Strategic Synthesis of Deoxy Taxane Analogs for Next-Generation Oncology APIs
Strategic Synthesis of Deoxy Taxane Analogs for Next-Generation Oncology APIs
The pharmaceutical industry continuously seeks robust synthetic pathways to overcome the limitations of natural product-derived chemotherapeutics, particularly regarding solubility and drug resistance. Patent CN110105368B, published in early 2022, introduces a groundbreaking methodology for synthesizing 10-deacetyl-9(R)-hydrogenation-9,10-O-(2,3-dihydroxypropylene)-1-deoxidized taxane analogs. This technology represents a significant leap forward for reliable API intermediate suppliers aiming to diversify their oncology portfolios. By modifying the northern hemisphere of the taxane skeleton, specifically the C-7 to C-10 region, this invention addresses the critical bottlenecks of poor water solubility and multi-drug resistance associated with traditional Paclitaxel. The disclosed process utilizes 1-dehydroxy baccatin VI, a readily available precursor extracted from Taxus mairei, ensuring a sustainable supply chain foundation for high-purity pharmaceutical intermediates.
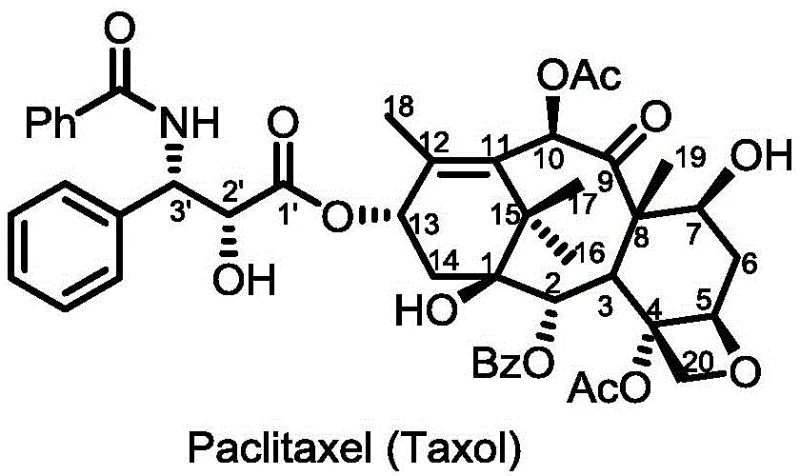
Understanding the structural baseline is crucial for appreciating the innovation behind these deoxy taxane analogs. As illustrated in the reference structure of Paclitaxel, the molecule possesses a complex diterpene core that is notoriously difficult to modify without losing biological activity. The conventional reliance on extraction from yew trees has long plagued the supply chain with issues of low natural content and seasonal variability. Furthermore, the inherent hydrophobicity of the natural product necessitates the use of toxic solvents like Cremophor EL in clinical formulations, which contributes to severe hypersensitivity reactions. The novel approach detailed in the patent strategically alters this scaffold to enhance pharmacokinetic properties while maintaining the essential binding affinity for microtubules, thereby offering a superior therapeutic index for modern cancer treatment regimens.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for producing taxane-based therapeutics are heavily constrained by the scarcity of natural resources and the chemical instability of the core structure. Natural Paclitaxel content in plant sources is exceedingly small, failing to meet the escalating global demand for clinical and basic research applications. Moreover, the structural rigidity of the natural product limits the ability to introduce polar functional groups that could improve bioavailability. The interaction with P-glycoprotein (P-gp) efflux pumps often leads to multi-drug resistance (MDR), rendering treatments ineffective in resistant tumor lines. Additionally, the low water solubility of natural taxanes complicates formulation development, requiring complex delivery systems that increase manufacturing costs and potential toxicity profiles for patients undergoing chemotherapy.
