic core requires aggressive nucleophilic substitutions that demand high temperatures and strong bases, leading to poor regioselectivity and difficult purification challenges. These inefficiencies result in higher manufacturing costs and increased environmental burden due to the generation of substantial chemical waste. Additionally, controlling the stereochemistry at the benzylic positions using conventional reduction methods often yields racemic mixtures, necessitating costly and material-intensive resolution steps later in the sequence.
The Novel Approach
In contrast, the approach detailed in the patent data utilizes a modern palladium-catalyzed cross-coupling strategy that dramatically streamlines the construction of the target molecule. By employing organozinc or organotin reagents in conjunction with palladium catalysts, the synthesis achieves efficient carbon-carbon bond formation under relatively mild thermal conditions. This method bypasses the need for extreme reaction parameters, thereby preserving sensitive functional groups and minimizing side reactions. The use of triflate leaving groups activates the aromatic ring effectively, allowing for high-yielding coupling events that are tolerant of various substituents. This novel pathway not only simplifies the operational complexity but also enhances the overall atom economy of the process. Consequently, manufacturers can achieve superior purity profiles with fewer downstream purification requirements, directly translating to cost efficiencies and improved throughput.
Mechanistic Insights into Palladium-Catalyzed Coupling and Stereoselective Reduction
The core of this synthetic innovation lies in the mechanistic elegance of the transition metal-catalyzed coupling reaction. The process typically begins with the activation of a halogenated or triflated benzopyran precursor, which serves as the electrophilic partner. Upon exposure to a palladium(0) catalyst, oxidative addition occurs, forming a reactive organopalladium intermediate. This species then undergoes transmetallation with the nucleophilic organometallic reagent, such as an arylzinc or arylstannane derivative, transferring the aryl group to the palladium center. The final step involves reductive elimination, which releases the coupled product and regenerates the active palladium catalyst to continue the cycle. This catalytic cycle is highly efficient and allows for the precise assembly of complex molecular architectures that would be difficult to access via classical organic synthesis.
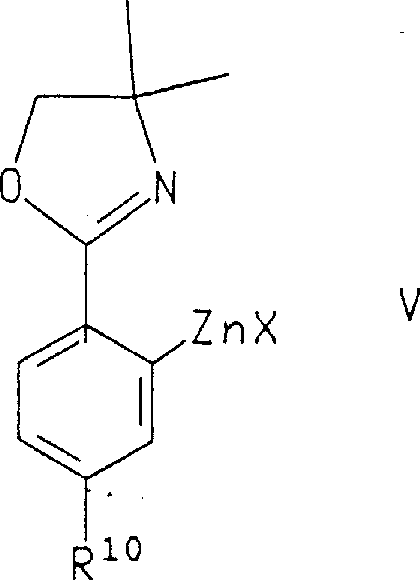
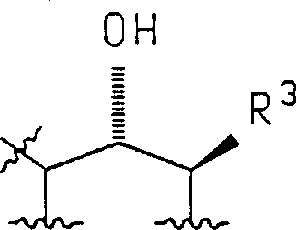
Following the construction of the carbon skeleton, the control of stereochemistry becomes paramount for ensuring biological potency. The reduction of the ketone functionality to the corresponding alcohol introduces two chiral centers, resulting in a mixture of cis and trans diastereomers. The patent describes the use of hydride reducing agents, such as sodium borohydride, which can be tuned to favor specific stereoisomers based on solvent choice and temperature. However, since a mixture is often obtained, the process incorporates robust resolution strategies. These may involve the formation of diastereomeric salts using chiral bases like methylbenzylamine or enzymatic resolution techniques. The ability to separate these isomers effectively ensures that the final active pharmaceutical ingredient meets stringent regulatory standards for enantiomeric purity, which is a critical quality attribute for LTB4 antagonists.
