Advanced Catalytic Distillation Technology for Scalable High-Purity Sulfoxide Manufacturing
The chemical industry is constantly evolving, driven by the need for more efficient and sustainable synthesis methods for critical intermediates. Patent CN111072528A introduces a groundbreaking approach to preparing sulfoxides, specifically addressing the long-standing challenges associated with purity and catalyst longevity in oxidation reactions. This technology leverages a sophisticated catalytic distillation reactor system that integrates a titanium silicalite molecular sieve with an alkaline ion exchange resin. By combining these catalytic and separation functions within a single unit operation, the process achieves a significant reduction in impurity accumulation, particularly in the recycled thioether streams. For R&D directors and procurement specialists seeking a reliable sulfoxide supplier, this innovation represents a pivotal shift towards more robust and cost-effective manufacturing protocols. The method not only enhances the selectivity of the sulfoxide product but also ensures the stability of the catalytic system over extended operational periods, thereby securing a consistent supply chain for high-value chemical intermediates used in pharmaceuticals and fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing sulfoxides, such as the oxidation of dimethyl sulfide, have historically relied on batch processes that suffer from significant inefficiencies and purity issues. Conventional techniques often involve the use of nitrogen dioxide or nitric acid as oxidants, which can lead to the formation of hazardous byproducts and require complex neutralization and distillation steps to achieve acceptable purity levels. Furthermore, in standard catalytic oxidation processes using molecular sieves, the accumulation of high-boiling impurities in the recycled unreacted thioether stream poses a critical bottleneck. These impurities tend to deposit on the active centers of the catalyst, gradually reducing its activity and necessitating frequent regeneration or replacement. This degradation of catalyst performance not only increases operational costs but also introduces variability in product quality, making it difficult to maintain the stringent purity specifications required for pharmaceutical applications. The inability to effectively separate the product from the reactants in situ often results in lower selectivity and higher energy consumption due to the need for downstream purification.
The Novel Approach
The novel approach disclosed in the patent overcomes these limitations by employing a catalytic distillation reactor packed with both a titanium silicalite molecular sieve and an alkaline ion exchange resin. This dual-catalyst system operates within a continuous flow regime where reaction and separation occur simultaneously, effectively shifting the equilibrium towards the desired sulfoxide product. The strategic placement of the alkaline ion exchange resin in the lower middle part of the reaction section or in the reboiler plays a crucial role in scavenging acidic impurities and preventing their accumulation in the recycle loop. By maintaining a cleaner reaction environment, the titanium silicalite catalyst retains its activity for much longer durations, ensuring stable production rates and consistent product quality. This integration allows for the use of greener oxidants like hydrogen peroxide, significantly reducing the environmental footprint compared to traditional nitric acid methods. For a reliable sulfoxide supplier, this technology offers a pathway to cost reduction in pharmaceutical intermediate manufacturing by minimizing waste generation and maximizing raw material utilization through efficient recycling of unreacted thioether.
Mechanistic Insights into Titanium Silicalite-Catalyzed Oxidation
The core of this innovative process lies in the synergistic interaction between the titanium silicalite molecular sieve and the alkaline ion exchange resin within the catalytic distillation column. The titanium silicalite, such as TS-1, acts as the primary oxidation catalyst, activating the peroxide oxidant to selectively oxidize the sulfide group to a sulfoxide without over-oxidation to sulfone. The unique framework structure of the titanium silicalite provides specific active sites that facilitate this transformation with high efficiency. However, the longevity of this catalyst is often compromised by the presence of impurities that can block these active sites. This is where the alkaline ion exchange resin becomes indispensable. Positioned strategically within the reactor, the resin neutralizes acidic byproducts and adsorbs polar impurities that would otherwise poison the molecular sieve. This protective mechanism ensures that the catalytic activity remains high throughout the continuous operation, allowing for sustained high-purity sulfoxide production. The hydrolytic condensation of organic silicon sources, represented by general structures such as the one shown below, is critical in forming the robust catalyst framework necessary for this demanding application.
