Advanced Synthesis of 3α-Hydroxy-2β-Amino-5α-Androst-17-One Compounds for Commercial Scale-Up
Advanced Synthesis of 3α-Hydroxy-2β-Amino-5α-Androst-17-One Compounds for Commercial Scale-Up
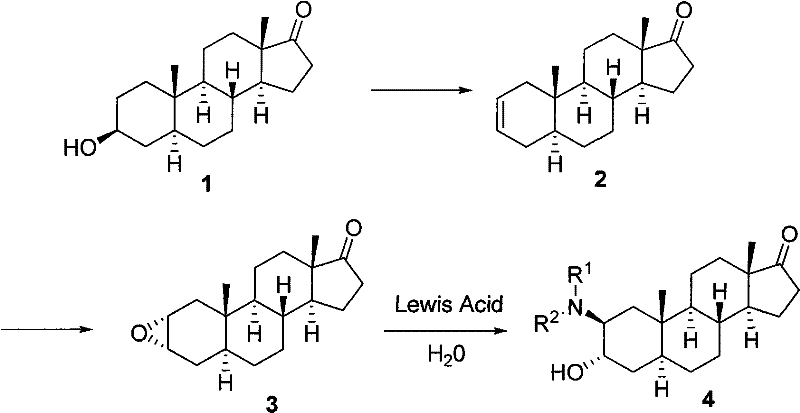
The pharmaceutical industry continuously seeks robust synthetic routes for bioactive steroid derivatives, particularly those exhibiting potent anti-leukemic and anesthetic properties. Patent CN101463063B introduces a groundbreaking methodology for synthesizing 3α-hydroxy-2β-amino-5α-androst-17-one compounds, addressing critical bottlenecks in traditional steroidal chemistry. This innovation leverages a unique water-soluble Lewis acid catalytic system that operates under remarkably mild conditions, contrasting sharply with the harsh environments typically required for epoxide aminolysis. By utilizing water as both a solvent and a reaction medium, this process not only enhances reaction kinetics through hydrophobic effects but also aligns with modern green chemistry principles by minimizing organic solvent waste. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more sustainable and economically viable manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of steroidal β-amino alcohols via epoxide aminolysis has been plagued by significant inefficiencies and operational hazards. Traditional protocols often necessitate the use of vast excesses of amine nucleophiles, sometimes exceeding ten molar equivalents, to drive the reaction to completion due to poor reactivity. Furthermore, these classical methods frequently require prolonged heating periods extending beyond three days, coupled with strict anhydrous conditions and inert gas protection to prevent catalyst deactivation. The reliance on expensive and scarce catalysts, such as rare earth metal triflates like gadolinium trifluoromethanesulfonate, further exacerbates production costs and complicates supply chain logistics. Additionally, the use of toxic organic solvents and the generation of substantial chemical waste pose severe environmental compliance challenges for large-scale manufacturing facilities.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a water-soluble Lewis acid catalyst system that dramatically simplifies the synthetic workflow. By employing common Lewis acids such as zinc chloride or aluminum chloride in an aqueous medium, the reaction achieves high conversion rates at moderate temperatures ranging from 60°C to 100°C. This method eliminates the need for rigorous moisture exclusion, as water actively participates in stabilizing the transition state and enhancing the electrophilicity of the epoxide ring. The versatility of this system is evident in its broad substrate scope, successfully accommodating both aliphatic and aromatic amines without the need for specialized activation strategies. Consequently, this approach offers a reliable pharmaceutical intermediate supplier pathway that significantly reduces lead times and operational complexity.
Mechanistic Insights into Lewis Acid-Water Catalyzed Epoxide Opening
The mechanistic foundation of this synthesis relies on the unique interaction between water-soluble Lewis acids and the epoxide substrate within a protic environment. When Lewis acids like ZnCl2 dissolve in water, they generate high concentrations of hydrated metal ions which exhibit strong affinity for the oxygen atom of the epoxide ring. This coordination significantly lowers the energy barrier for nucleophilic attack by polarizing the carbon-oxygen bonds, thereby facilitating ring opening even with weaker nucleophiles such as aromatic amines. The hydrophobic effect in the aqueous medium further drives the reaction forward by forcing organic reactants into closer proximity, effectively increasing their local concentration and reaction probability despite their limited solubility. This synergistic effect ensures that the reaction proceeds smoothly to yield a single stereoisomer with high fidelity.
