Advanced Synthesis of Cyclohexanealkanoic Acids for Bronchial Asthma Treatment Intermediates
Advanced Synthesis of Cyclohexanealkanoic Acids for Bronchial Asthma Treatment Intermediates
The pharmaceutical landscape for respiratory diseases continues to evolve, driven by the need for more potent and selective leukotriene antagonists. Patent CN86105165A introduces a sophisticated class of 2-hydroxy-4-tetradecylenecyclohexanealkanoic acids that serve as critical intermediates in the development of next-generation bronchial asthma therapeutics. These compounds, characterized by a unique mercaptoalkanoic acid substituent at the 3-position, exhibit significant antagonistic activity against Slow Reacting Substance of Anaphylaxis (SRS-A), specifically targeting Leukotriene D4 (LTD4). For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, understanding the synthetic versatility and biological efficacy of these structures is paramount. The patent outlines a robust methodology that transitions from complex epoxy-ketone precursors to biologically active acids through a series of highly controlled organic transformations, ensuring high purity and structural integrity essential for downstream drug formulation.
![Chemical structure of 3-[(carboxymethyl)thio]-2-hydroxy-4-tetradecylenecyclohexaneacetic acid example](/insights/img/cyclohexanealkanoic-acid-synthesis-pharma-supplier-20260308201536-04.webp)
This technical insight report delves deep into the mechanistic nuances and commercial viability of the processes described in CN86105165A. By leveraging advanced oxidation techniques like Swern oxidation and precise stereoselective reductions, manufacturers can achieve consistent quality in producing these high-value fine chemicals. The ability to modulate the alkyl chain length and the nature of the carboxylic acid side chain offers a versatile platform for structure-activity relationship (SAR) studies, allowing medicinal chemists to optimize potency and pharmacokinetic profiles. Furthermore, the synthetic routes described avoid the use of prohibited heavy metal catalysts in the final coupling steps, aligning with modern green chemistry initiatives and simplifying the regulatory approval pathway for new drug applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex cyclohexane derivatives with multiple chiral centers and specific side-chain functionalities has been plagued by low overall yields and cumbersome purification protocols. Traditional approaches often relied on non-selective alkylation reactions that produced difficult-to-separate mixtures of regioisomers and stereoisomers, drastically increasing the cost of goods sold (COGS). Moreover, older methodologies frequently utilized harsh acidic or basic conditions that could compromise sensitive functional groups, such as esters or unprotected hydroxyls, leading to degradation products that complicate the impurity profile. The lack of stereocontrol in early synthetic attempts meant that extensive chromatographic separation was required to isolate the biologically active enantiomer, resulting in significant material loss and extended production timelines. Additionally, the introduction of long-chain alkylidene groups at the 4-position was often achieved through inefficient coupling reactions that required expensive reagents and generated substantial chemical waste, posing environmental compliance challenges for large-scale manufacturing facilities.
The Novel Approach
The methodology presented in the patent data revolutionizes this synthetic challenge by employing a convergent strategy centered on the nucleophilic opening of a pre-formed epoxide ring. This approach allows for the precise installation of the mercaptoalkanoic acid moiety under mild conditions, typically utilizing tertiary amines like triethylamine as catalysts in appropriate alcohol solvents. By starting with well-defined 2,3-epoxy-4-alkylenecyclohexanealkanoates, the process ensures that the relative stereochemistry between the hydroxyl group and the sulfur substituent is strictly controlled, predominantly yielding the desired trans-configuration which is critical for biological activity. The use of Swern oxidation to generate the necessary ketone intermediates from alcohol precursors provides a high-yielding, metal-free alternative to chromium-based oxidations, thereby reducing toxic waste streams. Furthermore, the flexibility to convert the resulting esters into free acids or amides via standard hydrolysis or aminolysis post-reaction allows for the rapid generation of diverse compound libraries for screening purposes without altering the core synthetic backbone.
