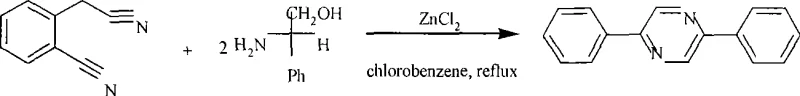
Advanced One-Step Synthesis of 2,5-Diphenylpyrazine for Commercial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes for high-value intermediates. A pivotal development in this domain is documented in patent CN101417983A, which discloses a novel method for the synthesis of 2,5-diphenylpyrazine, a structurally significant heterocyclic compound with potential applications in pharmaceutical and agrochemical sectors. This patent represents a significant departure from traditional multi-step methodologies by introducing a streamlined, one-pot condensation and cyclization strategy. The core innovation lies in the utilization of anhydrous zinc chloride as a Lewis acid catalyst within a chlorobenzene solvent system, facilitating the direct transformation of 2-cyanophenylacetonitrile and L-leucinol into the target pyrazine derivative. For R&D directors and process chemists, this approach offers a compelling alternative to legacy syntheses that often suffer from low atom economy and cumbersome purification steps. The ability to generate the target scaffold in a single operational step not only simplifies the workflow but also inherently reduces the accumulation of intermediate impurities, thereby enhancing the overall purity profile of the final product. This technical breakthrough underscores the potential for optimizing supply chains for complex heterocyclic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of substituted pyrazine rings has been a challenging endeavor in organic synthesis, often necessitating harsh reaction conditions and multiple discrete stages that erode overall yield and increase production costs. Traditional routes frequently rely on the condensation of alpha-dicarbonyl compounds with diamines or the oxidative coupling of imines, processes that are sensitive to moisture and require precise stoichiometric control to avoid oligomerization or polymerization side reactions. Furthermore, many classical methods employ expensive transition metal catalysts or toxic reagents that pose significant challenges for waste management and regulatory compliance in modern GMP environments. The purification of intermediates in these multi-step sequences often demands extensive chromatographic separation or recrystallization, leading to substantial material loss and extended lead times. For procurement managers, these inefficiencies translate directly into higher raw material costs and unpredictable delivery schedules, as any bottleneck in an early synthetic step can cascade through the entire production timeline. Additionally, the use of volatile or hazardous solvents in conventional protocols increases the safety risks associated with scale-up, requiring specialized equipment and rigorous safety protocols that further inflate capital expenditure.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology outlined in patent CN101417983A introduces a remarkably direct and robust pathway that consolidates the synthesis into a single, efficient transformation. By leveraging the unique reactivity of 2-cyanophenylacetonitrile with L-leucinol in the presence of a catalytic amount of anhydrous ZnCl2, the process achieves the formation of the 2,5-diphenylpyrazine core without the need for isolating unstable intermediates. This one-step protocol significantly reduces the operational complexity, eliminating the need for intermediate workups and drying steps that typically consume valuable reactor time and labor resources. The choice of chlorobenzene as the reaction medium provides a high-boiling environment conducive to driving the equilibrium towards product formation while maintaining solubility of the organic substrates throughout the extended reflux period. From a commercial perspective, this simplification of the synthetic tree offers profound advantages in terms of throughput and resource utilization, allowing manufacturers to produce larger batches with fewer unit operations. The robustness of the zinc chloride catalyst, which is inexpensive and readily available compared to precious metal alternatives, further enhances the economic viability of this route for large-scale industrial applications.
Mechanistic Insights into ZnCl2-Catalyzed Cyclization
The efficacy of this synthetic route is fundamentally rooted in the Lewis acidic properties of anhydrous zinc chloride, which plays a critical role in activating the nitrile functionalities of the starting material towards nucleophilic attack. In the proposed mechanistic pathway, the zinc center coordinates with the nitrogen atoms of the nitrile groups, increasing the electrophilicity of the carbon atoms and facilitating the initial condensation with the amino group of L-leucinol. This activation lowers the energy barrier for the formation of the amidine intermediate, which subsequently undergoes intramolecular cyclization and dehydration to establish the aromatic pyrazine ring system. The reaction conditions, specifically the reflux in chlorobenzene for 20 to 30 hours, are essential to provide the thermal energy required for the final aromatization step, ensuring the thermodynamic stability of the resulting heterocycle. Understanding this mechanism is crucial for process optimization, as it highlights the importance of maintaining strictly anhydrous conditions to prevent the hydrolysis of the nitrile groups or the deactivation of the Lewis acid catalyst by water coordination.
Furthermore, the control of impurity profiles in this reaction is intrinsically linked to the selectivity of the catalyst and the reaction parameters. The use of a specific molar ratio of reactants and the controlled addition of the catalyst help to minimize the formation of polymeric byproducts or incomplete condensation species that could complicate downstream purification. The subsequent workup procedure, involving extraction with chloroform and purification via silica gel chromatography using a petroleum ether and dichloromethane mixture, is designed to effectively separate the target pyrazine from any unreacted starting materials or minor side products. For quality control teams, this predictable impurity profile simplifies the validation of analytical methods and ensures consistent batch-to-batch reproducibility. The mechanistic clarity provided by this patent allows chemists to fine-tune reaction variables such as temperature and catalyst loading to maximize yield while maintaining high purity standards, which is a critical requirement for supplying intermediates to the regulated pharmaceutical industry.