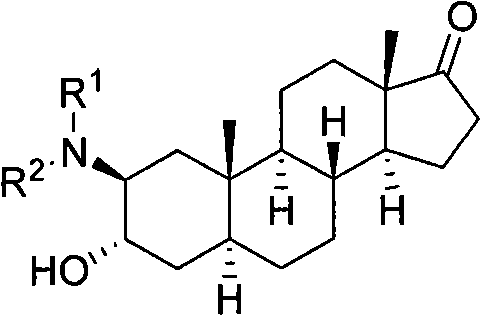
From an impurity control perspective, the regioselectivity and stereospecificity of this reaction are paramount for ensuring product purity. The nucleophilic attack occurs preferentially at the C2 position of the 2,3α-epoxide ring via a trans-diaxial opening mechanism, resulting in the exclusive formation of the 3α-hydroxy-2β-amino configuration. This high degree of stereocontrol minimizes the formation of regioisomeric byproducts that are notoriously difficult to separate in steroidal chemistry. Furthermore, the mild reaction conditions prevent the degradation of sensitive functional groups elsewhere on the steroid skeleton, such as the C17 ketone, which might otherwise undergo unwanted side reactions under harsher acidic or basic conditions. This inherent selectivity translates directly into simplified downstream purification processes and higher overall yields.
How to Synthesize 3α-Hydroxy-2β-Amino-5α-Androst-17-One Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the Lewis acid and the thermal profile of the reaction mixture to maximize yield. The process begins with the preparation of the 2,3α-epoxy-5α-androst-17-one precursor, which is subsequently mixed with the chosen amine nucleophile before introduction to the aqueous catalyst phase. Maintaining the reaction temperature within the specified 60°C to 100°C window is critical for balancing reaction rate against potential thermal decomposition. Detailed standardized synthetic steps for executing this transformation are provided in the guide below, ensuring reproducibility across different laboratory and production scales.
- Mix 2,3α-epoxy-5α-androst-17-one with the desired amine nucleophile at room temperature.
- Add the mixture to an aqueous solution of a water-soluble Lewis acid such as ZnCl2 or AlCl3.
- Heat the reaction mixture to 60-100°C for 4-36 hours, then concentrate and purify via extraction or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Lewis acid-water catalytic system presents compelling economic and logistical benefits. The substitution of expensive rare earth catalysts with commodity chemicals like zinc chloride results in substantial cost savings on raw material expenditures. Moreover, the elimination of strict anhydrous requirements reduces the capital investment needed for specialized drying equipment and inert atmosphere reactors, thereby lowering the barrier to entry for contract manufacturing organizations. The use of water as a primary solvent also drastically simplifies waste treatment protocols, aligning with increasingly stringent environmental regulations and reducing the total cost of ownership for the manufacturing process.
- Cost Reduction in Manufacturing: The replacement of high-cost catalytic systems with inexpensive, water-soluble Lewis acids directly impacts the bottom line by reducing material costs. Additionally, the shortened reaction times compared to traditional multi-day protocols increase reactor throughput, allowing for more batches to be produced within the same timeframe. The simplified workup procedure, which often requires only extraction and concentration rather than complex chromatographic separations for liquid amines, further reduces labor and solvent consumption expenses. These factors collectively contribute to a more competitive pricing structure for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Utilizing widely available and stable reagents such as zinc chloride and water mitigates the risk of supply disruptions associated with specialty catalysts. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental humidity, ensuring consistent batch-to-batch performance. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream API manufacturers. Furthermore, the broad applicability of the method allows for rapid switching between different amine derivatives without significant process re-optimization.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium inherently improves safety profiles by eliminating flammable organic solvents from the primary reaction step, facilitating safer scale-up to industrial volumes. The reduced generation of hazardous waste streams simplifies compliance with environmental discharge standards and lowers waste disposal costs. The mild thermal conditions also reduce energy consumption for heating and cooling, contributing to a lower carbon footprint for the manufacturing operation. These sustainability advantages are increasingly valued by global pharmaceutical partners seeking green supply chain solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. Understanding these nuances is essential for R&D teams planning to integrate this technology into their existing pipelines. The answers are derived directly from the experimental data and theoretical framework provided in the patent documentation, ensuring accuracy and relevance for practical application.
Q: What are the advantages of using water-soluble Lewis acids over traditional catalysts?
A: Water-soluble Lewis acids like ZnCl2 eliminate the need for expensive rare earth metals and strict anhydrous conditions, significantly reducing operational costs and environmental impact while maintaining high yields.
Q: Does this method support aromatic amines which are typically less reactive?
A: Yes, the patented Lewis acid/water system effectively activates the epoxide ring, allowing even low-reactivity aromatic amines to participate in the nucleophilic attack with high conversion rates.
Q: How is the stereochemistry controlled during the ring-opening reaction?
A: The reaction proceeds via a trans-diaxial opening mechanism, ensuring high stereoselectivity to produce the specific 3α-hydroxy-2β-amino configuration required for biological activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3α-Hydroxy-2β-Amino-5α-Androst-17-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Lewis acid-catalyzed synthesis for producing high-purity steroid intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop to plant. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for clinical and commercial applications. We are committed to delivering consistent quality and reliability for complex pharmaceutical intermediates.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis can optimize your supply chain. Request a Customized Cost-Saving Analysis today to understand the specific economic benefits for your project. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments tailored to your unique requirements. Let us help you accelerate your development timeline with our proven manufacturing capabilities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
