Advanced Manufacturing of Penfluridol: A High-Yield Route for Global API Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic viability, particularly for long-acting antipsychotic agents like Penfluridol. Patent CN111196781A introduces a transformative methodology that addresses the longstanding inefficiencies in producing this diphenylbutylpiperidine compound. By shifting the synthetic entry point to 4,4-bis(4-fluorophenyl)butyric acid, the inventors have engineered a streamlined three-step sequence involving reduction, esterification, and condensation. This approach not only elevates the total yield to an impressive 81.3 percent but also drastically simplifies the downstream purification protocols. For R&D directors and procurement strategists, this patent represents a critical opportunity to optimize the cost structure of antipsychotic API manufacturing while ensuring a reliable supply of high-purity intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Penfluridol has been plagued by convoluted reaction sequences that begin with simple fluorobenzene derivatives. As illustrated in prior art such as CN106187863A, traditional routes necessitate a series of energetically demanding steps including side-chain reactions, Friedel-Crafts acylations, and multiple reduction phases to construct the requisite carbon skeleton. These legacy processes suffer from cumulative yield losses, often resulting in a dismal overall yield ranging between 40 percent and 50 percent. Furthermore, the final amide reduction steps in these older methodologies are notorious for generating complex impurity profiles, necessitating repetitive and costly refining operations to meet pharmacopeial standards. This inefficiency creates a bottleneck for commercial scale-up, inflating the cost of goods sold and introducing significant supply chain volatility for manufacturers relying on these outdated synthetic strategies.
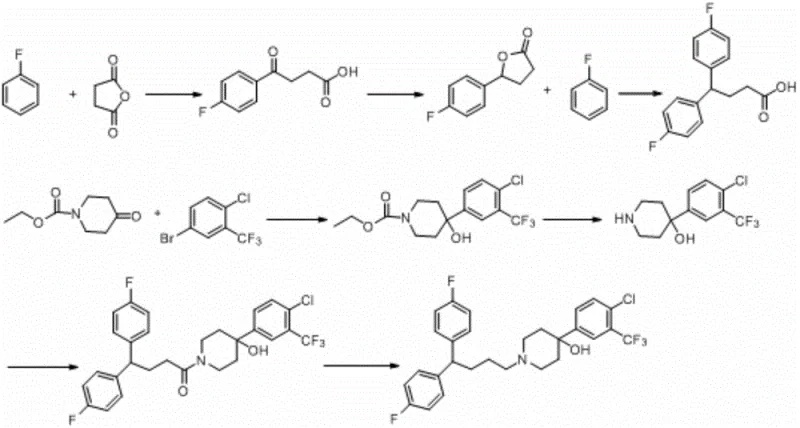
The Novel Approach
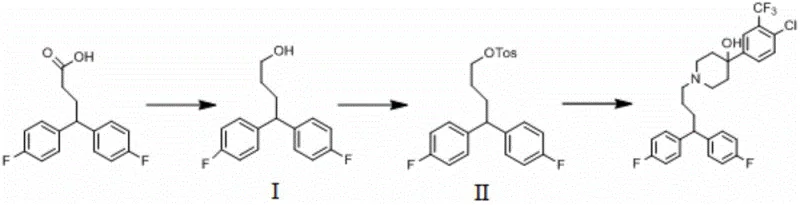
In stark contrast, the improved method disclosed in CN111196781A leverages a convergent strategy that utilizes 4,4-bis(4-fluorophenyl)butyric acid as a advanced building block. This strategic choice eliminates the need for constructing the diphenylbutyl framework from scratch, thereby removing several high-risk transformation steps. The new pathway proceeds through a logical progression: first reducing the carboxylic acid to the corresponding primary alcohol, then activating this alcohol via sulfonate ester formation, and finally coupling it with the piperidine moiety. This rational design results in a remarkably mild reaction profile with operationally simple workups. The visual representation of this streamlined process highlights how eliminating unnecessary synthetic detours directly correlates with enhanced process mass intensity and superior economic outcomes for industrial applications.
Mechanistic Insights into the Reduction and Activation Strategy
The cornerstone of this improved synthesis lies in the efficient conversion of the carboxylic acid starting material into a reactive electrophile suitable for nucleophilic substitution. The initial reduction step employs potent hydride sources such as lithium aluminum hydride or borane complexes to convert the carbonyl group of 4,4-bis(4-fluorophenyl)butyric acid into a primary hydroxyl group. This transformation is meticulously controlled within a temperature range of -30°C to 10°C to prevent over-reduction or side reactions, followed by a careful quenching protocol using aqueous alkaline solutions to manage exotherms and facilitate phase separation. The inclusion of decolorizing agents like potassium carbonate or neutral alumina during this stage is a critical quality control measure, ensuring that the resulting alcohol intermediate possesses the optical clarity and chemical purity necessary for subsequent high-fidelity transformations.
