Scalable One-Pot Synthesis of 2-Deoxy-Beta-D-Glucopyranosyl Triazoles for Advanced Drug Discovery
Scalable One-Pot Synthesis of 2-Deoxy-Beta-D-Glucopyranosyl Triazoles for Advanced Drug Discovery
In the rapidly evolving landscape of medicinal chemistry, the efficient construction of glycosylated heterocycles remains a pivotal challenge for developing next-generation enzyme inhibitors. Patent CN102276673A introduces a transformative methodology for the preparation of 2-deoxy-beta-D-glucopyranosyl triazole compounds, leveraging a robust one-pot copper-catalyzed azide-alkyne cycloaddition (CuAAC) strategy. This innovation addresses critical bottlenecks in carbohydrate chemistry by utilizing 1,2-deoxy-7-C-acetyl-alpha-D-glucopyranose derivatives as stable substrates, reacting them with sodium azide to generate reactive azide intermediates in situ. Subsequently, without the need for purification, terminal alkynes and a cuprous iodide catalyst are introduced to forge the triazole ring with exceptional regioselectivity. This approach not only streamlines the synthetic workflow but also significantly enhances the overall atom economy, making it an attractive route for reliable pharmaceutical intermediate supplier networks seeking to optimize their production pipelines for bioactive sugar analogues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of glycotriazoles has been plagued by inefficient multi-step protocols that rely on hazardous reagents and environmentally damaging solvents. As illustrated in prior art such as the method described in Tetrahedron Letters (2007), traditional routes often necessitate the use of expensive catalysts or complex protecting group manipulations that drive up the cost of goods significantly. Furthermore, alternative pathways documented in literature, such as those involving bromine-mediated activation, pose severe environmental and safety risks due to the generation of toxic halogenated waste streams that require costly disposal procedures. Another significant drawback in existing methodologies is the reliance on catalysts that are difficult to source or stabilize, leading to inconsistent batch-to-batch reproducibility which is unacceptable for GMP manufacturing environments. These cumulative inefficiencies result in prolonged lead times and reduced throughput, creating a substantial barrier for the commercial scale-up of complex polymer additives or drug candidates derived from these scaffolds.
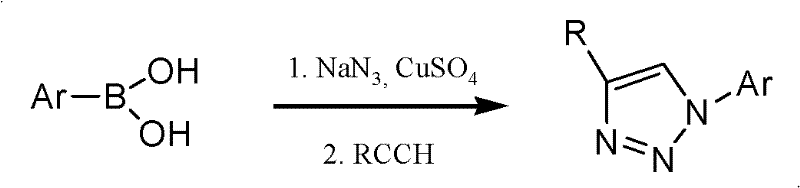
The Novel Approach
The methodology disclosed in CN102276673A represents a paradigm shift by integrating the azide formation and cycloaddition steps into a seamless one-pot operation within a single reactor. By employing polyethylene glycol 400 (PEG-400) as the reaction medium, the process capitalizes on a solvent that is not only non-toxic and biodegradable but also facilitates the dissolution of both organic substrates and inorganic salts like sodium azide. This eliminates the need for volatile organic solvents, thereby reducing the fire hazard and exposure risks associated with traditional organic synthesis. The use of readily available cuprous iodide (CuI) as the catalyst ensures that the reaction proceeds under mild thermal conditions, typically requiring heating to only 80°C for the initial azidation followed by room temperature for the cycloaddition. This gentle thermal profile preserves the integrity of sensitive carbohydrate moieties, preventing degradation and ensuring high-purity pharmaceutical intermediates are obtained with minimal downstream purification efforts.
Mechanistic Insights into CuI-Catalyzed Azide-Alkyne Cycloaddition
The core of this synthetic breakthrough lies in the precise orchestration of the copper-catalyzed azide-alkyne cycloaddition (CuAAC) mechanism, which is renowned for its click chemistry characteristics. In this specific transformation, the cuprous iodide catalyst activates the terminal alkyne to form a copper-acetylide species, which then undergoes a concerted cycloaddition with the in situ generated glycosyl azide. This mechanism is highly regioselective, exclusively yielding the 1,4-disubstituted 1,2,3-triazole isomer, which is crucial for maintaining the biological activity of the resulting enzyme inhibitors. The presence of the acetyl group at the C-7 position of the glucose scaffold plays a vital role in stabilizing the transition state and influencing the stereochemical outcome at the anomeric center, ensuring the formation of the desired beta-configuration. Understanding this mechanistic pathway allows process chemists to fine-tune reaction parameters, such as the molar ratio of sodium azide to substrate (optimized between 1:1.2 to 1:1.5), to maximize conversion while minimizing the formation of unreacted starting materials or side products.
