Advanced Chiral Reduction for Bimatoprost Intermediates: A Commercial Scale-Up Perspective
The global demand for effective glaucoma treatments continues to drive innovation in the synthesis of prostaglandin analogues, with bimatoprost standing as a cornerstone therapy for lowering intraocular pressure. Patent CN109232494B, published in 2020, introduces a transformative preparation method for the key chiral intermediate required for bimatoprost production, addressing long-standing challenges in stereoselectivity and process efficiency. This technical breakthrough utilizes a specialized aluminum-hydride complex, (S)-BINAL-H, to achieve asymmetric reduction with unprecedented precision, offering a viable pathway for manufacturers seeking to optimize their ophthalmic API supply chains. The ability to produce the intermediate with a chiral purity ratio exceeding 99.5:0.5 represents a significant leap forward from conventional methodologies, directly impacting the cost structure and scalability of the final drug substance. For R&D directors and procurement strategists, understanding the mechanistic advantages of this patent is crucial for evaluating potential technology transfers or licensing opportunities that can secure a competitive edge in the highly regulated pharmaceutical market. 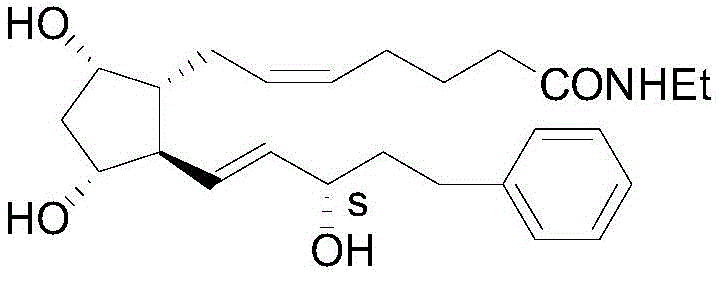
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the bimatoprost key intermediate has relied on asymmetric reduction agents such as (-)-diisopinocampheylchloroborane ((-)-DIP-chloride) or R-CBS catalysts, which, while effective, present substantial limitations for large-scale commercial manufacturing. These traditional reagents typically yield a mixture of isomers with a ratio of approximately 95:5 or 93:7 between the desired II-3S isomer and the unwanted II-3R impurity. This level of impurity is unacceptable for final pharmaceutical grade material, necessitating rigorous and expensive downstream purification processes such as complicated derivative crystallization or preparative chromatography. These additional steps not only drastically reduce the overall process yield but also introduce significant operational costs related to solvent consumption, waste disposal, and extended production cycles. Furthermore, the handling of boron-based reagents often requires specific safety protocols and can generate hazardous waste streams that complicate environmental compliance in modern chemical facilities. 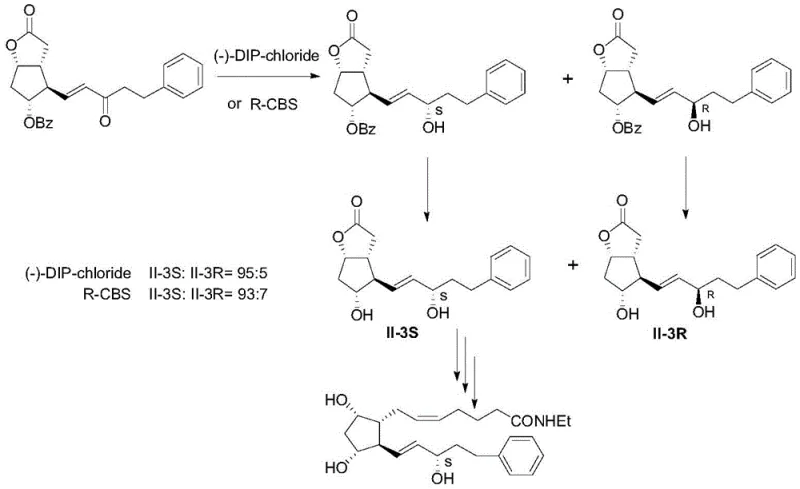
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the unique stereoelectronic properties of (S)-BINAL-H to drive the reduction reaction with exceptional selectivity, effectively bypassing the formation of the detrimental R-isomer. By utilizing this specific chiral reducing agent, the process achieves an isomer ratio greater than 99.5:0.5 directly from the reaction mixture, rendering the costly chiral resolution steps entirely obsolete. This methodological shift simplifies the workflow from a multi-stage purification nightmare into a streamlined sequence of reaction and standard work-up, significantly enhancing the throughput potential for commercial scale-up. The elimination of chromatographic separation not only accelerates the manufacturing timeline but also substantially reduces the consumption of silica gel and elution solvents, contributing to a greener and more cost-effective production profile. For supply chain managers, this translates to a more robust and predictable supply of high-purity intermediates, mitigating the risks associated with batch failures or purity deviations that often plague complex chiral syntheses.
