Advanced Synthetic Route for 9-Aminocamptothecin: Transforming Waste into High-Value API Intermediates
The pharmaceutical industry constantly seeks more efficient pathways for producing potent anticancer agents, and the synthesis of 9-aminocamptothecin stands as a prime example of process innovation. As detailed in patent CN1113384A, a groundbreaking methodology has been established that fundamentally alters the economic and environmental landscape of producing this critical topoisomerase I inhibitor. Traditionally, the semi-synthesis of 9-aminocamptothecin from natural camptothecin has been plagued by poor regioselectivity during the nitration step, resulting in a predominance of the undesired 12-nitro isomer. This new approach, however, ingeniously converts this problematic byproduct into the valuable starting material for the desired 9-amino derivative. By implementing a strategic sequence involving reduction, diazotization, halogenation, and selective nitration, the process achieves a closed-loop system that maximizes raw material utilization. This technical breakthrough not only addresses the long-standing challenge of isomer separation but also paves the way for a more sustainable and cost-effective supply chain for oncology drug manufacturers globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 9-aminocamptothecin has relied on the direct nitration of natural camptothecin, a process fraught with significant inefficiencies that hinder large-scale commercial viability. The fundamental chemical limitation lies in the electrophilic substitution pattern of the camptothecin ring system, which inherently favors nitration at the 12-position over the desired 9-position. Literature and prior art indicate that this reaction typically yields a mixture containing approximately 70% of the biologically inactive 12-nitrocamptothecin and only 30% of the therapeutically active 9-nitrocamptothecin. This skewed ratio creates a massive bottleneck for procurement and supply chain managers, as nearly three-quarters of the expensive natural starting material is converted into a waste stream that requires costly disposal. Furthermore, the separation of these closely related nitro isomers is technically demanding and often results in further yield losses during purification. The accumulation of this chemically stable but biologically useless 12-nitro derivative represents not just a financial loss but a significant environmental burden, complicating waste management protocols for pharmaceutical manufacturers aiming for green chemistry standards.
The Novel Approach
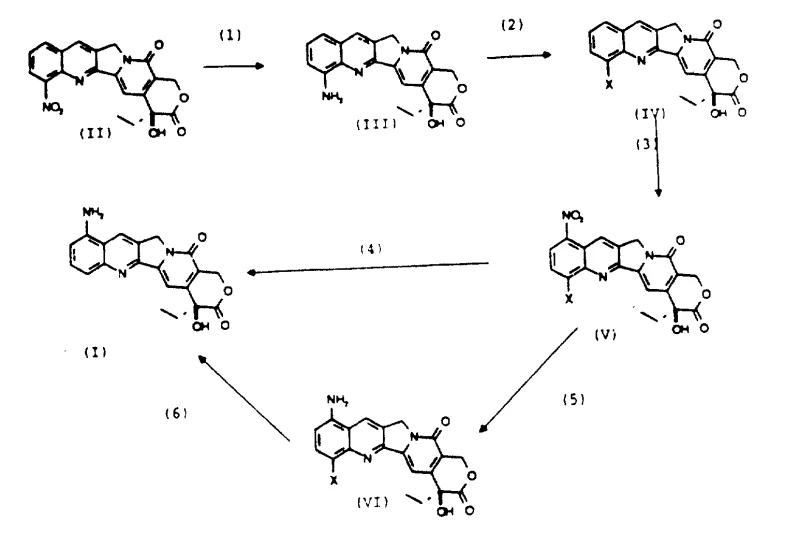
In stark contrast to the wasteful conventional routes, the novel process described in the patent data introduces a paradigm shift by treating the 12-nitro isomer not as waste, but as a valuable precursor. The core innovation involves the reductive conversion of 12-nitrocamptothecin into 12-aminocamptothecin, followed by a diazotization reaction that transforms the amino group into a halogen substituent, such as chlorine or bromine. This 12-halo intermediate serves a dual purpose: it blocks the 12-position from further reaction and acts as a directing group that facilitates highly selective nitration at the 9-position. This strategic modification ensures that subsequent nitration steps occur almost exclusively at the target site, dramatically improving the ratio of desired product to byproducts. The final stage involves a sophisticated reduction sequence that removes both the newly introduced nitro group at the 9-position and the temporary halogen blocking group at the 12-position, yielding pure 9-aminocamptothecin. This approach effectively recycles the previously discarded majority fraction of the nitration mixture, turning a liability into an asset and significantly enhancing the overall atom economy of the synthesis.
