Advanced Rhodium-Catalyzed Synthesis of Nitrogen-Oxygen Fused Aromatics for High-Performance Optoelectronics
Introduction to Patent CN114716454A and Technological Breakthroughs
The landscape of organic optoelectronic materials is undergoing a significant transformation driven by the demand for higher efficiency and stability in devices such as OLEDs and organic solar cells. Patent CN114716454A introduces a groundbreaking synthetic methodology for constructing nitrogen and oxygen heterocyclic aromatic hydrocarbons, a class of compounds pivotal for next-generation display and semiconductor technologies. This invention addresses critical bottlenecks in existing synthesis routes, specifically the reliance on complex substrates and limited structural diversity found in traditional polycyclic aromatic hydrocarbon production. By leveraging a rhodium-catalyzed C-H activation strategy, the patent outlines a pathway that is not only atom-economical but also operationally simple, utilizing mild reaction conditions that range from 80°C to 120°C. For R&D directors and procurement specialists in the fine chemical sector, this represents a substantial opportunity to secure reliable organic semiconductor intermediate suppliers who can deliver high-purity materials with improved process scalability. The ability to synthesize these complex fused-ring systems in fewer steps directly correlates to reduced manufacturing costs and enhanced supply chain resilience, making this technology a cornerstone for future industrial applications in luminescent materials and pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fused heterocyclic aromatic hydrocarbons has been dominated by methods utilizing strongly coordinating directing groups, such as pyridine derivatives, to guide transition metal-catalyzed C-H activation. While effective in specific contexts, these conventional approaches suffer from significant drawbacks that hinder large-scale commercial adoption. The synthesis of pyridine-containing substrates is often multi-step, expensive, and generates substantial chemical waste, thereby increasing the overall cost of goods sold. Furthermore, the strong coordination of pyridine can sometimes lead to catalyst poisoning or require harsher reaction conditions to drive the transformation, limiting the tolerance for sensitive functional groups. This rigidity restricts the structural diversity of the final products, confining manufacturers to a narrow range of nitrogen-only fused skeletons. For supply chain heads, this translates to vulnerability; reliance on complex, proprietary substrates creates single points of failure and extends lead times for high-purity organic electronic chemicals. The inability to easily modify the core structure without redesigning the entire synthetic route stifles innovation in material properties, such as bandgap tuning for specific OLED emission colors.
The Novel Approach
In stark contrast, the methodology described in CN114716454A utilizes benzoxazine imines as weakly coordinating directing groups, a strategic shift that unlocks unprecedented versatility in molecular design. This novel approach allows for the direct functionalization of inert C-H bonds using readily available diazo compounds, facilitating a [4+2] tandem annulation strategy that constructs the nitrogen-oxygen fused core in a single operational step. The use of a Rh(III) catalyst system, typically comprising [Cp*RhCl2]2 and a silver salt like AgSbF6, enables the reaction to proceed under mild thermal conditions with excellent functional group tolerance. This means that electron-withdrawing or electron-donating substituents such as halogens, alkyl groups, or alkoxy groups can be incorporated without compromising yield. 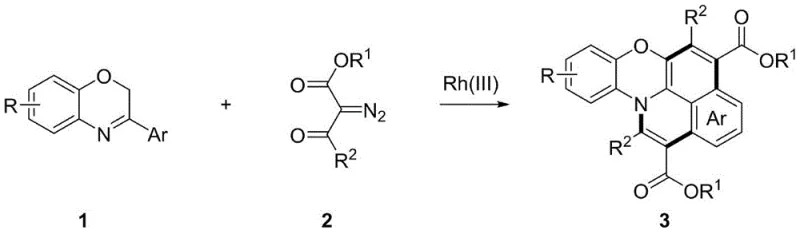 As illustrated in the general reaction scheme, the process transforms simple oxazine precursors into complex isoquinoline-phenoxazine derivatives with high atom economy. For procurement managers, this simplicity translates to cost reduction in organic electronic chemical manufacturing, as the starting materials are commodity chemicals rather than bespoke synthons. The robustness of this method ensures consistent quality and supply continuity, essential for maintaining production schedules in the fast-paced consumer electronics market.
As illustrated in the general reaction scheme, the process transforms simple oxazine precursors into complex isoquinoline-phenoxazine derivatives with high atom economy. For procurement managers, this simplicity translates to cost reduction in organic electronic chemical manufacturing, as the starting materials are commodity chemicals rather than bespoke synthons. The robustness of this method ensures consistent quality and supply continuity, essential for maintaining production schedules in the fast-paced consumer electronics market.
