Advanced Semi-Synthesis of Diosmin via Iodine Oxidation for Commercial Scale-up
Advanced Semi-Synthesis of Diosmin via Iodine Oxidation for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways for the production of high-value flavonoid compounds, which serve as critical active pharmaceutical ingredients (APIs) and nutraceuticals. Patent CN103772336A introduces a groundbreaking semi-synthesis method for phenolic hydroxyl flavonoid compounds, specifically addressing the long-standing challenges associated with the dehydrogenation of flavanones such as Hesperidin to produce Diosmin. This technology represents a significant leap forward in process chemistry, offering a universal method that drastically reduces the consumption of organic solvents like pyridine while simplifying the post-reaction treatment process. By leveraging a novel iodine recycling mechanism, this approach not only enhances the economic viability of the synthesis but also aligns with stringent environmental compliance standards required by modern regulatory bodies. For R&D Directors and Procurement Managers alike, understanding the nuances of this patent is essential for evaluating potential supply chain optimizations and cost reduction strategies in flavonoid manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for the oxidative dehydrogenation of flavanones often rely heavily on bromine oxidation or high-temperature iodine oxidation using dimethyl sulfoxide (DMSO) as a solvent. In bromine oxidation processes, the presence of phenolic hydroxyl groups necessitates complex protection and deprotection steps, such as methylation or acetylation, which significantly increase the number of synthetic steps and reduce the overall yield. Furthermore, the use of DMSO in high-temperature iodine oxidation presents severe drawbacks; DMSO acts as both a solvent and an oxidant, but its high boiling point makes recovery difficult and energy-intensive, often leading to its disposal as waste. Additionally, the coplanarity of flavonoid compounds often leads to strong intermolecular stacking, causing poor solubility in common organic solvents and requiring excessive amounts of solvent to maintain a homogeneous reaction mixture. These factors collectively contribute to high production costs, significant environmental pollution, and operational complexities that hinder commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The novel approach disclosed in the patent overcomes these limitations by utilizing a specialized solvent system and a unique complexing strategy. Instead of relying solely on high-boiling solvents, the method employs a mixture of pyridine derivatives with lower-boiling alcohols such as ethylene glycol or glycerol, which facilitates easier solvent recovery and reduces energy consumption. A key innovation is the use of trivalent aluminum compounds to form soluble complexes with the flavanone substrate, particularly for glycosides like Hesperidin that have poor solubility. This complexation prevents premature precipitation of the product during the reaction, ensuring a more uniform reaction environment and higher conversion rates. The dehydrogenation is achieved using iodine as a mild dehydrogenating agent, which is subsequently recycled from the acid filtrate using hydrogen peroxide, creating a closed-loop system that minimizes waste. 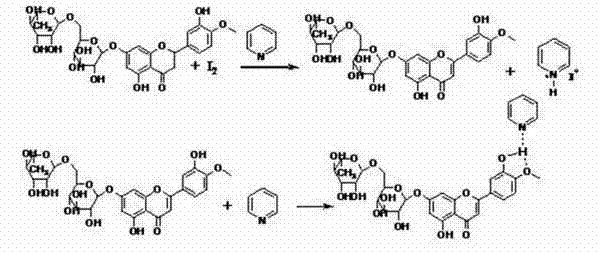
Mechanistic Insights into Iodine-Catalyzed Dehydrogenation and Aluminum Complexing
The core of this technology lies in the precise control of solubility and reaction kinetics through aluminum complexing. In the conventional iodine-pyridine method, the formation of pyridinium salts and pi-pi complexing between the pyridine ring and the flavonoid structure often traps the product, making it difficult to separate and reducing the recovery rate of pyridine. The new method introduces trivalent aluminum ions, such as those from aluminum chloride, which coordinate with the phenolic hydroxyl groups of the flavanone. This coordination significantly increases the solubility of the substrate and the intermediate dehydrogenation products in the alcohol solvent system, preventing the crystallization that typically halts the reaction in conventional processes. The reaction proceeds smoothly to form the flavonoid-aluminum complex, which remains in solution until the reaction is complete.