Mechanistic Insights into Diazotization and Selective Nitration
The success of this synthetic route hinges on the precise control of functional group transformations within the sensitive camptothecin scaffold, particularly the preservation of the E-ring lactone and the C20 stereocenter. The initial reduction of the 12-nitro group to an amine can be achieved using standard reducing agents like tin(II) chloride in aqueous hydrochloric acid or via catalytic hydrogenation with palladium on carbon. Following this, the critical diazotization step utilizes sodium nitrite under acidic conditions to generate a diazonium salt in situ. This unstable intermediate is immediately subjected to a Sandmeyer-type reaction with copper(I) halides, such as CuCl or CuBr, to install the halogen atom at the 12-position. This halogenation is crucial because the resulting 12-halo-camptothecin derivative exhibits distinct electronic properties that deactivate the 12-position towards electrophilic attack while activating or directing incoming electrophiles to the 9-position. When this 12-halo intermediate is subjected to nitration conditions using nitric acid or mixed acids, the reaction proceeds with high regioselectivity to form the 9-nitro-12-halo derivative. The final reductive removal of the halogen is particularly noteworthy; while reductive dehalogenation of quinolines is known, doing so on the base-sensitive camptothecin skeleton without ring opening requires careful selection of conditions. The patent highlights that the newly formed 9-amino group itself can act as a weak non-nucleophilic base, facilitating the reductive removal of the halogen in the presence of organic or inorganic non-nucleophilic bases, thereby preserving the integrity of the molecule.
Impurity control is another vital aspect of this mechanism, especially given the complexity of the polycyclic structure. The stepwise nature of the synthesis allows for the isolation and purification of key intermediates, such as the 12-amino and 12-halo derivatives, before proceeding to the next stage. This modularity prevents the carryover of impurities that could complicate the final reduction step. For instance, incomplete reduction of the 12-nitro starting material could lead to mixed nitro-amino species, but the robust conditions described, such as extended reaction times with tin(II) chloride or thorough catalytic hydrogenation, ensure complete conversion. Additionally, the selectivity of the nitration step minimizes the formation of dinitro species or nitration at other positions on the A-ring. The final hydrogenation step to remove the 12-halo group must be carefully monitored to prevent over-reduction of the quinoline nucleus or racemization at the C20 chiral center. The use of mild catalysts like palladium on carbon at atmospheric pressure and room temperature, as exemplified in the patent data, provides the necessary selectivity to reduce the nitro and carbon-halogen bonds while leaving the rest of the sensitive molecular architecture intact, ensuring a high-purity final product suitable for pharmaceutical applications.
How to Synthesize 9-Aminocamptothecin Efficiently
The synthesis of 9-aminocamptothecin via this recycling pathway offers a robust framework for laboratory and pilot-scale production, leveraging readily available reagents and standard unit operations. The process begins with the reduction of the 12-nitro precursor, followed by diazotization and halogenation to install the blocking group. Subsequent nitration targets the 9-position, and a final reduction yields the target API intermediate. While the general workflow is straightforward, precise control over reaction parameters such as temperature, pH, and stoichiometry is essential to maximize yield and purity. The detailed standardized synthesis steps, including specific solvent systems, reagent grades, and workup procedures, are outlined in the guide below to ensure reproducibility and safety during scale-up.
- Reduce 12-nitro-20(s)-camptothecin to the corresponding 12-amino derivative using tin(II) chloride or catalytic hydrogenation.
- Convert the 12-amino intermediate into a 12-halo derivative (e.g., 12-chloro or 12-bromo) via diazotization followed by a Sandmeyer-type reaction.