Mechanistic Insights into Rh(III)-Catalyzed C-H Activation and Annulation
The success of this synthetic route lies in the intricate catalytic cycle mediated by the cationic Rh(III) species generated in situ. The mechanism initiates with the coordination of the benzoxazine imine nitrogen to the rhodium center, followed by a concerted metalation-deprotonation (CMD) process that activates the proximal C-H bond to form a stable five-membered rhodacycle intermediate. This step is crucial as it determines the regioselectivity of the subsequent functionalization. Upon introduction of the diazo compound, the rhodium-carbene species is formed via nitrogen extrusion, which then undergoes a migratory insertion into the Rh-C bond. This insertion expands the ring system and sets the stage for the second C-H activation event. The catalytic cycle continues with a second equivalent of diazo compound reacting with the intermediate, leading to a double alkylation sequence. Subsequent tautomerization to an enol form triggers an intramolecular nucleophilic attack by the hydroxyl group on the imine bond, followed by a cascade of acidification, ring-opening, and dehydration steps. 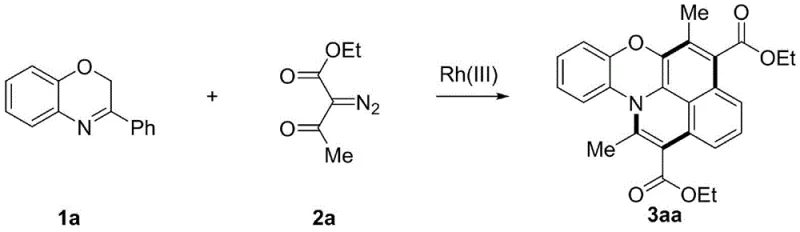 This complex cascade, detailed in the specific example of converting substrate 1a to product 3aa, ultimately furnishes the target nitrogen-oxygen heterocyclic aromatic hydrocarbon. Understanding this mechanism is vital for R&D teams aiming to optimize impurity profiles; controlling the stoichiometry of the diazo compound and the acidity of the additive (such as 2,4,6-trimethylbenzoic acid) is key to minimizing side reactions and maximizing the yield of the desired fused core.
This complex cascade, detailed in the specific example of converting substrate 1a to product 3aa, ultimately furnishes the target nitrogen-oxygen heterocyclic aromatic hydrocarbon. Understanding this mechanism is vital for R&D teams aiming to optimize impurity profiles; controlling the stoichiometry of the diazo compound and the acidity of the additive (such as 2,4,6-trimethylbenzoic acid) is key to minimizing side reactions and maximizing the yield of the desired fused core.
From an impurity control perspective, the mild nature of the Rh(III) catalyst system significantly reduces the formation of decomposition byproducts often seen with more aggressive Lewis acids or high-temperature thermal cyclizations. The specific choice of silver salt, such as AgSbF6, plays a dual role in generating the active cationic catalyst and scavenging chloride ions, which prevents catalyst deactivation. The reaction's tolerance for various solvents, including dichloroethane (DCE) and toluene, offers flexibility in process engineering to optimize solubility and heat transfer during scale-up. For quality assurance teams, the predictable formation of the isoquinoline[2,1,8-mna]phenoxazine skeleton ensures a clean impurity spectrum, simplifying downstream purification via standard silica gel chromatography. This mechanistic clarity provides a solid foundation for developing robust analytical methods, such as HPLC and NMR, to monitor reaction progress and certify the high purity required for electronic grade materials.
How to Synthesize Isoquinoline Phenoxazine Derivatives Efficiently
Implementing this synthesis in a pilot or production environment requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the reaction mixture under an inert atmosphere, typically nitrogen or argon, to prevent oxidation of the sensitive diazo compounds and the rhodium catalyst. Precise molar ratios are essential, with the oxazine substrate, diazo compound, catalyst, and additive typically employed in a ratio of 1:(2-3):(0.02-0.04):(0.08-0.16). The reaction concentration is maintained between 0.05M and 0.2M to balance reaction rate with heat dissipation. Following the reaction period of 6 to 12 hours at 80-120°C, the workup involves solvent removal under reduced pressure, followed by purification. The detailed standardized synthesis steps for scaling this process are provided in the guide below.
- Prepare the reaction mixture by adding oxazine compound 1, diazo compound 2, Rh(III) catalyst, silver salt additive, and carboxylic acid additive into a solvent like DCE under an inert nitrogen atmosphere.