Mechanistic Insights into Epoxide Ring-Opening and Functionalization
The core chemical transformation driving the synthesis of these asthma intermediates is the regioselective and stereoselective ring-opening of the epoxide functionality by a mercaptoalkanoate nucleophile. As illustrated in the reaction scheme below, the process begins with a 2,3-epoxy-4-alkylenecyclohexanealkanoate substrate, where the epoxide ring is activated for nucleophilic attack. In the presence of a tertiary amine base, the thiol group of the mercaptoalkanoate is deprotonated to form a thiolate anion, which is a potent soft nucleophile. This thiolate attacks the less hindered carbon of the epoxide ring (typically the C-3 position), leading to ring cleavage and the formation of a new carbon-sulfur bond. The reaction proceeds with inversion of configuration at the attack site, establishing the specific stereochemical relationship required for LTD4 antagonism. The choice of solvent is critical; lower alkanols corresponding to the ester moiety (e.g., methanol for methyl esters) are preferred to prevent transesterification side reactions, ensuring the integrity of the ester protecting group until the final hydrolysis step.
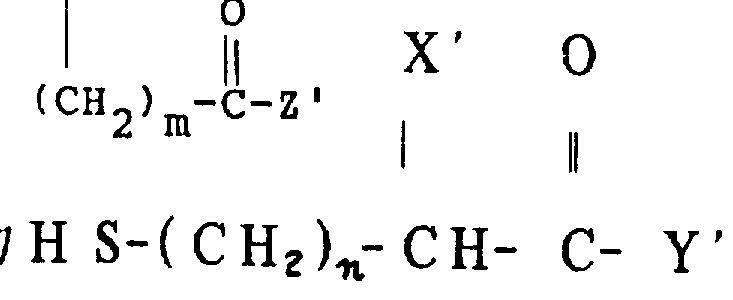
Beyond the coupling reaction, the preparation of the epoxide precursor itself involves a sequence of sophisticated organic transformations designed to maximize stereochemical purity. The patent details the use of Swern oxidation conditions, involving oxalyl chloride and dimethyl sulfoxide (DMSO) at cryogenic temperatures (-65°C to -55°C), to convert secondary alcohols into ketones without affecting the sensitive epoxide ring. This ketone intermediate is then subjected to a Wittig reaction using long-chain phosphonium salts, such as bromotetradecylphosphine, to install the tetradecenyl side chain with high E/Z selectivity. Following the olefination, the ketone can be reduced back to an alcohol using sodium borohydride, often in the presence of cerium chloride to control 1,2-reduction selectivity over 1,4-reduction. This redox manipulation allows chemists to invert the stereochemistry at the C-2 position if the cis-isomer is desired, although the trans-isomer is generally the primary target for asthma therapy. The entire sequence demonstrates a high level of chemoselectivity, preserving the ester and epoxide functionalities while modifying the carbon skeleton.
How to Synthesize 2-Hydroxy-4-Tetradecylenecyclohexanealkanoic Acids Efficiently
The synthesis of these high-purity pharmaceutical intermediates requires strict adherence to the reaction parameters outlined in the patent to ensure reproducibility and safety. The process generally initiates with the preparation of the epoxy-ketone intermediate, followed by Wittig olefination to introduce the lipid tail, and concludes with the thiol-epoxide coupling and hydrolysis. Operators must maintain rigorous temperature control during the Swern oxidation steps to prevent exothermic runaways and ensure the complete conversion of the alcohol to the ketone. The subsequent nucleophilic substitution with mercaptoalkanoates should be monitored via TLC or HPLC to determine the optimal reaction endpoint, typically ranging from 16 to 20 hours at room temperature. For detailed operational procedures, safety data sheets, and specific stoichiometric ratios required for GMP manufacturing, please refer to the standardized synthesis guide provided below.
- Preparation of the epoxy-ketone intermediate via Swern oxidation of hydroxy-epoxides derived from Birch reduction or silyl-protection routes.
- Execution of a Wittig reaction using bromotetradecylphosphine to introduce the long-chain alkylidene group at the 4-position.