- Nitrate the 12-halo derivative selectively at the 9-position, followed by a final reductive removal of both the nitro and halo groups to yield 9-aminocamptothecin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers substantial strategic benefits that extend beyond simple chemical yield improvements. The primary advantage lies in the drastic reduction of raw material costs and waste disposal expenses. By utilizing the 12-nitro isomer, which is traditionally considered a waste product comprising the majority of the output from natural camptothecin nitration, manufacturers can significantly lower their dependency on expensive, high-purity natural camptothecin feedstock. This recycling capability transforms a cost center (waste disposal) into a value-generating stream, effectively doubling the utility of the initial nitration batch. Furthermore, the process relies on common industrial reagents such as tin salts, copper halides, and hydrogen gas, avoiding the need for exotic or prohibitively expensive catalysts that often plague complex alkaloid syntheses. This reliance on commodity chemicals enhances supply chain resilience, reducing the risk of bottlenecks associated with specialized reagent shortages.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of waste and the optimization of raw material usage. In traditional methods, the 70% yield of unwanted 12-nitrocamptothecin represents a direct financial loss, requiring capital for both the initial material and its subsequent disposal. By converting this byproduct into the final drug substance, the effective cost per kilogram of the active ingredient is significantly reduced. Additionally, the process avoids the use of precious metal catalysts in the early stages, opting instead for base metals like copper and iron/tin, which are far more cost-effective. The ability to perform reactions under mild conditions, such as atmospheric pressure and moderate temperatures, also reduces energy consumption and the need for specialized high-pressure reactor equipment, further contributing to lower operational expenditures and a more competitive pricing structure for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is a critical concern for pharmaceutical manufacturers, and this synthetic route enhances reliability by diversifying the source of the key intermediate. Instead of relying solely on the scarce and variable supply of naturally derived 9-nitrocamptothecin, producers can generate the necessary precursors from the abundant 12-nitro byproduct. This flexibility buffers the supply chain against fluctuations in the harvest or extraction yields of natural camptothecin. Moreover, the synthetic steps involved are robust and scalable, utilizing well-established chemical transformations like reduction and diazotization that are easily transferred from laboratory to commercial manufacturing scales. The use of stable intermediates that can be isolated and stored allows for better inventory management and production planning, ensuring that downstream drug formulation teams receive a consistent and reliable supply of high-quality 9-aminocamptothecin without interruption.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns perfectly with modern green chemistry principles and stringent waste disposal regulations. The reduction in hazardous waste generation is substantial, as the toxic 12-nitro byproduct is consumed rather than discharged. This simplifies the environmental permitting process for manufacturing facilities and reduces the long-term liability associated with chemical waste storage and treatment. The scalability of the process is supported by the use of heterogeneous catalysts like palladium on carbon, which can be easily filtered and recycled, minimizing heavy metal contamination in the final product. The mild reaction conditions also enhance safety profiles for large-scale operations, reducing the risks associated with exothermic runaways or high-pressure incidents. Consequently, this route facilitates a smoother path to commercial approval and sustainable long-term production, meeting the increasing demands for environmentally responsible pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 9-aminocamptothecin using this advanced recycling methodology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of the process for potential partners and technical stakeholders. Understanding these details is crucial for evaluating the integration of this technology into existing production lines or new project developments.
Q: How does this process solve the waste issue in traditional camptothecin nitration?
A: Traditional nitration of camptothecin produces a 70/30 mixture favoring the unwanted 12-nitro isomer. This novel process recycles that 12-nitro byproduct by converting it into a 12-halo intermediate, which directs subsequent nitration exclusively to the desired 9-position, effectively turning waste into the primary feedstock.
Q: What are the critical conditions for preserving the C20 stereochemistry?
A: The process utilizes mild reaction conditions, specifically avoiding strong bases that could racemize the sensitive C20 stereocenter or open the E-ring lactone. Reductions are performed using catalytic hydrogenation or mild chemical reducing agents like tin(II) chloride under controlled acidic or neutral conditions.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the method is designed for scalability. It eliminates the need to dispose of large quantities of biologically inactive 12-nitro derivatives, significantly reducing waste treatment costs and improving overall atom economy, which are critical factors for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Aminocamptothecin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes for high-value oncology intermediates like 9-aminocamptothecin. Our technical team has extensively analyzed the recycling process described in CN1113384A and possesses the expertise to implement this technology at a commercial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant with global regulatory standards. Our state-of-the-art facilities are equipped with rigorous QC labs capable of monitoring every step of this multi-stage synthesis, guaranteeing stringent purity specifications and consistent quality for every batch delivered. We understand that the stability of the camptothecin lactone ring is paramount, and our process controls are designed to maintain the integrity of the C20 stereocenter throughout the manufacturing cycle.
We invite pharmaceutical companies and research institutions to collaborate with us to optimize their supply chains for 9-aminocamptothecin and related derivatives. By leveraging our technical capabilities, you can achieve significant cost reductions and improve the sustainability of your API production. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of this novel recycling technology can meet your exacting standards for quality and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
