Advanced Synthetic Route for Osthole Derivatives: Enhancing Commercial Viability and Purity
Advanced Synthetic Route for Osthole Derivatives: Enhancing Commercial Viability and Purity
The pharmaceutical and agrochemical industries have long recognized the potent biological activities of osthole derivatives, ranging from antitumor and anti-osteoporosis effects to significant antiviral properties. However, the commercial realization of these benefits has historically been hindered by inefficient synthetic pathways that suffer from low yields and harsh reaction conditions. Patent CN101456852B introduces a transformative two-step methodology that leverages a palladium-catalyzed Heck reaction followed by a chromium-mediated olefin conversion. This innovative approach not only streamlines the construction of the critical carbon-carbon bonds found in the coumarin scaffold but also operates under remarkably mild conditions, specifically utilizing room temperature for the final conversion step. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 7-hydroxyl-8-isopentene group coumarins was plagued by significant inefficiencies that rendered large-scale production economically unviable. Early methodologies, such as the one reported in 1985, relied on complex multi-step sequences involving iodine and chloro-ethyl acetylene, which introduced unnecessary complexity and impurity profiles. Another prominent method from 1986 utilized a slurry of alumina and required an staggering reaction time of five days to achieve a meager yield of merely 29%, creating a massive bottleneck for supply chain continuity. Furthermore, radical-based approaches developed in 2004 utilizing AIBN and THB still struggled to push yields beyond 40%, while generating substantial chemical waste. These legacy processes are characterized by their inability to provide consistent quality, their excessive energy consumption due to prolonged heating, and their reliance on difficult-to-handle reagents that complicate safety protocols in a manufacturing environment.
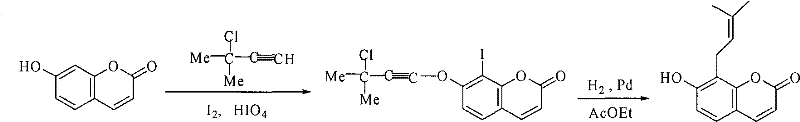
The Novel Approach
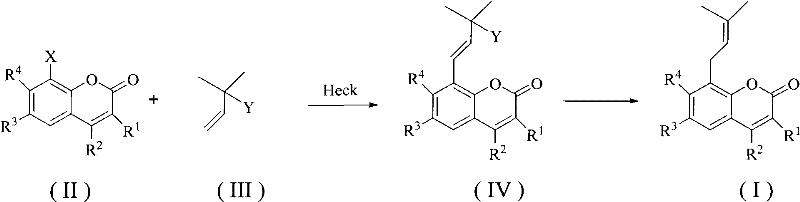
In stark contrast to these archaic techniques, the novel pathway disclosed in the patent utilizes a robust Heck coupling strategy to install the prenyl side chain directly onto the halogenated coumarin core. This method dramatically reduces the number of unit operations and allows for precise control over the regioselectivity of the substitution. By employing palladium acetate with triphenylphosphine ligands, the reaction achieves superior conversion rates in a fraction of the time required by previous methods, typically completing within 1 to 48 hours depending on the specific substrate. The subsequent transformation of the intermediate is equally impressive, utilizing a chromium trichloride and lithium aluminum hydride system that proceeds efficiently at room temperature. This eliminates the thermal stress on the sensitive coumarin lactone ring, thereby preserving the structural integrity of the molecule and minimizing the formation of degradation byproducts that are common in high-temperature processes.
Mechanistic Insights into Pd-Catalyzed Heck Coupling and Cr-Mediated Reduction
The core of this synthetic breakthrough lies in the mechanistic elegance of the palladium-catalyzed Heck reaction, which facilitates the formation of the C-C bond between the aryl halide and the olefin. In this catalytic cycle, the palladium species undergoes oxidative addition into the carbon-halogen bond of the coumarin substrate, forming a reactive organopalladium intermediate. This is followed by the coordination and migratory insertion of the olefin, such as 3-bromo-3-methyl-butylene, into the palladium-carbon bond. The cycle is completed by beta-hydride elimination, which regenerates the active palladium catalyst and releases the coupled product. The presence of triphenylphosphine as a ligand is crucial here, as it stabilizes the palladium center and prevents the formation of inactive palladium black, ensuring that the catalyst loading can be kept relatively low while maintaining high turnover numbers. This mechanistic efficiency is what allows the reaction to tolerate various functional groups, including nitro and amino substituents, without compromising the yield.
Following the coupling, the second step involves a sophisticated olefin conversion mediated by chromium trichloride and lithium aluminum hydride. While the exact mechanistic details of this reduction-isomerization sequence are complex, it is believed to involve the generation of low-valent chromium species that facilitate the rearrangement of the double bond or the reduction of specific functionalities depending on the desired final structure. The use of a mixed solvent system comprising DMF and THF, along with water or alcohol, creates a unique polarity environment that solubilizes both the inorganic chromium salts and the organic intermediate. This biphasic-like behavior enhances the interaction between the catalyst and the substrate, driving the reaction to completion at room temperature. From an impurity control perspective, this mildness is paramount; it prevents the polymerization of the olefin side chain and avoids the hydrolysis of the lactone ring, which are common failure modes in harsher acidic or basic conditions.
