Advanced Solvent-Free Synthesis of Scopolamine Butylbromide for Commercial Scale-Up
Advanced Solvent-Free Synthesis of Scopolamine Butylbromide for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with economic efficiency, particularly for established therapeutic agents like Scopolamine Butylbromide. A pivotal advancement in this domain is documented in patent CN102146079A, which outlines a novel preparation method that fundamentally alters the traditional quaternization landscape. This technology addresses critical bottlenecks associated with solvent toxicity and prolonged reaction cycles, offering a streamlined approach that leverages the dual functionality of n-butyl bromide as both a reactant and a reaction medium. For R&D Directors and Procurement Managers evaluating long-term supply strategies, understanding the mechanistic shifts in this patent is essential for securing a reliable Active Pharmaceutical Ingredients supplier capable of delivering consistent quality. The transition from conventional solvent-based systems to this solvent-free protocol represents a significant leap in process intensification, directly impacting the cost structure and environmental footprint of production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Scopolamine Butylbromide has relied heavily on methods utilizing acetonitrile as the primary reaction medium, a practice fraught with logistical and safety challenges. Traditional protocols typically require heating Scopolamine and n-butyl bromide at 65°C for an exhaustive duration of up to 160 hours to achieve a modest yield of approximately 65%. The reliance on acetonitrile introduces severe complications, as it is classified as a highly flammable and toxic liquid, necessitating stringent safety measures and complex waste treatment procedures that inflate operational expenditures. Furthermore, the extended reaction time not only ties up reactor capacity, reducing overall throughput, but also increases the energy consumption required to maintain reflux conditions over nearly a week. These inefficiencies create a fragile supply chain vulnerable to raw material price volatility and regulatory scrutiny regarding volatile organic compound (VOC) emissions, making the conventional route increasingly unsustainable for modern cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the innovative methodology disclosed in the patent data eliminates the need for external solvents entirely, capitalizing on the excellent solubility of Scopolamine in n-butyl bromide and the poor solubility of the resulting product. By increasing the reaction temperature to a range of 70°C to 100°C, specifically optimizing at 90°C, the reaction kinetics are dramatically accelerated, reducing the total processing time to merely 30 hours while boosting yields to 82%. This solvent-free strategy not only simplifies the post-reaction workup by avoiding solvent removal steps but also allows for the easy recovery and recycling of excess n-butyl bromide, creating a closed-loop system that minimizes waste. For Supply Chain Heads, this translates to a more resilient production model where reactor turnover is significantly faster, and the dependency on hazardous auxiliary chemicals is removed, thereby enhancing supply chain reliability and reducing lead time for high-purity Active Pharmaceutical Ingredients.
Mechanistic Insights into Solvent-Free Quaternization
The core of this technological breakthrough lies in the exploitation of specific physicochemical properties to drive the quaternization equilibrium forward without the dilution effects of an inert solvent. In this system, n-butyl bromide serves a dual purpose: it acts as the alkylating agent providing the butyl group and simultaneously functions as the reaction medium, ensuring high local concentration of reactants which favors the nucleophilic attack of the tertiary amine nitrogen on the alkyl halide. The reaction proceeds through a standard SN2 mechanism where the lone pair electrons on the nitrogen atom of the Scopolamine tropane ring attack the electrophilic carbon of the n-butyl bromide, displacing the bromide ion. The absence of acetonitrile prevents the stabilization of the transition state by polar aprotic solvation in a way that might hinder product precipitation, instead allowing the product, Scopolamine Butylbromide, to precipitate out of the excess reagent as it forms due to its low solubility in the alkyl halide matrix.
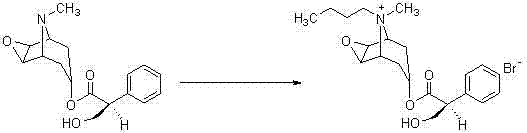
Controlling impurities in this solvent-free environment is achieved through precise thermal management and a rigorous recrystallization protocol using dehydrated ethanol. The elevated temperature of 90°C ensures complete conversion of the starting material, minimizing the presence of unreacted Scopolamine which could complicate downstream purification. Following the reaction, the crude product undergoes a specialized refining process where it is dissolved in heated dehydrated ethanol and subsequently cooled to 8°C to 10°C, inducing selective crystallization of the target molecule while leaving soluble impurities in the mother liquor. This step is critical for achieving the reported purity of 99.2% as verified by HPLC analysis, meeting the stringent specifications required for gastrointestinal therapeutics. The ability to wash the crystals with ice-cold absolute ethanol further removes surface-adhered impurities and residual reagents, ensuring a final product that is chemically stable and suitable for direct formulation or further processing.