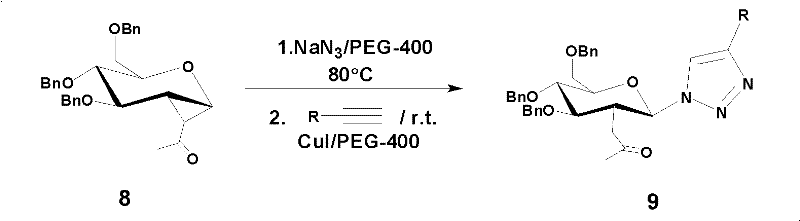
Impurity control in this system is inherently managed by the choice of solvent and the one-pot nature of the reaction. PEG-400 acts as a phase-transfer medium that keeps the inorganic azide salt in close proximity to the organic sugar derivative, facilitating rapid conversion to the azide intermediate before it can decompose. Furthermore, because the azide is not isolated, the risk of handling potentially explosive organic azides on a large scale is mitigated, enhancing operational safety. The subsequent addition of the alkyne and catalyst occurs in the same medium, where the copper species coordinates with the triazole product, often aiding in its precipitation or ease of extraction. This intrinsic self-purification effect reduces the burden on chromatographic separation, allowing for the isolation of high-purity compounds with yields ranging from 67% to 82% across various substituted alkynes, demonstrating the robustness of the catalytic cycle against electronic variations in the alkyne partner.
How to Synthesize 2-Deoxy-Beta-D-Glucopyranosyl Triazoles Efficiently
Implementing this synthesis requires strict adherence to the optimized stoichiometric ratios and thermal profiles established in the patent data to ensure consistent quality. The process begins with the dissolution of the 1,2-deoxy-7-C-acetyl-alpha-D-glucopyranose derivative and sodium azide in PEG-400, followed by heating to promote nucleophilic substitution. Once the azide formation is complete, typically monitored by TLC, the reaction mixture is cooled, and the alkyne component along with the CuI catalyst is introduced. The detailed standardized synthesis steps, including specific workup procedures and purification protocols using petroleum ether and ethyl acetate gradients, are outlined below to guide process development teams in replicating these results accurately.
- React 1,2-deoxy-7-C-acetyl-alpha-D-glucopyranose derivative with sodium azide in PEG-400 at 80°C for 10 hours to form the azide intermediate.
- Without isolation, add terminal alkyne and CuI catalyst to the same reactor at room temperature.
- Stir until completion, extract with ethyl acetate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers profound strategic benefits that extend beyond mere technical feasibility. The elimination of intermediate isolation steps drastically reduces the total processing time and labor costs associated with multiple filtration and drying operations, leading to substantial cost savings in API manufacturing. By utilizing PEG-400, a commodity chemical with a stable global supply chain, the process avoids reliance on exotic or fluctuating specialty solvents, thereby enhancing supply chain reliability and reducing the risk of production stoppages due to raw material shortages. Furthermore, the mild reaction conditions and the use of non-corrosive reagents minimize equipment wear and tear, extending the lifecycle of reactor vessels and reducing maintenance expenditures over the long term.
- Cost Reduction in Manufacturing: The one-pot design inherently lowers the cost of goods by consolidating two distinct chemical transformations into a single vessel operation, which significantly reduces solvent consumption and energy usage. Since the process avoids the use of expensive ligands or precious metal catalysts like palladium or rhodium, the raw material costs are kept exceptionally low, allowing for competitive pricing in the generic drug market. Additionally, the high atom economy of the click reaction ensures that the majority of the input mass is converted into the final product, minimizing waste disposal fees and maximizing the yield per batch.
- Enhanced Supply Chain Reliability: All key reagents, including sodium azide, cuprous iodide, and various terminal alkynes, are bulk commodities available from multiple global vendors, ensuring that the supply chain is resilient against single-source disruptions. The robustness of the reaction to minor variations in reagent quality means that procurement teams have greater flexibility in sourcing materials without compromising the final product specification. This flexibility is critical for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The use of PEG-400 aligns perfectly with green chemistry principles, as it is non-volatile and can potentially be recycled, reducing the environmental footprint of the manufacturing site. This eco-friendly profile simplifies the regulatory approval process for new drug filings, as solvent residue limits are less stringent compared to Class 2 or Class 1 solvents. The scalability of the process is further supported by the exothermic nature of the cycloaddition being manageable under the described conditions, allowing for safe translation from gram-scale laboratory synthesis to multi-kilogram commercial production without significant engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent's background and beneficial effects sections. These insights are intended to clarify the operational advantages and technical nuances for stakeholders evaluating this technology for integration into their existing manufacturing portfolios. Understanding these details is essential for making informed decisions about process adoption and resource allocation.
Q: What are the advantages of using PEG-400 in this triazole synthesis?
A: PEG-400 serves as a green, non-toxic, and recyclable solvent that simplifies the workup process and eliminates the need for volatile organic compounds, aligning with modern environmental compliance standards.
Q: Why is the CuI catalyst preferred over other copper sources for this reaction?
A: CuI is highly effective for promoting the regioselective formation of 1,4-disubstituted triazoles under mild conditions, ensuring high purity and minimizing side reactions compared to harsher catalytic systems.
Q: Can this one-pot method be scaled for industrial production?
A: Yes, the elimination of intermediate isolation steps and the use of stable, commercially available reagents make this process highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Deoxy-Beta-D-Glucopyranosyl Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative academic research into robust industrial processes that meet the highest standards of quality and efficiency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless and risk-mitigated. We are committed to delivering high-purity pharmaceutical intermediates with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch against the detailed impurity profiles defined in patents like CN102276673A.
We invite potential partners to engage with our technical procurement team to discuss how this green synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this one-pot methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is equipped with the most advanced and cost-effective solutions for producing next-generation glycosyl triazole therapeutics.
