Advanced Semi-Synthetic Route for 7-Silylcamptothecin and Homocamptothecin Analogs
The pharmaceutical landscape for anticancer therapeutics is continuously evolving, with a specific focus on enhancing the bioavailability and stability of potent natural products. Patent CN1514841A introduces a groundbreaking semi-synthetic methodology for the production of 7-silylcamptothecin and 7-silylhomocamptothecin, collectively known as silatecans. These compounds represent a critical class of DNA topoisomerase I inhibitors, exhibiting superior lipophilicity and plasma stability compared to their parent alkaloids. The disclosed technology addresses the longstanding bottleneck in accessing these high-value pharmaceutical intermediates by shifting away from laborious total synthesis towards a streamlined functionalization of naturally abundant camptothecin. This strategic pivot not only simplifies the molecular construction but also opens new avenues for cost reduction in pharmaceutical intermediate manufacturing, making these promising anticancer candidates more viable for large-scale clinical development and commercial deployment.
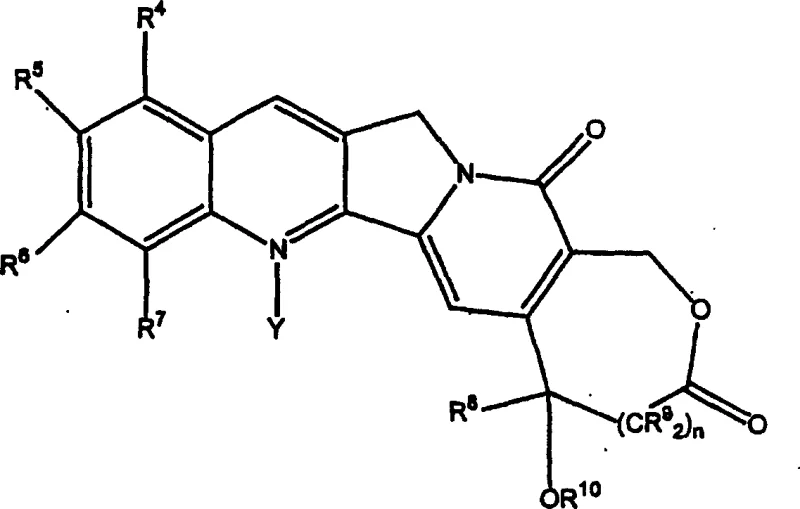
The structural complexity of the camptothecin core, characterized by its fused pentacyclic ring system, has historically posed significant challenges for synthetic chemists. Traditional approaches often struggled with the delicate balance of introducing lipophilic silyl groups without compromising the integrity of the sensitive lactone E-ring. The innovation detailed in this patent leverages the inherent reactivity of the quinoline moiety within the camptothecin scaffold, allowing for direct C-H functionalization. By targeting the C7 position, the process generates analogs with enhanced membrane permeability and reduced protein binding, properties that are essential for high-purity API intermediates intended for oncology applications. This method stands as a testament to the power of modern radical chemistry in solving classical problems in natural product modification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the primary route to access silatecans and homosilatecans involved complex cascade radical annulation strategies, often requiring thirteen or more synthetic steps from simple starting materials. These total synthesis pathways, while intellectually elegant, suffered from abysmal overall yields, typically hovering around 2 percent, which rendered them economically unfeasible for commercial scale-up. The extensive step count inherently multiplied the risk of impurity accumulation, necessitating rigorous and costly purification protocols at each stage to meet the stringent quality standards required for commercial scale-up of complex pharmaceutical intermediates. Furthermore, the reliance on specialized reagents and the generation of significant chemical waste associated with long linear sequences created substantial environmental and logistical burdens, limiting the ability of supply chains to respond flexibly to market demand for these critical anticancer agents.
The Novel Approach
In stark contrast, the novel semi-synthetic approach described in the patent capitalizes on the availability of natural camptothecin, effectively truncating the synthetic timeline to a single, highly efficient functionalization step. By employing a radical silylation strategy, the process directly installs the requisite silyl group onto the pre-formed pentacyclic core, bypassing the need to construct the rings de novo. This drastic simplification translates to a massive improvement in material throughput and a significant reduction in the consumption of solvents and reagents. The ability to recover a substantial portion of unreacted starting material further enhances the atom economy of the process, providing a robust foundation for reducing lead time for high-purity pharmaceutical intermediates. This methodology transforms the production of silatecans from a boutique laboratory exercise into a viable industrial process capable of supporting clinical and commercial needs.
Mechanistic Insights into Radical Silylation Chemistry
The core of this technological breakthrough lies in the generation and controlled application of silyl radicals under mild thermal conditions. The reaction mechanism initiates with the homolytic cleavage of a peroxide initiator, such as di-tert-butyl peroxide, which generates reactive alkoxy radicals. These radicals subsequently abstract a hydrogen atom from a silane precursor, facilitated by the presence of a thiol promoter, to generate the active silyl radical species. This silyl radical then adds competitively to the electron-deficient quinoline ring system of the camptothecin substrate. The presence of the thiol is crucial, as it acts as a polarity reversal catalyst, accelerating the hydrogen abstraction step and ensuring a steady flux of reactive silyl species without inducing excessive decomposition of the sensitive camptothecin backbone.