Mechanistic Insights into (S)-BINAL-H Catalyzed Asymmetric Reduction
The core of this technological advancement lies in the in-situ generation and application of (S)-BINAL-H, a chiral aluminum hydride complex derived from lithium aluminum hydride, an alcohol, and S-binaphthol. The mechanism involves the coordination of the carbonyl oxygen of the ketone intermediate to the aluminum center, where the bulky binaphthyl ligand creates a highly defined chiral environment that sterically directs the hydride delivery to the si-face of the carbonyl group. This precise spatial arrangement ensures that the hydride attack occurs with minimal competition from the re-face, thereby suppressing the formation of the R-configured alcohol impurity at the molecular level. The reaction is conducted in an inert solvent such as tetrahydrofuran under strict argon protection to prevent the decomposition of the sensitive hydride species by moisture or oxygen, which is critical for maintaining the reagent's activity and selectivity throughout the batch cycle. Understanding this coordination chemistry is vital for process chemists aiming to replicate the high yields reported, as the stoichiometry of the reagents—specifically the molar ratio of intermediate I to (S)-BINAL-H—plays a pivotal role in driving the reaction to completion without excess reagent waste. 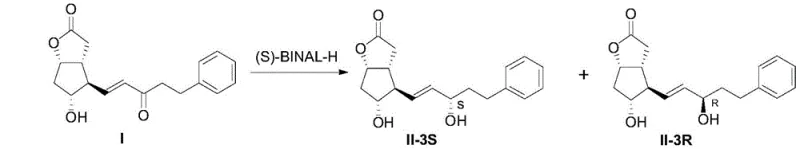
Impurity control in this system is inherently built into the reaction design rather than relying on post-reaction filtration, which is a paradigm shift from traditional quality control strategies. The high chiral purity of the resulting intermediate II ensures that the downstream synthesis of bimatoprost proceeds with minimal carryover of the R-hydroxy isomer, keeping the final drug substance impurity levels well below the 0.2% threshold required by pharmacopeial standards. This intrinsic purity reduces the burden on analytical laboratories, as less intensive monitoring is required to ensure batch conformity, and it minimizes the risk of regulatory rejection during the drug approval or audit phases. The thermal profile of the reaction, involving an initial cooling to -105°C followed by a controlled warm-up to -70°C, is engineered to maximize the kinetic selectivity of the reduction while maintaining a practical reaction rate. This balance between thermodynamic control and kinetic efficiency is what allows the process to achieve yields exceeding 90% while maintaining the rigorous stereochemical integrity demanded by ophthalmic therapeutics.
How to Synthesize Bimatoprost Key Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this high-efficiency route in a GMP-compliant environment, focusing on the precise preparation of the reducing agent followed by the controlled addition of the substrate. The process begins with the generation of the active (S)-BINAL-H species at ambient temperatures before transitioning to cryogenic conditions for the reduction step, a sequence that optimizes reagent stability and reaction performance. Detailed standardized operating procedures regarding reagent concentrations, addition rates, and temperature ramping are essential to replicate the >99.5% chiral purity demonstrated in the experimental examples.
- Preparation of the chiral reducing agent (S)-BINAL-H by reacting lithium aluminum hydride with methanol or ethanol and S-binaphthol in tetrahydrofuran at 20-30°C under argon protection.
- Cooling the (S)-BINAL-H solution to -105 to -95°C and adding the solution of Intermediate I to initiate the asymmetric reduction reaction.