How to Synthesize Scopolamine Butylbromide Efficiently
Implementing this synthesis route requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility and safety on a commercial scale. The process begins with the precise charging of Scopolamine and n-butyl bromide into a reactor equipped with efficient reflux condensation capabilities, maintaining a molar excess of the alkylating agent to drive the reaction to completion. Operators must monitor the temperature closely to sustain the 90°C reflux condition for the designated 30-hour period, after which the mixture is cooled to facilitate phase separation and solvent recovery. The detailed standardized synthetic steps, including specific filtration rates, drying pressures of -0.095Mpa, and washing volumes, are critical for maintaining batch-to-batch consistency and are outlined in the technical guide below.
- Mix Scopolamine and n-butyl bromide in a 1: 3 ratio and reflux at 90°C for 30 hours without additional solvent.
- Cool the reaction mixture to room temperature, separate oily liquids, and evaporate residue to dryness.
- Recrystallize the crude product using dehydrated ethanol, filter, wash with ice-cold ethanol, and vacuum dry at 70°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this solvent-free quaternization process offers profound advantages that extend beyond simple yield improvements, fundamentally reshaping the cost basis of production. By eliminating the procurement, handling, and disposal of acetonitrile, manufacturers can realize substantial cost savings associated with raw material acquisition and hazardous waste management compliance. The drastic reduction in reaction time from 160 hours to 30 hours significantly enhances asset utilization, allowing existing infrastructure to produce higher volumes without capital expansion, which is a key driver for cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the ability to recover and reuse excess n-butyl bromide creates a circular economy within the reactor, lowering the effective consumption rate of this key reagent and insulating the production cost from market fluctuations.
- Cost Reduction in Manufacturing: The elimination of acetonitrile removes a significant line item from the bill of materials, as this solvent is not only expensive but also incurs high costs for recovery and environmental abatement. Additionally, the energy demand is drastically lowered due to the shortened reaction cycle, meaning less steam or electricity is consumed per kilogram of product produced. The recovery of unreacted n-butyl bromide further contributes to margin expansion, as the effective stoichiometric ratio is optimized, ensuring that every dollar spent on raw materials contributes maximally to the final output value.
- Enhanced Supply Chain Reliability: Shortening the production cycle from nearly a week to just over a day dramatically improves responsiveness to market demand spikes, reducing the risk of stockouts for downstream formulators. The removal of toxic solvents simplifies logistics and storage requirements, as there is no longer a need to manage large inventories of flammable Class I liquids, thereby reducing insurance premiums and regulatory overhead. This streamlined process ensures a more predictable delivery schedule, making it easier for procurement managers to plan inventory levels and reduce the safety stock required to buffer against production delays.
- Scalability and Environmental Compliance: The solvent-free nature of the reaction inherently reduces the volume of waste generated, simplifying the effluent treatment process and ensuring easier compliance with increasingly strict environmental regulations. The process is highly scalable because it avoids the heat transfer limitations often associated with large volumes of solvent, allowing for safer operation in larger reactors without the risk of thermal runaway associated with flammable solvents. This environmental robustness future-proofs the supply chain against regulatory changes, ensuring long-term continuity of supply without the risk of production shutdowns due to non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method, derived directly from the patent specifications and process benefits. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios or for procurement specialists assessing supplier capabilities. The answers provided reflect the specific advantages of the solvent-free approach over traditional acetonitrile-based methods, highlighting the tangible improvements in efficiency and safety.
Q: How does the solvent-free method improve yield compared to acetonitrile methods?
A: By eliminating acetonitrile and using excess n-butyl bromide as the solvent, the reaction temperature can be safely increased to 90°C, reducing reaction time from 160 hours to 30 hours and improving yield from 65% to 82%.
Q: What are the safety advantages of removing acetonitrile from the process?
A: Acetonitrile is a toxic, flammable Class I liquid. Removing it eliminates significant safety hazards and environmental disposal costs, simplifying regulatory compliance and workplace safety protocols.
Q: Can the excess n-butyl bromide be recovered?
A: Yes, the physicochemical properties allow for easy separation of excess n-butyl bromide after the reaction, enabling recovery and reuse which further drives down raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Scopolamine Butylbromide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN102146079A to maintain competitiveness in the global pharmaceutical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this solvent-free process are fully realized in large-scale operations. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of Scopolamine Butylbromide meets the highest international standards for safety and efficacy required by regulatory bodies worldwide.
We invite you to collaborate with us to optimize your supply chain and leverage these technological advancements for your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this improved synthesis method can lower your total landed cost. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both your product quality and bottom-line performance.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
