Advanced Manufacturing of Docetaxel Chiral Intermediates via Efficient L-Phenylglycine Route
Advanced Manufacturing of Docetaxel Chiral Intermediates via Efficient L-Phenylglycine Route
The global demand for next-generation antineoplastic agents continues to drive innovation in the synthesis of complex taxane derivatives, with Docetaxel remaining a cornerstone therapy for breast, lung, and prostate cancers. However, the commercial viability of Docetaxel has historically been constrained by the intricate and costly synthesis of its chiral C-13 side chain. A groundbreaking technological advancement detailed in patent CN113636956A, published in late 2021, introduces a robust and economically superior preparation method for Docetaxel chiral side chain intermediates. This novel approach departs from traditional reliance on scarce natural precursors or expensive asymmetric catalysis, instead leveraging the abundant chiral pool of L-phenylglycine. By establishing a concise synthetic pathway that avoids toxic heavy metals and achieves exceptional stereoselectivity, this patent provides a critical blueprint for reliable pharmaceutical intermediate supplier networks aiming to secure the supply chain for life-saving oncology drugs.
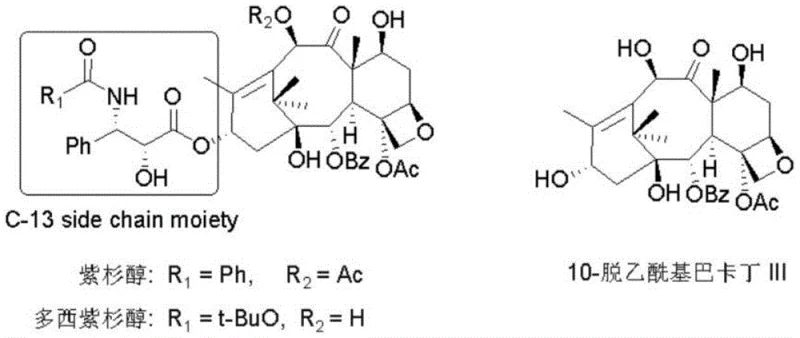
Docetaxel, a semi-synthetic analogue of paclitaxel, exerts its therapeutic effect by stabilizing microtubules and inhibiting cell division, yet its production is bottlenecked by the complexity of attaching the N-tert-butoxycarbonyl-3-phenylisoserine side chain to the 10-deacetylbaccatin III core. The structural integrity of this side chain, specifically the (2R,3S) or (2S,3S) configuration, is paramount for biological activity. As illustrated in the structural overview, the side chain moiety is the differentiating factor that grants Docetaxel superior water solubility and potency compared to its predecessor. The new methodology addresses the urgent industry need for cost reduction in pharmaceutical intermediates manufacturing by replacing multi-step, low-yield routes with a streamlined process that begins with inexpensive, commercially available amino acids, thereby democratizing access to high-quality anticancer ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the Docetaxel side chain has been plagued by significant economic and technical hurdles that impede efficient commercial scale-up of complex pharmaceutical intermediates. Prominent literature methods, such as the Sharpless asymmetric dihydroxylation, rely heavily on potassium osmate (K2OsO2(OH)4) and chiral ligands, which are not only prohibitively expensive but also introduce severe environmental liabilities due to osmium toxicity. Furthermore, alternative strategies involving the resolution of racemic mixtures using resolving agents like ephedrine suffer from an inherent theoretical yield limit of 50%, effectively wasting half of the raw material input. These conventional pathways often require harsh reaction conditions, cryogenic temperatures, and tedious purification steps to remove trace heavy metals, creating substantial operational risks and inflating the cost of goods sold (COGS) for API manufacturers.