- Heat the sealed reaction vessel to a temperature between 80°C and 120°C and maintain stirring for a duration of 6 to 12 hours to ensure complete conversion.
- Upon completion, remove the solvent under reduced pressure and purify the crude residue using silica gel column chromatography with a petroleum ether/ethyl acetate/dichloromethane eluent system.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this Rh(III)-catalyzed synthesis offers profound strategic advantages for organizations managing the supply of advanced chemical intermediates. By shifting from complex pyridine-based substrates to simple benzoxazine imines, manufacturers can drastically simplify their raw material sourcing strategies. Benzoxazine derivatives are widely available and inexpensive compared to specialized heterocyclic amines, leading to substantial cost savings in the bill of materials. Furthermore, the high atom economy of the [4+2] annulation means less waste generation, which lowers disposal costs and aligns with increasingly stringent environmental regulations. For supply chain heads, the robustness of the reaction conditions—operating at moderate temperatures without the need for cryogenic cooling or ultra-high pressures—reduces energy consumption and equipment wear, enhancing the overall reliability of the production line. This process stability minimizes the risk of batch failures, ensuring a consistent flow of materials to downstream device manufacturers.
- Cost Reduction in Manufacturing: The elimination of multi-step substrate synthesis and the use of commodity-grade diazo compounds significantly lower the entry barrier for production. The high yields, often exceeding 80% and reaching up to 98% in optimized cases, mean that less raw material is required per kilogram of finished product. Additionally, the simplified purification process reduces solvent usage and processing time, contributing to a leaner manufacturing footprint and improved margin potential for high-value electronic chemicals.
- Enhanced Supply Chain Reliability: Sourcing risks are mitigated by the use of broadly available starting materials that are not subject to the same supply constraints as exotic directing groups. The reaction's tolerance for diverse functional groups allows for the production of a wide library of derivatives from a common platform, enabling rapid response to changing market demands for specific material properties. This flexibility ensures that procurement teams can maintain inventory levels of critical intermediates without relying on single-source suppliers for complex precursors.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic heavy metal contaminants in the final product (due to efficient catalyst removal protocols) facilitate easier regulatory approval for pharmaceutical and consumer electronic applications. The process is inherently scalable from gram to tonnage quantities without significant re-engineering, supporting the commercial scale-up of complex organic semiconductors. This scalability, combined with reduced waste generation, positions manufacturers to meet global sustainability goals while maintaining competitive pricing structures.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the synthesis and application of these nitrogen-oxygen fused aromatics. These insights are derived directly from the experimental data and technical specifications provided in patent CN114716454A, ensuring accuracy and relevance for industrial implementation. Understanding these nuances is critical for integrating this chemistry into existing R&D pipelines and supply chain frameworks effectively.
Q: What is the primary advantage of using benzoxazine imines over traditional pyridine directing groups?
A: Benzoxazine imines serve as weakly coordinating directing groups, which simplifies substrate synthesis compared to strongly coordinating pyridines. This approach allows for broader functional group tolerance and access to novel nitrogen-oxygen fused skeletons that are difficult to achieve with conventional methods.
Q: What are the typical yield ranges for this Rh(III)-catalyzed annulation process?
A: The patented method demonstrates robust efficiency, with isolated yields generally exceeding 80% across various substrates. In optimized cases, such as the synthesis of bromo-substituted derivatives, yields can reach as high as 98%, indicating high atomic economy.
Q: Which industries benefit most from these nitrogen-oxygen heterocyclic aromatic hydrocarbons?
A: These compounds are critical for the organic optoelectronics industry, specifically for Organic Light-Emitting Diodes (OLEDs), Organic Field-Effect Transistors (OFETs), and Organic Solar Cells (OSCs). They also hold significant potential as intermediates in pharmaceutical synthesis and natural product development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoquinoline Phenoxazine Derivatives Supplier
As the demand for high-performance organic optoelectronic materials continues to surge, partnering with a chemically proficient CDMO is essential for translating patented innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for OLED and semiconductor applications. We understand the critical nature of impurity control in electronic materials and employ advanced analytical techniques to guarantee the quality of every batch of nitrogen-oxygen heterocyclic aromatic hydrocarbons we produce.
We invite you to collaborate with our technical procurement team to explore how this Rh(III)-catalyzed technology can optimize your material costs and accelerate your product development cycles. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique project goals, ensuring a seamless transition from laboratory discovery to industrial manufacturing.
