Advanced Synthesis of Chiral (R)-Alpha-Phenylethylamine Acetate for Pharmaceutical Catalysis
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access chiral building blocks, which are essential for the synthesis of active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in patent CN101973890B, which details a novel method for preparing and synthesizing chiral (R)-alpha-phenylethylamine acetate. This compound, initially discovered somewhat serendipitously during attempts to synthesize a cobalt complex, has emerged as a highly effective catalyst for carbon-carbon bond-forming reactions. The patent outlines a streamlined, one-step synthetic route that converts (R)-alpha-phenylethylamine and cobalt acetate tetrahydrate into the target acetate salt with remarkable ease. This development represents a pivotal shift towards simpler, more cost-effective methodologies for generating chiral catalysts, addressing the growing demand for reliable suppliers of high-performance chemical intermediates in the global market.
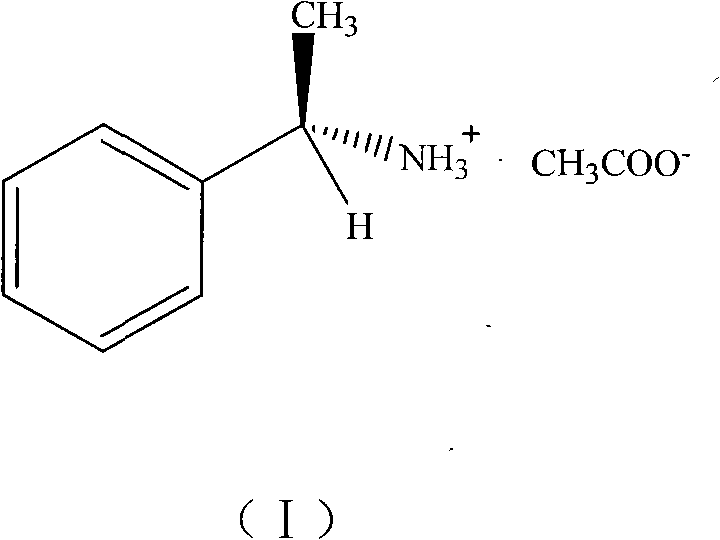
The structural integrity and purity of this chiral salt are paramount for its downstream applications, particularly in asymmetric catalysis where stereochemical fidelity dictates the success of the final drug substance. The methodology described ensures that the chiral information from the starting amine is preserved and effectively utilized in the resulting salt form. By leveraging this specific acetate salt, manufacturers can access a versatile tool for organic synthesis that bridges the gap between academic discovery and industrial application. The implications for supply chain stability are profound, as the simplified production process reduces the risk of batch-to-batch variability and ensures a consistent supply of this critical reagent for R&D teams worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the preparation of chiral ammonium salts and related catalytic species has been fraught with challenges that hinder large-scale adoption. Conventional synthetic routes often involve multi-step procedures requiring stringent anhydrous conditions, expensive chiral ligands, or complex resolution processes to achieve the desired enantiomeric purity. These legacy methods frequently necessitate the use of hazardous solvents and generate significant amounts of waste, complicating environmental compliance and driving up production costs. Furthermore, the isolation of pure chiral salts from reaction mixtures can be technically demanding, often requiring recrystallization from specific solvent systems or chromatographic purification, which are not feasible for ton-scale manufacturing. The reliance on transition metal complexes that are difficult to remove from the final product also poses a significant hurdle for pharmaceutical applications, where residual metal limits are strictly regulated.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data offers a paradigm shift towards operational simplicity and economic efficiency. The synthesis of chiral (R)-alpha-phenylethylamine acetate is achieved through a direct reaction between (R)-alpha-phenylethylamine and cobalt acetate tetrahydrate in an absolute ethanol solution. This one-pot strategy eliminates the need for intermediate isolation or complex workup procedures. The process involves a straightforward reflux for 24 hours, followed by hot filtration to remove insolubles and natural volatilization of the solvent to yield the product as clear crystals. This drastic simplification of the workflow not only reduces the time required for production but also minimizes the consumption of resources and energy. The ability to obtain the target compound in high purity without extensive purification steps makes this method exceptionally attractive for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Cobalt-Catalyzed Henry Reaction
The true value of chiral (R)-alpha-phenylethylamine acetate lies in its application as a catalyst for the Henry reaction, a classic carbon-carbon bond-forming reaction between nitroalkanes and carbonyl compounds. In this mechanistic pathway, the catalyst facilitates the deprotonation of the nitroalkane (such as nitromethane) to generate a nucleophilic nitronate species. This activated species then attacks the electrophilic carbonyl carbon of the aldehyde (such as benzaldehyde), leading to the formation of a beta-nitroalcohol. The presence of the chiral ammonium salt influences the stereochemical outcome of this addition, although the primary focus here is on the high conversion efficiency. The cobalt center likely plays a role in coordinating the reactants, stabilizing the transition state, and enhancing the acidity of the alpha-protons in the nitroalkane, thereby accelerating the reaction rate under mild conditions.
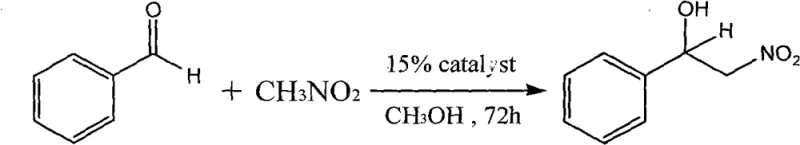
Experimental data from the patent highlights the robustness of this catalytic system. When employed in the reaction between benzaldehyde and nitromethane in methanol, the catalyst achieves a remarkable yield of 96% after 72 hours of stirring at room temperature. This high yield is achieved with a catalyst loading of only 15%, demonstrating the efficiency of the system. The reaction produces 2-nitro-1-phenylethyl alcohol, a valuable bifunctional intermediate that can be further transformed into amino alcohols or other pharmacologically active scaffolds. The mild reaction conditions (room temperature, atmospheric pressure) and the use of common solvents like methanol make this process highly adaptable for various substrates, offering R&D directors a reliable platform for exploring new chemical space without the need for specialized high-pressure equipment or cryogenic cooling.