The Novel Approach
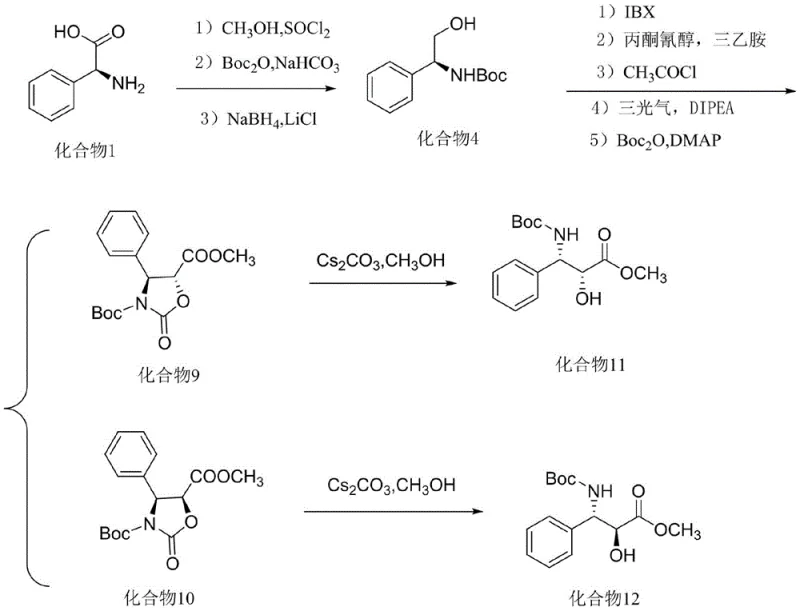
In stark contrast, the methodology disclosed in patent CN113636956A revolutionizes the production landscape by utilizing L-phenylglycine as a foundational chiral building block. This strategy bypasses the need for external chiral catalysts entirely, as the stereochemistry is derived directly from the starting material. The process features a clever sequence involving IBX oxidation, cyanohydrin formation, and a triphosgene-mediated cyclization that generates rigid oxazolidinone intermediates. These intermediates, designated as Compounds 9 and 10, exhibit distinct polarity differences (Rf value difference of 0.15-0.2), allowing for rapid and efficient separation via standard silica gel chromatography. This innovative route not only drastically simplifies the operational workflow but also ensures that the final products, Compounds 11 and 12, are obtained with an enantiomeric excess (ee) exceeding 98%, setting a new benchmark for purity in the sector.
Mechanistic Insights into IBX Oxidation and Triphosgene Cyclization
The chemical elegance of this synthesis lies in its precise control over stereochemistry through the formation of transient cyclic structures. The process initiates with the conversion of L-phenylglycine into a protected amino alcohol (Compound 4) through esterification and reduction. The pivotal step involves the oxidation of this alcohol using 2-iodoxybenzoic acid (IBX), a hypervalent iodine reagent known for its mildness and selectivity, transforming the hydroxyl group into an aldehyde without racemization. Subsequent nucleophilic attack by acetone cyanohydrin introduces the necessary carbon framework, which is then locked into place via cyclization with triphosgene. This cyclization forms a five-membered oxazolidinone ring, effectively freezing the rotational freedom of the molecule and preserving the chiral information inherited from the L-phenylglycine. The resulting diastereomers are stable and easily distinguishable, facilitating high-fidelity separation.
From an impurity control perspective, this mechanism offers distinct advantages over transition-metal catalyzed routes. The absence of osmium or ruthenium eliminates the risk of heavy metal contamination, a critical quality attribute for regulatory compliance in oncology drug manufacturing. Furthermore, the use of cesium carbonate in the final deprotection step ensures mild conditions that prevent epimerization of the sensitive alpha-amino acid center. The patent data indicates that the final products achieve purity levels of 98.5% to 99.0%, demonstrating that the mechanistic design inherently suppresses the formation of unwanted stereoisomers. This level of control is essential for meeting the stringent specifications required by global health authorities for injectable chemotherapy agents.