Following the reduction, the activation of the hydroxyl group is achieved through esterification with sulfonyl chlorides, such as p-toluenesulfonyl chloride or methanesulfonyl chloride. This step converts the poor leaving group (-OH) into an excellent leaving group (-OTs or -OMs), thereby priming the molecule for the final nucleophilic attack by the piperidine nitrogen. The reaction is conducted under inert atmosphere protection to prevent moisture interference, utilizing triethylamine as a base to scavenge the generated hydrogen chloride. This activation strategy is superior to direct alkylation methods because it offers precise control over reaction kinetics and minimizes the formation of elimination byproducts. The resulting sulfonate ester is sufficiently stable for isolation yet reactive enough to undergo smooth condensation with the piperidinol derivative in polar aprotic solvents like acetonitrile, ensuring high conversion rates and minimal impurity generation.
How to Synthesize Penfluridol Efficiently
The implementation of this patented technology requires strict adherence to the specified stoichiometric ratios and thermal profiles to maximize the 81.3 percent total yield. The process is designed to be scalable, utilizing common industrial solvents such as tetrahydrofuran, dichloromethane, and acetonitrile, which facilitates easy technology transfer from laboratory to pilot plant. Operators must pay particular attention to the temperature control during the hydride reduction and the pH adjustment during the workup phases to ensure safety and product integrity. The following guide outlines the critical operational parameters derived from the patent examples, serving as a foundational reference for process engineers aiming to replicate this high-efficiency route.
- Reduce 4,4-bis(4-fluorophenyl)butyric acid using lithium aluminum hydride or borane to form the corresponding butanol intermediate.
- Activate the alcohol intermediate via esterification with sulfonyl chloride (e.g., p-toluenesulfonyl chloride) to create a sulfonate ester.
- Condense the activated sulfonate ester with 4-[4-chloro-3-(trifluoromethyl)phenyl]-4-piperidinol in acetonitrile to yield Penfluridol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this improved synthesis route offers tangible strategic benefits that extend beyond simple yield metrics. By consolidating the synthetic pathway and eliminating low-yielding steps, the process inherently reduces the consumption of raw materials and solvents per kilogram of finished product. This reduction in material intensity directly lowers the variable costs associated with manufacturing, providing a competitive edge in pricing negotiations for bulk API contracts. Furthermore, the use of stable, commercially available starting materials like 4,4-bis(4-fluorophenyl)butyric acid mitigates the risk of supply disruptions often associated with custom-synthesized precursors, thereby enhancing the overall resilience of the supply chain against market fluctuations.
- Cost Reduction in Manufacturing: The dramatic improvement in total yield from approximately 45 percent in legacy methods to over 81 percent in this new process fundamentally alters the cost equation. This efficiency gain means that nearly double the amount of product can be generated from the same quantity of input materials, effectively halving the raw material cost burden per unit. Additionally, the simplified purification requirements reduce the consumption of energy and chromatography media, leading to substantial operational expenditure savings. The elimination of complex transition metal catalysts or harsh reagents further decreases waste disposal costs, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is paramount for maintaining regulatory compliance in the pharmaceutical sector. The reliance on standard reagents and solvents means that sourcing is not dependent on niche suppliers, reducing lead times and minimizing the risk of production stoppages due to material shortages. The mild reaction conditions also reduce equipment wear and tear, allowing for longer campaign runs and more predictable production schedules. This reliability makes the manufacturer a more attractive partner for long-term supply agreements with multinational pharmaceutical companies seeking stability in their API sourcing.
- Scalability and Environmental Compliance: From an environmental perspective, the shorter synthetic sequence generates less chemical waste, aligning with modern green chemistry principles and reducing the burden on effluent treatment facilities. The process avoids the use of highly toxic or persistent organic pollutants where possible, simplifying the permitting process for new manufacturing lines. The scalability is evidenced by the use of standard unit operations such as filtration, crystallization, and distillation, which are easily replicated in large-scale reactors. This ease of scale-up allows manufacturers to rapidly respond to increases in market demand without requiring significant capital investment in specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented Penfluridol synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this technology. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What is the primary advantage of the new Penfluridol synthesis method over prior art?
A: The new method described in CN111196781A achieves a total yield of 81.3%, significantly higher than the 40-50% yield of conventional methods, while simplifying purification and reducing impurity profiles.
Q: Which starting material is utilized to improve process efficiency?
A: The process utilizes 4,4-bis(4-fluorophenyl)butyric acid as the initial raw material, bypassing the complex multi-step construction of the diphenylbutyl skeleton required in older fluorobenzene-based routes.
Q: How does this process impact supply chain reliability for antipsychotic APIs?
A: By employing mild reaction conditions and commercially available reagents like lithium aluminum hydride and sulfonyl chlorides, the process ensures robust scalability and consistent batch-to-batch quality, critical for long-term API supply.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Penfluridol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in maintaining a competitive edge in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN111196781A are fully realized in practical manufacturing settings. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to process optimization allows us to offer clients a secure and cost-effective supply of complex organic molecules.
We invite you to collaborate with us to leverage this advanced synthesis technology for your Penfluridol requirements. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can drive value and efficiency in your supply chain.