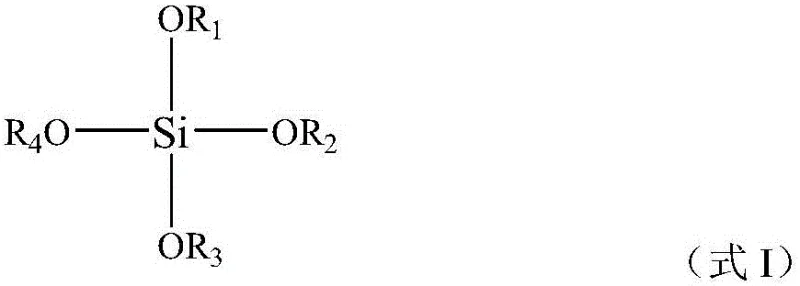
Furthermore, the preparation of the titanium silicalite catalyst itself involves precise control over the templating agents used during hydrothermal crystallization. The use of quaternary ammonium bases, as illustrated by the general structure below, directs the formation of the specific zeolite topology required for optimal catalytic performance. The patent details a method where the template agent is partially recycled via condensate return, reducing the overall consumption of these expensive organic modifiers. This not only lowers the manufacturing cost of the catalyst but also minimizes the environmental impact associated with template disposal. The resulting catalyst exhibits a high degree of crystallinity and titanium incorporation, which are essential for achieving the high selectivity observed in the sulfoxide oxidation reaction. By understanding these mechanistic details, R&D teams can better appreciate the technical depth behind the process and its potential for producing high-purity dimethyl sulfoxide and other sulfoxide derivatives suitable for sensitive electronic or pharmaceutical applications.
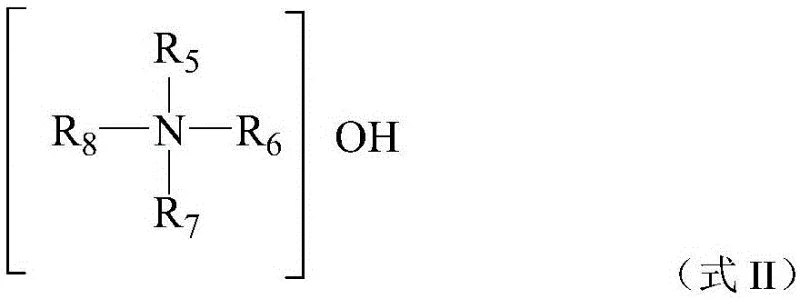
How to Synthesize Dimethyl Sulfoxide Efficiently
Implementing this advanced synthesis route requires a systematic approach to reactor configuration and operational parameter control to fully realize its benefits. The process begins with the careful preparation of the titanium silicalite catalyst, ensuring the correct silicon-to-titanium ratio and crystal structure are achieved through controlled hydrothermal synthesis. Once the catalyst is prepared and shaped, it is loaded into the catalytic distillation reactor along with the alkaline ion exchange resin in the designated zones. The reactor is then brought to the desired operating temperature and pressure, typically using methanol as a solvent to facilitate the reaction and separation dynamics. Thioether and oxidant are fed into the reactor at specific theoretical plate positions to optimize contact time and conversion efficiency. The detailed standardized synthesis steps see the guide below.
- Prepare the titanium silicalite molecular sieve catalyst using hydrolytic condensation of organic silicon and titanium sources followed by hydrothermal crystallization.
- Load the catalytic distillation reactor with the catalyst and alkaline ion exchange resin in the specified reaction zones.