Upon completion, the addition of a dealumination agent, preferably phosphoric acid, rapidly decomposes the aluminum complex. This step releases the free flavonoid compound, which, being insoluble in the acidic aqueous medium, precipitates out of the solution in high purity. This mechanism effectively bypasses the need for extensive chromatographic purification or recrystallization steps that are common in older methods. Furthermore, the iodine generated as hydrogen iodide during the dehydrogenation is retained in the acid filtrate. By adding hydrogen peroxide to this filtrate, the iodide ions are oxidized back to elemental iodine, which precipitates and can be filtered and reused. 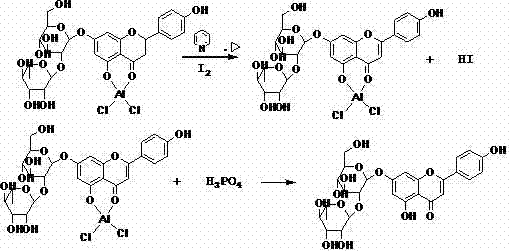
How to Synthesize Diosmin Efficiently
The synthesis of Diosmin from Hesperidin using this patented method involves a carefully controlled sequence of mixing, heating, and precipitation steps that are designed for industrial reproducibility. The process begins with the formation of the aluminum complex in a pyridine-alcohol solvent system, followed by the addition of iodine and heating to facilitate dehydrogenation. The reaction progress is monitored using TLC or HPLC to ensure complete conversion before the addition of the acid for precipitation. The detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining phenolic hydroxyl flavanone, iodine, and a pyridine derivative in a glycol or glycerol solvent system.
- For glycosides like Hesperidin, introduce a trivalent aluminum compound to form a soluble complex, preventing premature precipitation.
- Terminate the reaction with phosphoric acid to decompose the aluminum complex, precipitate the product, and recycle iodine from the filtrate using hydrogen peroxide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers substantial strategic advantages beyond mere technical feasibility. The reduction in solvent usage and the ability to recycle expensive reagents like iodine directly translate to a lower cost of goods sold (COGS), making the final product more competitive in the global market. The simplified work-up procedure, which eliminates the need for complex protection-deprotection sequences and extensive solvent exchanges, significantly reduces the manufacturing cycle time. This efficiency gain allows for faster response to market demand fluctuations and reduces the inventory holding costs associated with long production runs. Moreover, the environmental benefits of reduced waste discharge and lower energy consumption for solvent recovery align with the sustainability goals of major pharmaceutical buyers, enhancing the supplier's qualification status in vendor audits.
- Cost Reduction in Manufacturing: The elimination of high-boiling solvents like DMSO and the drastic reduction in pyridine consumption remove the need for expensive distillation and waste treatment processes. By recycling iodine directly from the reaction filtrate, the raw material cost for the dehydrogenating agent is minimized, leading to significant cost savings in API manufacturing without compromising on quality. The simplified process flow also reduces labor costs and equipment wear, contributing to a more lean and efficient production model.
- Enhanced Supply Chain Reliability: The use of readily available and stable reagents such as aluminum chloride and phosphoric acid ensures that the supply chain is not vulnerable to the volatility of specialized catalyst markets. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent ratios, means that production can be maintained consistently even with minor variations in raw material quality. This reliability is crucial for reducing lead time for high-purity APIs and ensuring continuous supply to downstream formulation partners.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing solvents that are easier to recover and recycle on a large scale. The closed-loop iodine recycling system prevents the release of halogenated waste into the environment, ensuring compliance with strict environmental regulations. The high purity of the precipitated product reduces the need for energy-intensive purification steps, further lowering the carbon footprint of the manufacturing process and facilitating the commercial scale-up of complex intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this semi-synthesis method. These answers are derived directly from the patent specifications and are intended to provide clarity on the operational benefits and chemical principles underlying the technology. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: How does the aluminum complexing method improve yield compared to traditional pyridine methods?
A: The aluminum complexing method significantly enhances solubility of the reaction intermediates in alcohol solvents, preventing premature crystallization and allowing for more complete dehydrogenation, which leads to higher overall yields.
Q: Is the iodine recycling process viable for large-scale industrial production?
A: Yes, the patent describes a robust recycling method where iodide ions in the acid filtrate are oxidized back to elemental iodine using hydrogen peroxide, allowing for high recovery rates and reduced raw material costs.
Q: What are the environmental benefits of this semi-synthesis route?
A: This route minimizes the usage of high-boiling organic solvents like DMSO and reduces pyridine consumption, while the closed-loop iodine recycling prevents heavy halogen waste discharge.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diosmin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively analyzed the technology disclosed in CN103772336A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with rigorous QC labs and stringent purity specifications to ensure that every batch of Diosmin or related flavonoid intermediate meets the highest international standards. Our commitment to process innovation allows us to offer cost-effective solutions that do not compromise on the quality or safety of the final product.
We invite you to collaborate with us to optimize your supply chain for flavonoid intermediates. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you achieve greater efficiency and reliability in your manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