How to Synthesize Docetaxel Chiral Side Chain Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible roadmap for laboratory and pilot-scale production. The procedure is divided into four logical stages: protection and reduction, oxidative functionalization and cyclization, diastereomer separation, and final deprotection. Each step utilizes common organic solvents such as methanol, ethyl acetate, and tetrahydrofuran, ensuring compatibility with standard reactor setups. The reaction conditions are notably mild, with most transformations occurring at room temperature or under gentle reflux, minimizing energy consumption and thermal degradation risks. For a detailed breakdown of the specific molar ratios, solvent volumes, and workup procedures required to execute this synthesis, please refer to the standardized guide below.
- Convert L-phenylglycine to protected amino alcohol (Compound 4) via esterification, Boc-protection, and borohydride reduction.
- Oxidize Compound 4 using IBX, react with acetone cyanohydrin, and perform cyclization with triphosgene to separate diastereomers 9 and 10.
- Deprotect compounds 9 and 10 using cesium carbonate in methanol to yield the final chiral intermediates 11 and 12 with >98% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this L-phenylglycine-based route represents a strategic opportunity to optimize the cost structure and resilience of the Docetaxel supply chain. By shifting away from precious metal catalysts and resolution-heavy processes, manufacturers can achieve substantial cost savings while mitigating supply risks associated with specialized reagents. The following points detail how this technology translates into tangible business value:
- Cost Reduction in Manufacturing: The elimination of expensive osmium catalysts and chiral ligands removes a significant variable cost component from the production budget. Additionally, the high overall yield and the ability to utilize both diastereomers (by converting them into the respective active intermediates) mean that raw material utilization is maximized. The use of commodity chemicals like L-phenylglycine, thionyl chloride, and cesium carbonate ensures that the bill of materials remains stable and predictable, shielding the project from the volatility often seen in the market for fine chemical catalysts.
- Enhanced Supply Chain Reliability: Sourcing constraints are a major concern for API production, particularly when relying on single-source suppliers for complex chiral auxiliaries. This process relies on L-phenylglycine, a bulk chemical available from multiple global vendors, thereby diversifying the supply base and reducing lead times. The robustness of the chemistry, which tolerates standard industrial solvents and equipment, further ensures that production schedules can be maintained without the delays often caused by sensitive catalytic systems requiring inert atmospheres or ultra-low temperatures.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly superior to traditional methods. By avoiding heavy metals, the burden on wastewater treatment facilities is significantly reduced, lowering the cost of environmental compliance and waste disposal. The simplified purification process, which relies on standard chromatography rather than complex crystallization or distillation sequences, facilitates easier scale-up from kilogram to tonne quantities. This scalability ensures that the technology can support commercial volume demands without requiring massive capital investment in specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical teams evaluating this technology for potential licensing or adoption.
Q: What are the primary advantages of this L-phenylglycine route over Sharpless asymmetric dihydroxylation?
A: This route eliminates the need for expensive and toxic osmium catalysts (K2OsO2(OH)4) and complex ligands required in Sharpless reactions. It utilizes cheap, readily available L-phenylglycine as a chiral pool starting material, significantly reducing raw material costs and simplifying heavy metal removal processes.
Q: How does the process ensure high stereoselectivity without resolution losses?
A: Unlike traditional resolution methods that theoretically cap yield at 50%, this synthesis leverages the inherent chirality of L-phenylglycine. The subsequent cyclization step forms rigid oxazolidinone rings (Compounds 9 and 10), which lock the stereochemistry and allow for easy chromatographic separation of diastereomers, achieving an ee value greater than 98%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly highlights the use of mild reaction conditions, non-toxic reagents, and simple purification methods like flash column chromatography. The avoidance of cryogenic temperatures and hazardous heavy metals makes the process highly scalable and environmentally compliant for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Docetaxel Side Chain Intermediate Supplier
The technological breakthroughs encapsulated in patent CN113636956A highlight the immense potential for optimizing the production of high-value oncology intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust industrial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench to plant is seamless. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Docetaxel side chain intermediate meets the highest global standards for safety and efficacy.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain for this critical cancer therapeutic is both secure and economically optimized for the future.