Regioselectivity in this radical addition is a function of thermodynamic and kinetic control governed by reaction temperature. At lower temperatures, approximately 105 degrees Celsius, the reaction favors addition at the C7 position, yielding the desired 7-silylcamptothecin isomer which possesses the optimal biological profile. Conversely, elevating the temperature to 160 degrees Celsius shifts the selectivity towards the C12 position, demonstrating the fine tunability of the process parameters. Following the radical addition, an oxidative rearomatization step restores the aromaticity of the quinoline ring, locking the silyl group in place. This mechanistic understanding allows for precise optimization of reaction conditions to maximize the yield of the target isomer while minimizing the formation of regioisomeric by-products, ensuring a clean impurity profile suitable for downstream pharmaceutical processing.
How to Synthesize 7-Silylcamptothecin Efficiently
The practical execution of this synthesis involves dissolving the camptothecin starting material in a high-boiling ether solvent such as p-dioxane, which provides excellent solubility for the polar alkaloid substrate. To this solution, a slight excess of the chosen silane, such as tert-butyldimethylsilane, is added along with a catalytic amount of a thiol promoter like triisopropylsilanethiol. The reaction is initiated by the addition of a peroxide oxidant, and the mixture is maintained under an inert atmosphere at reflux temperatures for a period ranging from one to two days. This standardized protocol ensures consistent generation of the silyl radical and its subsequent capture by the substrate, providing a reliable method for producing multi-kilogram quantities of the intermediate. The detailed standardized synthesis steps are outlined in the guide below.
- Mix camptothecin or homocamptothecin with a silyl precursor (e.g., silane) and a silyl generating agent in a suitable solvent like p-dioxane.
- Add a thiol promoter (e.g., triisopropylsilanethiol) and a peroxide initiator (e.g., di-tert-butyl peroxide) to the reaction mixture.
- Heat the mixture under reflux conditions (approx. 105°C) for 1 to 2 days to facilitate radical silylation at the C7 position.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the transition from total synthesis to this semi-synthetic route offers profound strategic benefits that extend beyond simple unit cost metrics. The reliance on natural camptothecin as a feedstock leverages an established agricultural supply chain, mitigating the risks associated with sourcing complex synthetic building blocks that may be subject to geopolitical or manufacturing disruptions. The drastic reduction in synthetic steps inherently lowers the working capital tied up in inventory and reduces the facility time required for production, thereby increasing the overall agility of the supply network. This efficiency gain is critical for maintaining continuity of supply for life-saving oncology medications, where interruptions can have severe consequences for patient care and clinical trial timelines.
- Cost Reduction in Manufacturing: The elimination of over ten synthetic steps fundamentally alters the cost structure of producing silatecan intermediates. By removing the need for multiple protection-deprotection sequences and intermediate isolations, the process significantly reduces the consumption of expensive reagents, chromatography media, and energy-intensive purification operations. The ability to recover and recycle unreacted camptothecin further drives down the effective cost of goods sold, creating a sustainable economic model that can withstand market fluctuations in raw material pricing. This structural cost advantage allows for more competitive pricing strategies without compromising on the quality or purity of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Utilizing a semi-synthetic approach anchored on a natural product simplifies the bill of materials, reducing the number of critical vendors and potential points of failure in the supply chain. The robustness of the radical silylation chemistry means that the process is less sensitive to minor variations in reagent quality compared to multi-step organometallic cascades, leading to higher batch-to-batch consistency. This reliability is paramount for securing long-term supply agreements with major pharmaceutical partners who require guaranteed delivery schedules to support their global clinical and commercial launch plans for new cancer therapies.
- Scalability and Environmental Compliance: The simplified reaction profile facilitates easier scale-up from laboratory to pilot and commercial manufacturing scales, as there are fewer unit operations to optimize and validate. The use of standard organic solvents and the absence of heavy metal catalysts commonly found in cross-coupling reactions simplify waste stream management and reduce the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only lowers disposal costs but also ensures compliance with increasingly stringent environmental regulations, safeguarding the manufacturing license to operate in key global markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silylation technology. These insights are derived directly from the experimental data and process descriptions within the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project requirements. Understanding these nuances is essential for R&D teams planning process validation and for procurement teams assessing vendor capabilities.
Q: What is the primary advantage of this semi-synthetic route over total synthesis?
A: The semi-synthetic route significantly reduces the number of steps from over thirteen to just a few key transformations, utilizing abundant natural camptothecin as a starting material, which drastically improves overall yield and cost-efficiency.
Q: How is regioselectivity controlled between C7 and C12 positions?
A: Regioselectivity is primarily controlled by reaction temperature; lower temperatures (around 105°C) favor the desired 7-silyl isomer, while higher temperatures shift the preference towards the 12-silyl isomer.
Q: Can this method be used to synthesize 10-hydroxy derivatives like DB-67?
A: Yes, the 7-silylcamptothecin produced can be subsequently converted to 10-hydroxy-7-silylcamptothecin (DB-67) through a two-step process involving N-oxidation followed by photolysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Silylcamptothecin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this semi-synthetic technology in advancing the development of next-generation anticancer drugs. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate this patented chemistry into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in early-stage discovery or late-stage commercialization. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 7-silylcamptothecin meets the highest international standards for safety and efficacy.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this semi-synthetic supply chain. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can accelerate your drug development timeline and enhance your market competitiveness.