How to Synthesize Benzopyran Derivatives Efficiently
Executing this synthesis requires careful attention to reaction parameters to maximize yield and purity. The process generally initiates with the cyclization of substituted resorcinols to form the chromanone core, followed by activation with triflic anhydride. The subsequent coupling step is the pivotal transformation, requiring anhydrous conditions and inert atmosphere handling to maintain catalyst activity. Once the coupled ketone is obtained, the reduction step must be monitored closely to manage the ratio of stereoisomers produced. Finally, the resolution and hydrolysis steps convert the intermediate esters or salts into the free acid form of the drug substance. Detailed standard operating procedures are essential for replicating these results on a commercial scale, ensuring consistency across batches.
- Preparation of the chromanone core followed by triflation to activate the aromatic ring for coupling.
- Palladium-catalyzed cross-coupling with organometallic reagents (zinc or tin) to introduce the aryl side chain.
- Stereoselective reduction of the ketone and subsequent chiral resolution to isolate the active enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for organizations focused on cost reduction in pharmaceutical intermediate manufacturing. The reliance on palladium catalysis, while involving a precious metal, is offset by the high turnover numbers and the ability to recover and recycle the catalyst in many modern setups. More importantly, the elimination of multiple protection-deprotection sequences significantly reduces the consumption of reagents and solvents, leading to a leaner and more cost-effective process. The use of commodity chemicals such as resorcinol and simple alkyl halides as starting materials ensures a stable and predictable supply chain, shielding manufacturers from the volatility associated with exotic or proprietary reagents. This stability is vital for long-term planning and budget forecasting in the competitive pharmaceutical landscape.
- Cost Reduction in Manufacturing: The streamlined nature of the coupling reaction eliminates several unit operations that are typically required in traditional syntheses. By reducing the total number of steps, the process inherently lowers labor costs, energy consumption, and equipment usage time. Furthermore, the high selectivity of the palladium-catalyzed step minimizes the formation of difficult-to-remove impurities, which reduces the burden on purification resources like chromatography columns or crystallization vessels. This efficiency gain allows for a significant optimization of the cost of goods sold (COGS), making the final API more economically viable for mass production without compromising on quality standards.
- Enhanced Supply Chain Reliability: The raw materials identified in this patent, such as substituted phenols and common organometallic reagents, are produced by a wide network of global chemical suppliers. This diversification of the supply base reduces the risk of single-source dependency, which is a common vulnerability in complex drug synthesis. Additionally, the robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality or environmental factors, ensuring consistent output. For supply chain heads, this translates to reduced lead times for high-purity pharmaceutical intermediates and a lower probability of production delays caused by material shortages or failed batches.
- Scalability and Environmental Compliance: The chemistry described is inherently scalable, moving seamlessly from laboratory benchtop experiments to pilot plant and full commercial production. The reactions do not require extreme pressures or temperatures that would necessitate specialized high-cost reactor infrastructure. Moreover, the improved atom economy and reduced solvent usage align well with modern green chemistry principles and environmental regulations. By generating less hazardous waste and utilizing safer reagents, manufacturers can simplify their waste management protocols and reduce the environmental footprint of their operations. This compliance advantage is increasingly important for maintaining operational licenses and meeting the sustainability goals of major pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of these benzopyran derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details helps stakeholders assess the feasibility and advantages of implementing this technology in their own production pipelines. The focus is on practical implementation, quality control, and the strategic benefits of this specific chemical pathway.
Q: What are the key advantages of the palladium-catalyzed route described in CN1041311C?
A: The process utilizes robust palladium catalysis to form carbon-carbon bonds under mild conditions, significantly reducing reaction times and improving overall yield compared to traditional nucleophilic substitutions.
Q: How is stereochemical purity controlled in this synthesis?
A: Stereocontrol is achieved through a combination of selective reduction conditions and classical resolution techniques using chiral acids or bases to separate diastereomeric salts.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the use of commercially available starting materials like resorcinol derivatives and standard catalytic systems makes the route highly adaptable for kilogram to ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzopyran Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the production of complex pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to delivering products that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in palladium-catalyzed reactions and chiral resolution allows us to optimize the synthesis of benzopyran antagonists, guaranteeing consistent quality and batch-to-batch reproducibility that meets global regulatory standards.
We invite you to collaborate with us to leverage these advanced synthetic capabilities for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing excellence can accelerate your path to market while optimizing your overall production costs.