The Novel Approach
The invention presents a sophisticated semi-synthetic route that transforms 1-dehydroxy baccatin VI into highly active analogs through a series of selective modifications. By introducing a 9,10-O-(2,3-dihydroxypropylene) acetal group, the method effectively increases the polarity of the molecule, drastically improving water solubility without compromising the integrity of the taxane ring skeleton. This structural modification is designed to weaken interactions with P-gp transporters, potentially reversing drug resistance mechanisms in tumor cells. The process leverages the abundance of 1-dehydroxy baccatin VI in regional Taxus species, providing a cost-reduction in pharmaceutical intermediates manufacturing by utilizing a more accessible starting material compared to Baccatin III or Paclitaxel itself.
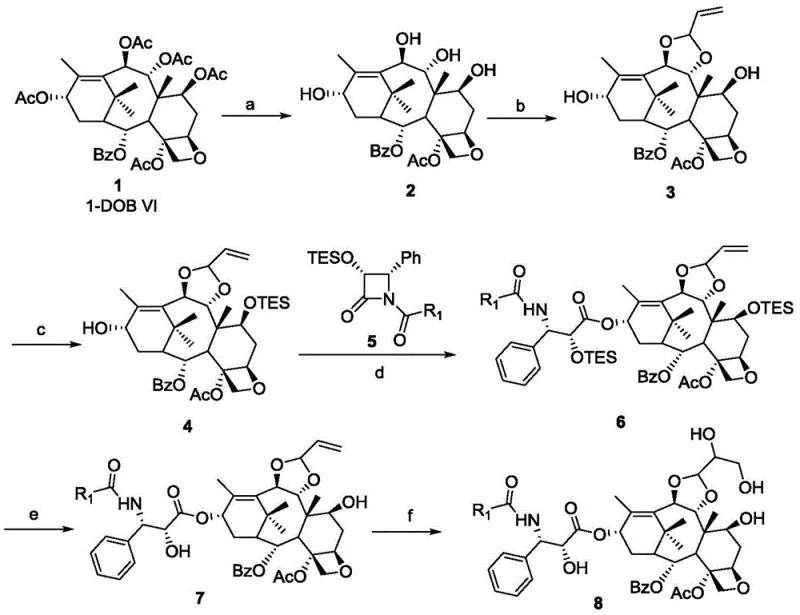
Mechanistic Insights into Semi-Synthetic Taxane Modification
The synthetic pathway relies on precise control of stereochemistry and protective group chemistry to navigate the complex functionality of the taxane core. The initial step involves the selective deacetylation of 1-dehydroxy baccatin VI using hydrazine hydrate in ethanol, which removes acetate groups at positions 7, 9, 10, and 13 to generate the tetra-deacetyl intermediate. This is followed by a critical acetalization reaction where the 9,10-diol system is protected as an allyl acetal using acetal diethanol and camphorsulfonic acid. This step is vital for locking the conformation of the C-ring and preventing unwanted side reactions during subsequent transformations. The selectivity achieved here ensures that the biological activity associated with the C-1/C-2 oxetane ring and the C-4/C-5 acetate remains intact throughout the synthesis.
Further functionalization involves the installation of the C-13 side chain, which is essential for antitumor activity. The process employs a beta-lactam coupling strategy using sodium bis(trimethylsilyl)amide as a strong base to facilitate the nucleophilic attack on the C-13 position. Following the coupling, the removal of the triethylsilyl protecting group at C-7 using tetrabutylammonium fluoride restores the hydroxyl functionality. The final oxidation step utilizes an NMO-osmium tetroxide system to convert the allyl group of the acetal into a 2,3-dihydroxypropylene moiety. This oxidative dihydroxylation is performed under mild conditions to prevent degradation of the sensitive ester linkages, resulting in the final target analogs with enhanced solubility profiles and potent cytotoxicity against various cancer cell lines.
How to Synthesize Deoxy Taxane Analogs Efficiently
The synthesis of these advanced intermediates requires strict adherence to reaction conditions to maintain high purity and yield. The process begins with the preparation of the deacetylated core, followed by sequential protection and coupling steps that build molecular complexity. Each stage demands careful monitoring of stoichiometry and temperature to avoid epimerization or hydrolysis of the sensitive oxetane ring. The detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures that ensure reproducibility on a commercial scale.