How to Synthesize Osthole Derivatives Efficiently
The execution of this synthesis requires careful attention to stoichiometry and solvent quality to maximize the efficiency of the palladium cycle and the subsequent chromium reduction. Operators must ensure that the halogenated coumarin and the olefin are mixed in the correct molar ratios, typically ranging from 1:1 to 1:5, to drive the equilibrium towards the product. The reaction environment must be strictly controlled to exclude oxygen, which can oxidize the phosphine ligand and deactivate the palladium catalyst. Following the Heck coupling, the workup involves standard extraction and washing procedures to remove inorganic salts before the crude intermediate is subjected to the second step. The detailed standardized operating procedures, including specific temperature ramps and quenching protocols for the lithium aluminum hydride, are critical for ensuring operator safety and product consistency.
- Perform a Heck reaction between halogenated coumarin and an olefin using palladium acetate and triphenylphosphine in a polar aprotic solvent at elevated temperatures.
- Isolate the intermediate osthole product through extraction and column chromatography purification.
- Conduct an olefin conversion reaction on the intermediate using chromium trichloride and lithium aluminum hydride in a DMF-THF mixture at room temperature.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic advantages that extend far beyond simple yield improvements. The primary benefit is the drastic simplification of the manufacturing process, which directly translates to reduced operational expenditures. By condensing what was previously a multi-day ordeal into a streamlined two-step sequence, facilities can significantly increase their reactor turnover rates, effectively expanding production capacity without the need for capital-intensive infrastructure upgrades. The use of commodity chemicals like palladium acetate and chromium trichloride, rather than exotic or proprietary reagents, ensures a stable and competitive supply chain for raw materials, mitigating the risk of price volatility or sourcing bottlenecks that often plague specialty chemical manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of energy-intensive heating steps and the reduction in reaction time. Traditional methods requiring five days of reflux consume vast amounts of utility resources, whereas this novel method operates largely at moderate temperatures or even room temperature for the final step. Additionally, the higher yields mean that less raw material is wasted per kilogram of finished product, directly lowering the cost of goods sold. The ability to use cheaper halogenated starting materials instead of complex pre-functionalized precursors further drives down the input costs, making the final osthole derivatives much more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The tolerance of the Heck reaction to various substituents means that the same platform technology can be used to synthesize a library of different derivatives simply by swapping the starting coumarin. This flexibility allows manufacturers to respond rapidly to changing market demands for specific analogues without revalidating entirely new processes. Furthermore, the stability of the reagents and the simplicity of the workup procedures reduce the likelihood of batch failures, ensuring a consistent and reliable flow of material to downstream customers who depend on just-in-time delivery for their own formulation schedules.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of extreme pressures or temperatures. The room temperature operation of the second step is particularly advantageous for safety, as it minimizes the risk of thermal runaway events in large reactors. From an environmental standpoint, the improved atom economy and higher yields result in less chemical waste per unit of product, simplifying effluent treatment and helping facilities meet increasingly stringent environmental regulations. The use of standard solvents like DMSO and THF, which are readily recyclable, further enhances the green chemistry profile of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing production lines or for procurement teams negotiating supply agreements based on these specifications.
Q: What are the primary advantages of this Heck-based synthesis over traditional methods?
A: This method significantly improves reaction yields compared to prior art which often suffered from yields below 40%. It utilizes milder conditions, particularly in the second step which proceeds at room temperature, reducing energy consumption and operational hazards associated with high-temperature reflux.
Q: Is the chromium trichloride catalyst system scalable for industrial production?
A: Yes, the use of chromium trichloride and lithium aluminum hydride in a DMF-THF solvent system is highly amenable to scale-up. The reaction operates at room temperature, eliminating the need for complex heating infrastructure and allowing for safer handling of exothermic potentials in large reactors.
Q: How does this route impact the cost of goods for osthole derivatives?
A: The route utilizes cheap and easily obtainable raw materials such as halogenated coumarins and simple olefins. Furthermore, the elimination of prolonged reaction times (reducing from 5 days in older methods to under 48 hours total) drastically lowers utility costs and increases reactor throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Osthole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a robust commercial process requires deep technical expertise and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the Heck-coupling route are fully realized in a GMP-compliant environment. We understand that high-purity osthole derivatives are critical for downstream drug development, which is why our stringent purity specifications and rigorous QC labs are designed to detect and eliminate even trace levels of palladium or chromium residues, guaranteeing a product that meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to optimize your supply chain for these valuable bioactive compounds. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this efficient synthetic route can lower your overall procurement costs. We encourage potential partners to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than estimates. Let us help you secure a stable, high-quality supply of osthole derivatives that empowers your research and commercial success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