- Nucleophilic ring-opening of the epoxide using mercaptoalkanoates in the presence of a tertiary amine catalyst like triethylamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthetic route described in CN86105165A offers substantial strategic advantages in terms of cost stability and raw material security. The reliance on commodity chemicals such as anisole propionic acid, dimethyl sulfoxide, and common phosphonium salts mitigates the risk of supply disruptions associated with exotic or single-source reagents. By eliminating the need for precious metal catalysts in the key coupling steps, the process significantly reduces the cost reduction in pharmaceutical intermediate manufacturing, as there is no requirement for expensive metal scavenging resins or complex wastewater treatment for heavy metal removal. The modular nature of the synthesis allows for the decoupling of production stages; the epoxy-ketone intermediate can be stockpiled and then diversified into various final analogs based on market demand, enhancing inventory flexibility and reducing lead time for high-purity pharmaceutical intermediates. Furthermore, the use of standard unit operations like extraction, distillation, and crystallization facilitates seamless technology transfer from laboratory scale to commercial production.
- Cost Reduction in Manufacturing: The synthetic pathway leverages widely available starting materials and avoids the use of costly chiral catalysts or enzymes, relying instead on substrate-controlled stereochemistry and classical resolution methods where necessary. This fundamental simplification of the reagent profile translates directly into lower variable costs per kilogram of finished product. Additionally, the high atom economy of the Wittig reaction and the epoxide opening step minimizes waste generation, reducing disposal costs and improving the overall environmental footprint of the manufacturing process. The ability to perform reactions in common solvents like dichloromethane, ether, and alcohols further optimizes solvent recovery and recycling loops, contributing to a more sustainable and economically viable production model.
- Enhanced Supply Chain Reliability: The robustness of the chemical transformations described ensures consistent batch-to-batch quality, which is critical for maintaining long-term supply contracts with major pharmaceutical companies. The process tolerances are well-defined, reducing the likelihood of batch failures due to minor fluctuations in reaction conditions. By establishing a supply chain based on stable, non-perishable intermediates, manufacturers can buffer against upstream volatility in the petrochemical sector. The scalability of the route has been demonstrated through examples ranging from gram-scale laboratory preparations to multi-kilogram pilot runs, proving its viability for commercial scale-up of complex pharmaceutical intermediates without the need for specialized high-pressure or cryogenic equipment beyond standard industrial capabilities.
- Scalability and Environmental Compliance: The avoidance of chromium-based oxidants in favor of Swern oxidation, while requiring careful handling of DMSO byproducts, aligns better with modern environmental regulations regarding heavy metal discharge. The process generates primarily organic waste streams that can be incinerated for energy recovery, simplifying waste management protocols. The final hydrolysis steps utilize standard aqueous bases and acids, generating salt byproducts that are easily treated in conventional effluent treatment plants. This compliance-ready profile reduces the regulatory burden on manufacturing sites and accelerates the audit process for new vendor approvals, ensuring uninterrupted supply continuity for downstream drug product manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these cyclohexanealkanoic acid derivatives. The answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating these intermediates into their existing development pipelines.
Q: What is the primary therapeutic application of these cyclohexanealkanoic acid derivatives?
A: These compounds function as potent antagonists of Leukotriene D4 (LTD4), making them highly effective intermediates for developing treatments for bronchial asthma and other anaphylactic diseases.
Q: How is the stereochemistry controlled during the synthesis process?
A: The process utilizes specific reduction steps, such as sodium borohydride reduction of ketones, to establish the desired trans-configuration between the hydroxyl and sulfur substituents on the cyclohexane ring.
Q: Are there specific safety considerations for the oxidation steps described in the patent?
A: Yes, the Swern oxidation method employed requires low temperatures (-65°C) and generates volatile byproducts like carbon monoxide, necessitating rigorous ventilation and temperature control systems during scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclohexanealkanoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of respiratory therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and potency of every batch. Our commitment to quality extends beyond mere compliance; we actively collaborate with our clients to optimize synthetic routes for maximum efficiency and cost-effectiveness, acting as a true extension of your R&D department.
We invite you to contact our technical procurement team to discuss your specific requirements for asthma intermediate synthesis. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new analogs, we are ready to provide the support you need. Let us partner with you to accelerate the delivery of life-saving medications to patients worldwide through superior chemical manufacturing excellence.
