Advanced Cyclization Technology for High-Purity Xanthotoxol and Derivatives Manufacturing
Introduction to Novel Xanthotoxol Synthesis Technology
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to produce bioactive molecules, particularly those with significant therapeutic potential like psoralens. A groundbreaking patent, CN102134247A, discloses a novel synthetic methodology for Xanthotoxol and its derivatives, addressing critical bottlenecks in traditional manufacturing. This technology introduces a highly efficient cyclization strategy that constructs the core furocoumarin skeleton in a single step, marking a substantial departure from the tediously long multi-step sequences previously reported in literature. For R&D directors and procurement specialists, this represents a pivotal opportunity to optimize the supply chain for dermatological and anti-inflammatory agents.
The significance of this innovation lies in its ability to bypass the limitations of natural extraction, which is often plagued by low yields and resource scarcity. By shifting to a robust chemical synthesis, manufacturers can achieve consistent quality and scalability. The patent outlines a versatile approach where various substituted benzofuranones react with beta-keto esters or acids under acidic conditions. This not only simplifies the process but also opens the door to a wide array of derivatives by merely adjusting the substituents on the starting materials. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic advancements is crucial for evaluating the feasibility of large-scale production.
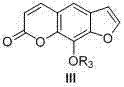
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Xanthotoxol and related compounds like Xanthotoxin has relied heavily on extraction from natural plant sources such as Fructus Cnidii or Ammi majus. While natural, this approach is inherently unstable; crop yields fluctuate due to environmental factors, leading to volatile pricing and supply interruptions. Furthermore, the isolation process is labor-intensive and results in low recovery rates, making it economically unviable for meeting the demands of the global market. On the synthetic front, prior art methods, such as those reported by Carl or Wang, often involve convoluted pathways exceeding nine reaction steps. These lengthy syntheses suffer from cumulative yield losses, where the overall yield rarely exceeds 9%, rendering them impractical for industrial application.
Additionally, traditional synthetic routes often employ harsh reagents or require complex purification steps to remove impurities generated at each stage. The accumulation of by-products necessitates extensive chromatographic separations, which drives up both the cost of goods sold (COGS) and the environmental footprint of the manufacturing process. For a procurement manager, these inefficiencies translate into higher raw material costs and longer lead times. The inability to scale these archaic methods without compromising purity or safety has long been a barrier to the widespread availability of high-quality psoralen intermediates for drug development.
The Novel Approach
The methodology presented in CN102134247A revolutionizes this landscape by introducing a convergent synthesis strategy centered on a key cyclization reaction. Instead of building the molecule piece by piece over many steps, this approach utilizes readily available benzofuran-3(2H)-one derivatives as building blocks. These precursors undergo a direct condensation with beta-keto esters or acids, such as 3,3-diethoxypropionic acid or 3-oxopropionic acid, to form the target hexa-atomic unsaturated cyclic lactone ring in a single operation. This drastic reduction in step count—from nine steps down to just four when starting from pyrogallol—results in a dramatic improvement in overall yield, reaching up to 27%.
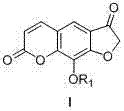
This novel route is not only shorter but also chemically elegant. It employs a robust acid-catalyzed system using trifluoroacetic acid and sulfuric acid, which promotes the cyclization efficiently at moderate temperatures (around 120°C). The simplicity of the operation means that reaction control is easier, and the formation of side products is minimized. For supply chain heads, this translates to a process that is inherently safer and easier to manage in a multi-ton reactor setting. The versatility of the method allows for the easy introduction of different alkoxy groups, enabling the production of a library of derivatives like Xanthotoxin and 8-ethoxypsoralene without redesigning the entire synthetic pathway.
Mechanistic Insights into Acid-Catalyzed Cyclization
The core of this technological breakthrough is the acid-catalyzed cyclization mechanism, which resembles a Pechmann condensation but is adapted for the specific electronic requirements of the benzofuranone system. In this reaction, the carbonyl oxygen of the beta-keto ester is protonated by the strong acid catalyst, increasing its electrophilicity. This activated species then undergoes nucleophilic attack by the electron-rich aromatic ring of the benzofuranone precursor. The subsequent elimination of alcohol (from the ester) or water (from the acid) drives the formation of the new carbon-carbon bond, closing the pyrone ring to complete the furocoumarin scaffold.
Understanding this mechanism is vital for R&D teams aiming to optimize the process further. The choice of acid catalyst is critical; the combination of trifluoroacetic acid and sulfuric acid provides the necessary acidity to drive the equilibrium towards the product while maintaining the stability of the sensitive furan ring. Moreover, the reaction conditions are tuned to prevent polymerization or degradation of the starting materials. By controlling the temperature at 120°C and the reaction time at 4 hours, the process ensures high conversion rates. This precise control over the reaction environment is what allows the method to achieve yields as high as 78% in specific derivative syntheses, a figure that is exceptionally high for heterocyclic chemistry.