- Gradually warming the reaction mixture to -80 to -70°C to complete the reduction, followed by quenching, extraction, and purification to isolate Intermediate II with >99.5% chiral purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this (S)-BINAL-H mediated reduction pathway offers profound advantages for procurement and supply chain operations, primarily through the simplification of the manufacturing process and the reduction of material costs. By eliminating the need for expensive chiral resolving agents and the associated chromatographic purification infrastructure, manufacturers can significantly lower the variable cost per kilogram of the intermediate. This cost reduction is not merely theoretical but is derived from the tangible removal of unit operations that traditionally consume high volumes of solvents, stationary phases, and energy. For procurement managers, this means a more stable pricing structure for the intermediate, as the process is less susceptible to fluctuations in the cost of specialized purification materials or the capacity constraints of third-party purification service providers. 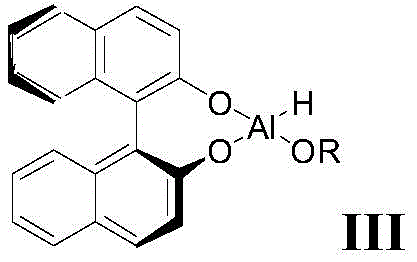
- Cost Reduction in Manufacturing: The primary driver for cost efficiency in this process is the drastic simplification of the downstream processing requirements. Traditional routes require extensive chromatographic separation to remove the R-isomer, a step that is both time-consuming and resource-intensive, often accounting for a significant portion of the total manufacturing cost. By achieving high stereoselectivity directly in the reaction pot, this new method removes the need for silica gel columns and large volumes of elution solvents, leading to substantial savings in raw material procurement and waste treatment expenses. Furthermore, the higher overall yield of the reaction means that less starting material is required to produce the same amount of final product, effectively amplifying the purchasing power of the raw material budget and improving the overall margin profile of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that have multiple failure points, particularly in chiral resolution steps where batch-to-batch variability can be high. The robust nature of the (S)-BINAL-H reduction, with its wide operational window and high intrinsic selectivity, reduces the risk of batch failures and off-spec material that could disrupt the supply of the final API. Additionally, the reagents required for this method, such as lithium aluminum hydride and binaphthol derivatives, are commercially available from multiple global suppliers, reducing the risk of single-source dependency that often plagues proprietary chiral catalysts. This diversification of the supply base ensures that production schedules can be maintained even in the face of regional supply disruptions or logistical delays, providing a more resilient foundation for long-term product availability.
- Scalability and Environmental Compliance: Scaling chiral processes from the laboratory to commercial tonnage often reveals hidden bottlenecks related to heat transfer and mass transfer during purification, issues that are largely circumvented by this streamlined approach. The absence of chromatographic steps simplifies the equipment train required for production, allowing for larger batch sizes in standard reactor vessels without the need for specialized preparative HPLC systems. From an environmental standpoint, the reduction in solvent usage and the elimination of silica waste align with increasingly stringent global regulations regarding chemical manufacturing emissions and waste disposal. This compliance advantage not only mitigates regulatory risk but also enhances the corporate sustainability profile, a factor that is becoming increasingly important for partnerships with major multinational pharmaceutical companies who prioritize green chemistry initiatives in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route, providing clarity on its operational feasibility and quality advantages. These insights are derived directly from the experimental data and claims within the patent documentation, ensuring that the information provided is grounded in verified scientific evidence.
Q: How does the (S)-BINAL-H method improve chiral purity compared to traditional DIP-chloride reduction?
A: Traditional methods using (-)-DIP-chloride or R-CBS typically yield an isomer ratio of 95:5 or 93:7 (II-3S:II-3R), necessitating complex chromatographic separation. The (S)-BINAL-H method described in patent CN109232494B achieves a ratio exceeding 99.5:0.5, effectively eliminating the need for costly chiral resolution steps.
Q: What are the critical reaction conditions for maintaining high yield in this synthesis?
A: Maintaining a strict inert atmosphere (argon protection) and precise temperature control are vital. The reduction must be initiated at cryogenic temperatures between -105°C and -95°C, then carefully warmed to -80°C to -70°C. Deviations can compromise the stereoselectivity and overall yield, which is otherwise reported above 90%.
Q: Is chromatographic separation required for the final intermediate purification?
A: No. A significant advantage of this novel process is that the post-treatment involves standard work-up procedures such as methanol quenching, ethyl acetate extraction, and drying. The high intrinsic stereoselectivity of the (S)-BINAL-H reagent removes the requirement for preparative chromatography, significantly reducing processing time and solvent waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bimatoprost Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the production of life-saving ophthalmic medications, and we possess the technical expertise to bring this advanced (S)-BINAL-H reduction technology to commercial reality. Our R&D team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent concept to industrial reality is seamless and compliant with international GMP standards. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art chiral HPLC capabilities to verify that every batch of bimatoprost intermediate meets the >99.5% chiral purity benchmark required for downstream API synthesis. Our commitment to quality assurance ensures that our partners receive material that is ready for immediate use in their own synthesis campaigns, minimizing the need for additional testing or reprocessing upon receipt.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain to achieve significant operational efficiencies. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your production volume and regional constraints. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics rather than theoretical projections. Our goal is to serve as a strategic extension of your manufacturing capabilities, providing the reliability and technical depth necessary to support the global demand for high-quality glaucoma treatments.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