- Feed thioether and oxidant into the reactor under controlled temperature and pressure to achieve continuous oxidation and separation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic distillation technology offers substantial strategic advantages beyond mere technical performance. The primary benefit lies in the significant stabilization of the supply chain for critical sulfoxide intermediates. By enabling continuous operation with extended catalyst life, the process reduces the frequency of shutdowns for catalyst regeneration or replacement, thereby ensuring a more consistent and reliable output of high-purity dimethyl sulfoxide. This reliability is crucial for downstream manufacturers who depend on just-in-time delivery of solvents and intermediates for their own production schedules. Additionally, the enhanced selectivity of the process means that less raw material is wasted in the formation of byproducts, leading to a more efficient use of resources and a reduction in the overall cost of goods sold. The ability to recycle unreacted thioether with minimal impurity buildup further contributes to cost optimization by lowering the demand for fresh feedstock. These factors combined create a more resilient and cost-effective supply chain model that can better withstand market fluctuations and raw material price volatility.
- Cost Reduction in Manufacturing: The integration of reaction and separation in a single unit significantly reduces capital expenditure and operational complexity compared to traditional multi-step batch processes. By eliminating the need for separate neutralization and extensive purification steps, the process streamlines the production workflow, leading to lower labor and utility costs. The reduced consumption of template agents during catalyst preparation also contributes to lower material costs, while the extended catalyst life minimizes the frequency of costly catalyst purchases. Furthermore, the use of hydrogen peroxide as a clean oxidant reduces waste treatment costs associated with hazardous nitrogen oxides, providing a clear economic advantage in terms of environmental compliance and waste disposal fees.
- Enhanced Supply Chain Reliability: The continuous nature of the catalytic distillation process ensures a steady stream of product, mitigating the risks associated with batch-to-batch variability and production delays. The robust design of the reactor system, which effectively manages impurity accumulation, means that unplanned maintenance events are significantly reduced, leading to higher overall equipment effectiveness. This reliability allows supply chain managers to plan inventory levels more accurately and reduce the need for safety stock, freeing up working capital. The ability to scale this technology from pilot to commercial production with consistent performance provides confidence in long-term supply agreements, making it an attractive option for companies seeking a reliable sulfoxide supplier for their critical manufacturing needs.
- Scalability and Environmental Compliance: The process is inherently scalable, as the catalytic distillation columns can be designed to handle increased throughput without compromising reaction efficiency or product quality. This scalability supports the growing demand for high-purity sulfoxides in emerging markets such as electronics and advanced pharmaceuticals. From an environmental perspective, the process aligns with green chemistry principles by using safer oxidants and generating less hazardous waste. The reduced energy consumption due to the integration of reaction and separation also lowers the carbon footprint of the manufacturing process. These environmental benefits not only help companies meet regulatory requirements but also enhance their corporate sustainability profiles, which is increasingly important for stakeholders and customers in the global chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this sulfoxide preparation technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of adopting this method for large-scale production. The insights provided here aim to clarify the operational advantages and the specific mechanisms that drive the improved performance of this catalytic system.
Q: How does the alkaline ion exchange resin improve sulfoxide purity?
A: The alkaline ion exchange resin effectively reduces impurity content in the unreacted thioether stream, inhibiting impurity accumulation and catalyst deactivation.
Q: What are the advantages of using catalytic distillation for sulfoxide production?
A: Catalytic distillation integrates reaction and separation, allowing for continuous operation, higher selectivity, and easier recycling of unreacted materials.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for continuous operation with stable catalyst performance, making it highly suitable for commercial scale-up and consistent supply.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dimethyl Sulfoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced manufacturing technologies to meet the evolving demands of the global pharmaceutical and fine chemical industries. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in patent CN111072528A can be successfully translated into reliable industrial operations. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging our deep technical knowledge and robust infrastructure, we can provide our partners with a secure and consistent supply of high-quality sulfoxides that adhere to the highest industry standards. Our dedication to quality and reliability makes us a trusted partner for companies seeking to optimize their supply chains and enhance their product portfolios.
We invite you to collaborate with us to explore how this advanced catalytic distillation technology can benefit your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your production requirements, demonstrating the potential economic and operational advantages of switching to this superior method. Please contact us to request specific COA data and route feasibility assessments that will help you make informed decisions about your sourcing strategy. By partnering with NINGBO INNO PHARMCHEM, you gain access to not just a product, but a comprehensive solution that drives efficiency and value across your entire manufacturing process.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