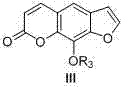
Impurity control is another aspect where this mechanism shines. Because the reaction is a direct cyclization, there are fewer intermediate isolation steps where impurities can be introduced or carried over. The major by-products are typically small molecules like ethanol or water, which are easily removed during the aqueous workup. The crude product can often be purified simply by recrystallization from ethanol, avoiding the need for expensive column chromatography. This streamlined purification process is a key factor in reducing the overall cost of manufacturing and ensuring that the final API intermediate meets stringent purity specifications required by regulatory bodies.
How to Synthesize Xanthotoxol Efficiently
To implement this synthesis in a laboratory or pilot plant setting, one must strictly adhere to the optimized protocols derived from the patent data. The process begins with the preparation of the key benzofuranone intermediate, which can be obtained via hydrogenation of the corresponding coumarin ketone using palladium on carbon. Once the precursor is secured, it is mixed with the appropriate beta-keto component in a solvent system containing trifluoroacetic acid and sulfuric acid. The mixture is then heated to reflux conditions to effect the cyclization. Detailed standardized operating procedures regarding stoichiometry, addition rates, and quenching protocols are essential for reproducibility.
- Prepare the benzofuran-3(2H)-one precursor (Formula IV, VII, or VIII) via hydrogenation or reduction of corresponding coumarin or benzofuranone ketones.
- Mix the precursor with a beta-keto ester or acid (Formula V or VI) in the presence of trifluoroacetic acid and sulfuric acid.
- Heat the reaction mixture to 120°C for approximately 4 hours to effect ring-closure, followed by aqueous workup and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling economic and logistical benefits. The primary advantage is the significant reduction in manufacturing costs driven by the shortened synthetic sequence. By eliminating five or more reaction steps compared to traditional methods, the consumption of solvents, reagents, and energy is drastically lowered. Furthermore, the high overall yield means that less raw material is required to produce the same amount of final product, directly improving the margin profile. This efficiency makes the production of high-purity pharmaceutical intermediates much more cost-competitive in the global market.
- Cost Reduction in Manufacturing: The elimination of multiple isolation and purification steps significantly lowers the operational expenditure. Traditional methods requiring chromatography are replaced by simple crystallization, which reduces solvent waste and processing time. The use of commodity chemicals like sulfuric acid and ethanol as solvents further keeps raw material costs low. Additionally, the high atom economy of the cyclization step minimizes waste generation, contributing to lower disposal costs and a smaller environmental footprint.
- Enhanced Supply Chain Reliability: Unlike plant extraction, which is subject to seasonal variations and agricultural risks, this chemical synthesis relies on stable, petrochemical-derived feedstocks. This ensures a continuous and predictable supply of Xanthotoxol and its derivatives, mitigating the risk of stockouts that can halt downstream drug production. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in operating parameters, ensuring consistent batch-to-batch quality.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind. The reaction uses standard equipment and does not require exotic catalysts or extreme pressures, making it easy to transfer from the lab to multi-ton reactors. The simplified workup procedure reduces the volume of hazardous waste, facilitating compliance with increasingly strict environmental regulations. This scalability ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Xanthotoxol derivatives using this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this manufacturing route.
Q: What are the primary advantages of the new synthetic route for Xanthotoxol compared to traditional extraction?
A: The novel synthetic route described in CN102134247A significantly reduces the number of reaction steps from nine down to four when starting from pyrogallol, increasing the overall yield from less than 9% to approximately 27%. Unlike plant extraction, which is limited by natural resources, this chemical synthesis ensures a stable and scalable supply chain.
Q: What specific reaction conditions are required for the key cyclization step?
A: The critical ring-closure reaction utilizes a strong acid catalytic system comprising trifluoroacetic acid and sulfuric acid. The reaction is typically conducted at elevated temperatures around 120°C for 4 hours, facilitating the formation of the hexa-atomic unsaturated cyclic lactone structure in a single step.
Q: Can this method be adapted for producing methyl or ethyl ether derivatives?
A: Yes, the process is highly versatile. By substituting the hydroxyl groups on the benzofuranone precursor with alkoxy groups (such as methoxy or ethoxy), the same cyclization protocol successfully produces derivatives like Xanthotoxin (methyl ether) and 8-ethoxypsoralene with high efficiency and purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Xanthotoxol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic routes disclosed in CN102134247A for the production of psoralen-based therapeutics. As a leading CDMO partner, we possess the technical expertise to adapt and optimize these cyclization methodologies for your specific project needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to factory floor is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Xanthotoxol or derivative meets the highest international standards.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective supply of these critical intermediates for your next-generation pharmaceutical products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