- Deacetylate 1-dehydroxy baccatin VI using hydrazine hydrate in ethanol to obtain the tetra-deacetyl intermediate.
- Protect the 9,10-diol system as an allyl acetal using acetal diethanol and camphorsulfonic acid catalyst.
- Perform selective silylation at the C-7 position using triethylchlorosilane and imidazole.
- Couple the C-13 side chain using a beta-lactam precursor and sodium bis(trimethylsilyl)amide base.
- Remove silyl protecting groups with TBAF and oxidize the allyl group to a diol using NMO and osmium tetroxide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical feasibility. The shift from scarce natural extracts to semi-synthetic derivatives based on abundant 1-dehydroxy baccatin VI mitigates the risks associated with agricultural variability and geopolitical supply disruptions. By utilizing a starting material that is easier to separate and exists in higher concentrations within plant biomass, manufacturers can achieve significant cost optimization in raw material acquisition. This transition supports a more resilient supply chain capable of meeting the rigorous quality standards required for oncology drug production while reducing the environmental footprint associated with harvesting large quantities of yew biomass.
- Cost Reduction in Manufacturing: The elimination of complex extraction processes for the final active core allows for a streamlined production workflow that significantly lowers operational expenditures. By avoiding the need for extensive purification of natural Paclitaxel, the process reduces solvent consumption and waste generation, leading to substantial cost savings in downstream processing. The use of standard reagents and ambient temperature reactions for key steps further minimizes energy consumption and equipment requirements, enhancing the overall economic viability of large-scale production runs.
- Enhanced Supply Chain Reliability: Sourcing 1-dehydroxy baccatin VI from established regional supplies of Taxus mairei ensures a consistent and reliable feedstock for continuous manufacturing operations. This stability is crucial for long-term contract fulfillment and prevents the bottlenecks often seen with seasonally dependent natural products. The robustness of the synthetic pathway allows for flexible scaling, enabling suppliers to respond rapidly to fluctuations in market demand without compromising on the stringent purity specifications required for clinical-grade intermediates.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing common organic solvents and reagents that are readily available in the global chemical market. The process avoids the use of heavy metal catalysts in the final steps, simplifying the removal of impurities and ensuring compliance with strict regulatory limits for residual metals in pharmaceutical products. This environmentally conscious approach aligns with green chemistry principles, reducing the burden on waste treatment facilities and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these deoxy taxane analogs. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating these intermediates into existing drug development pipelines.
Q: How do these deoxy taxane analogs compare to natural Paclitaxel in terms of solubility?
A: According to patent CN110105368B, the introduction of the 9,10-O-(2,3-dihydroxypropylene) group significantly enhances water solubility compared to natural paclitaxel, addressing a major formulation challenge.
Q: What is the primary advantage of using 1-dehydroxy baccatin VI as a starting material?
A: 1-dehydroxy baccatin VI is more abundant in Taxus mairei (0.05-0.08%) compared to paclitaxel, making it a more sustainable and cost-effective substrate for large-scale semi-synthesis.
Q: Do these analogs retain antitumor activity against drug-resistant cells?
A: Yes, the data indicates that specific analogs (like compound 8a) exhibit stronger cytotoxicity against A549/T drug-resistant lung cancer cells than paclitaxel, suggesting improved interaction with P-gp transporters.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deoxy Taxane Analog Supplier
As the demand for next-generation oncology therapeutics grows, partnering with an experienced CDMO is essential for navigating the complexities of taxane synthesis. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to industrial reactor. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of deoxy taxane analog meets the highest international standards for safety and efficacy. We understand the critical nature of timeline and quality in the pharmaceutical sector and are committed to delivering consistent results.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific development needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this novel synthetic route for your pipeline. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to excellence in the manufacturing of high-value pharmaceutical intermediates.