How to Synthesize 2,5-Diphenylpyrazine Efficiently
To implement this advanced synthesis in a laboratory or pilot plant setting, operators must adhere to strict protocols regarding atmospheric control and reagent quality to ensure optimal outcomes. The process begins with the preparation of the reaction vessel under an inert atmosphere, followed by the precise charging of anhydrous zinc chloride, chlorobenzene solvent, and the organic substrates in the specified ratios. Detailed standard operating procedures for the heating profile, reflux duration, and quenching steps are essential to replicate the success reported in the patent literature and to ensure safety during scale-up operations. The following guide outlines the critical phases of this synthesis, providing a structured framework for technical teams to adopt this efficient methodology.
- Combine 2-cyanophenylacetonitrile and L-leucinol in chlorobenzene solvent with anhydrous ZnCl2 catalyst under inert atmosphere.
- Reflux the mixture for 20 to 30 hours to ensure complete cyclization and aromatization of the pyrazine ring.
- Perform workup by removing solvent, extracting with chloroform, and purifying via silica gel chromatography using petroleum ether and dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route presents a strategic opportunity to optimize costs and enhance the reliability of the supply base for key heterocyclic intermediates. The elimination of multiple synthetic steps and the use of commodity-grade reagents significantly reduce the direct material costs associated with production, while the simplified workflow decreases labor overhead and utility consumption. By transitioning to a one-step process, manufacturers can achieve faster turnaround times from order to delivery, mitigating the risks associated with long lead times that often plague complex multi-step syntheses. This agility is particularly valuable in dynamic market environments where demand fluctuations require responsive supply chains capable of scaling production up or down without excessive penalty. Moreover, the reduced complexity of the process lowers the barrier to entry for secondary suppliers, fostering a more competitive sourcing landscape that drives down prices and improves service levels for end-users.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the drastic simplification of the synthetic sequence, which eliminates the need for expensive isolation and purification of intermediates that characterize traditional multi-step routes. By consolidating the reaction into a single pot, the process minimizes solvent usage, reduces waste generation, and lowers the energy intensity per kilogram of product produced. The substitution of precious metal catalysts with inexpensive zinc chloride further contributes to substantial cost savings, making the final product more price-competitive in the global marketplace. Additionally, the high atom economy of the condensation reaction ensures that a greater proportion of the raw material mass is incorporated into the final product, reducing the effective cost of goods sold and improving overall margin potential for manufacturers.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable starting materials such as 2-cyanophenylacetonitrile and L-leucinol ensures a robust supply chain that is less susceptible to disruptions caused by the scarcity of exotic reagents. The simplicity of the reaction conditions, which do not require extreme pressures or cryogenic temperatures, allows for production in a broader range of facilities, thereby diversifying the geographic sources of supply and reducing logistical risks. This resilience is critical for pharmaceutical companies seeking to secure long-term supplies of key intermediates against the backdrop of global geopolitical instability and trade volatility. Furthermore, the predictable nature of the reaction kinetics facilitates accurate production planning and inventory management, enabling suppliers to maintain optimal stock levels and meet just-in-time delivery requirements with greater confidence.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process offers significant advantages due to its reduced solvent footprint and the absence of heavy metal contaminants that are difficult to remove from the final product. The use of chlorobenzene, while requiring careful handling, is a well-understood solvent in industrial chemistry with established recovery and recycling protocols that align with green chemistry principles. The simplified waste stream, resulting from fewer unit operations and higher selectivity, eases the burden on wastewater treatment facilities and reduces the costs associated with hazardous waste disposal. As regulatory scrutiny on chemical manufacturing intensifies globally, adopting cleaner and more efficient technologies like this ZnCl2-catalyzed route positions companies favorably for future compliance audits and sustainability certifications, enhancing their corporate reputation and market access.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis method for 2,5-diphenylpyrazine. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making by technical and commercial stakeholders. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production portfolios.
Q: What are the critical reaction conditions for this synthesis?
A: The reaction requires strictly anhydrous and oxygen-free conditions using chlorobenzene as the solvent, with reflux temperatures maintained for 20 to 30 hours to drive the cyclization to completion.
Q: How does the ZnCl2 catalyst improve the process efficiency?
A: Anhydrous Zinc Chloride acts as a potent Lewis acid, activating the nitrile groups for nucleophilic attack by the amino alcohol, thereby enabling a direct one-step formation of the pyrazine core without complex multi-stage sequences.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the use of common solvents like chlorobenzene and robust inorganic catalysts suggests high scalability, although the extended reflux time requires energy optimization strategies for massive tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Diphenylpyrazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the ZnCl2-catalyzed route described in patent CN101417983A for delivering high-quality chemical intermediates to the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent supply regardless of their volume requirements. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 2,5-diphenylpyrazine meets the exacting standards required for pharmaceutical and fine chemical applications. We are committed to leveraging our technical expertise to optimize this process further, driving down costs and improving efficiency for our partners.
We invite forward-thinking organizations to collaborate with us to unlock the full commercial value of this innovative synthesis. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs and quality targets. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments that demonstrate how our capabilities can streamline your supply chain and accelerate your product development timelines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