How to Synthesize (R)-Alpha-Phenylethylamine Acetate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing this valuable catalyst with minimal infrastructure requirements. The process is designed to be robust and forgiving, making it suitable for both laboratory-scale optimization and pilot-plant production. By adhering to the specified molar ratios and reaction times, manufacturers can consistently produce the catalyst with the necessary quality attributes for downstream applications. The following guide summarizes the critical operational parameters derived from the patent examples, serving as a foundation for process engineers to develop standardized operating procedures (SOPs).
- React (R)-alpha-phenylethylamine with cobalt acetate tetrahydrate in an absolute ethanol solution under reflux conditions for 24 hours.
- Perform hot filtration on the reaction mixture to remove any insoluble impurities or unreacted solids while maintaining temperature.
- Allow the filtrate to stand and naturally volatilize the solvent to obtain the target chiral (R)-alpha-phenylethylamine acetate as clear crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible benefits that extend beyond mere technical performance. The shift towards a simpler, one-step manufacturing process directly translates to reduced operational expenditures and enhanced supply security. By eliminating complex purification stages and reducing the dependency on exotic reagents, the overall cost structure of producing this chiral intermediate is significantly optimized. This economic efficiency allows for more competitive pricing strategies in the global market, ensuring that downstream pharmaceutical manufacturers can access high-quality catalysts without compromising their margins. Furthermore, the use of widely available starting materials mitigates the risk of supply disruptions caused by geopolitical factors or raw material shortages.
- Cost Reduction in Manufacturing: The streamlined nature of the synthesis process drives substantial cost savings by minimizing unit operations. Traditional methods often require multiple reaction vessels, extensive washing steps, and energy-intensive drying processes, all of which contribute to higher utility and labor costs. In contrast, the new method relies on a single reflux step followed by simple filtration and evaporation. This reduction in processing time and equipment usage leads to a lower cost of goods sold (COGS). Additionally, the high yield of the subsequent catalytic application (96%) means that less catalyst is wasted, further improving the overall atom economy and reducing the effective cost per kilogram of the final pharmaceutical intermediate produced.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals as feedstocks. Both (R)-alpha-phenylethylamine and cobalt acetate tetrahydrate are commercially available in bulk quantities from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions—specifically the tolerance for standard solvents like ethanol and methanol—means that production can be easily transferred between different manufacturing sites without the need for specialized infrastructure. This flexibility ensures continuous availability of the catalyst, preventing bottlenecks in the production schedules of client companies who rely on timely delivery of critical raw materials for their own API synthesis campaigns.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns well with modern green chemistry principles. The avoidance of chlorinated solvents in the synthesis step (using ethanol instead) and the ability to recover solvents through distillation reduce the environmental footprint of the manufacturing process. The simplicity of the workup (filtration and volatilization) generates less aqueous waste compared to extraction-heavy protocols. As regulatory pressures on chemical manufacturing intensify, having a process that is inherently cleaner and easier to manage from a waste disposal standpoint provides a strategic advantage. This ease of scale-up ensures that production can be ramped from kilograms to tons to meet surging market demand without encountering unforeseen engineering challenges.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new catalytic system is crucial for stakeholders evaluating its integration into their existing workflows. The following questions address common inquiries regarding the stability, application scope, and handling of chiral (R)-alpha-phenylethylamine acetate. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of this synthesis method over traditional quaternary ammonium salt preparation?
A: The primary advantage is the simplicity and efficiency of a one-step synthesis that bypasses complex multi-stage reactions. Unlike traditional methods that may require intricate separation techniques or harsh conditions, this process utilizes a straightforward reflux in ethanol followed by simple filtration and natural volatilization, significantly reducing operational complexity and energy consumption.
Q: How effective is this catalyst in Henry reactions for pharmaceutical intermediates?
A: The catalyst demonstrates exceptional efficacy in Henry reactions, specifically achieving a yield of 96% in the synthesis of 2-nitro-1-phenylethyl alcohol from benzaldehyde and nitromethane. Operating at a catalytic loading of just 15% under mild conditions (room temperature stirring in methanol), it offers a robust solution for forming critical C-C bonds in drug synthesis.
Q: Is this process scalable for industrial production of chiral intermediates?
A: Yes, the process is highly scalable due to its reliance on common solvents like ethanol and methanol and standard unit operations such as reflux and filtration. The absence of sensitive reagents or extreme pressure requirements makes it ideal for commercial scale-up, ensuring consistent supply chain reliability for high-purity chiral intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-Alpha-Phenylethylamine Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral catalysts play in accelerating drug discovery and development. Our team of expert chemists has thoroughly analyzed the potential of the technology described in patent CN101973890B and is prepared to support your projects with premium-grade (R)-alpha-phenylethylamine acetate. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the early stages of process research or full-scale manufacturing. Our commitment to quality is unwavering, with stringent purity specifications enforced through our rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced catalytic technology for your next synthesis campaign. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production needs, helping you identify opportunities to optimize your bill of materials. We encourage you to contact our technical procurement team today to request specific COA data for our current inventory and to discuss route feasibility assessments for your target molecules. Let us help you streamline your supply chain and achieve your project milestones with confidence and efficiency.
