Scaling High-Capacity Organic Polymer Anodes for Next-Gen Lithium Batteries
Scaling High-Capacity Organic Polymer Anodes for Next-Gen Lithium Batteries
The global transition towards sustainable energy storage has intensified the search for alternatives to traditional transition metal oxide cathodes, which suffer from resource scarcity and environmental concerns. Patent CN110590789B introduces a breakthrough nitrogen-rich triphenylamine derivative conjugated polymer, specifically poly[2,8,14-tris(4-diphenylaminophenyl)-hexaazanaphthalene], designed to serve as a high-performance organic positive electrode material for lithium-ion batteries. This innovation addresses the critical trade-off between energy density and cycle stability by chemically fusing the high-voltage characteristics of triphenylamine with the high-capacity potential of hexaazanaphthalene. For R&D directors and procurement strategists in the energy sector, this technology represents a viable pathway toward cost-effective, environmentally friendly, and high-specific-capacity battery solutions that do not rely on cobalt or nickel supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional lithium-ion battery正极 materials, such as LiCoO2 and LiNiO2, are heavily dependent on the mining of finite transition metals, leading to volatile pricing and significant geopolitical supply chain risks. Furthermore, purely organic alternatives have historically struggled with performance deficits; for instance, polytriphenylamine exhibits excellent redox reversibility and a high discharge voltage platform around 3.6V, yet its theoretical specific capacity is limited to a mere 109 mAh/g, which is insufficient for modern high-energy applications. Conversely, small-molecule hexaazanaphthalenes offer high theoretical capacities up to 418 mAh/g but suffer from severe dissolution into the electrolyte during charge-discharge cycles, resulting in rapid capacity degradation and poor cycling stability. These inherent limitations of single-component organic systems have hindered their commercial adoption in large-scale energy storage devices.
The Novel Approach
The patented methodology overcomes these deficiencies through a sophisticated molecular design that covalently links triphenylamine units to a central hexaazanaphthalene core, creating a conjugated system that synergizes the benefits of both moieties. The synthesis begins with a robust Suzuki coupling reaction to construct the precursor framework, ensuring high purity and structural integrity before the final polymerization step. 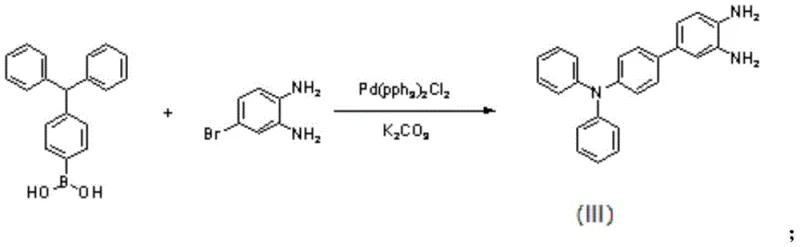 As illustrated in the reaction scheme, the coupling of 4-triphenylamine borate with 4-bromo-o-phenylenediamine under palladium catalysis establishes the necessary carbon-nitrogen backbone with high efficiency. This structural integration prevents the active material from dissolving while simultaneously elevating the theoretical specific capacity to 216 mAh/g, nearly double that of pure polytriphenylamine, thereby offering a compelling value proposition for next-generation battery manufacturing.
As illustrated in the reaction scheme, the coupling of 4-triphenylamine borate with 4-bromo-o-phenylenediamine under palladium catalysis establishes the necessary carbon-nitrogen backbone with high efficiency. This structural integration prevents the active material from dissolving while simultaneously elevating the theoretical specific capacity to 216 mAh/g, nearly double that of pure polytriphenylamine, thereby offering a compelling value proposition for next-generation battery manufacturing.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Polymerization
The formation of the final conjugated polymer relies on a precise oxidative polymerization mechanism mediated by anhydrous ferric chloride (FeCl3), which acts as both an oxidant and a Lewis acid catalyst to facilitate C-N or C-C coupling between the monomer units. In this process, the 2,8,14-tris(4-diphenylaminophenyl)-hexaazanaphthalene monomer is dissolved in re-distilled chloroform to ensure an anhydrous environment, preventing the hydrolysis of the ferric chloride which would otherwise deactivate the catalyst. The reaction proceeds under a nitrogen atmosphere at controlled temperatures between 20°C and 60°C, typically optimized at 30°C for 24 hours, allowing for the gradual growth of the polymer chain without inducing structural defects or cross-linking that could impair electrochemical performance. 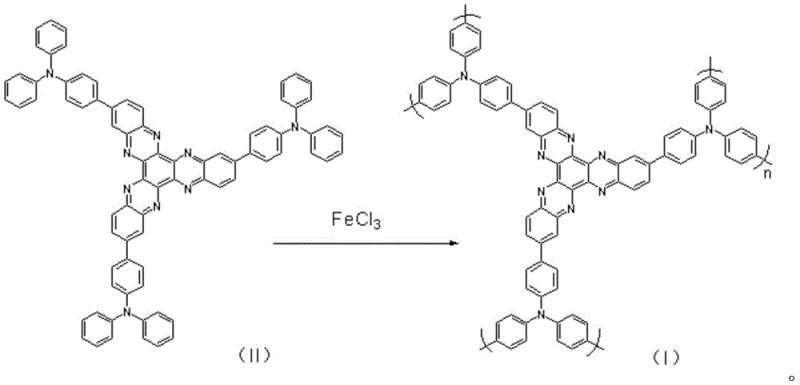 This mild reaction condition is crucial for maintaining the integrity of the nitrogen-rich heterocyclic core, ensuring that the resulting polymer retains the multiple electron transfer centers necessary for high-capacity energy storage.
This mild reaction condition is crucial for maintaining the integrity of the nitrogen-rich heterocyclic core, ensuring that the resulting polymer retains the multiple electron transfer centers necessary for high-capacity energy storage.
Impurity control is rigorously managed through the stoichiometry of the oxidant and the post-polymerization purification protocol. The molar ratio of monomer to anhydrous ferric chloride is strictly maintained between 1:4 and 1:6, preferably 1:5, to ensure complete oxidation while minimizing residual metal contaminants that could act as parasitic reaction sites within the battery cell. Following the reaction, the crude polymer is precipitated in methanol and subjected to extensive Soxhlet extraction using tetrahydrofuran (THF) to remove oligomers and unreacted monomers, which is essential for achieving the high molecular weight distribution required for mechanical stability in electrode coatings. This meticulous purification ensures that the final material exhibits uniform particle morphology and consistent electrochemical behavior, critical factors for reliable battery operation over hundreds of cycles.
How to Synthesize 2,8,14-tris(4-diphenylaminophenyl)-hexaazanaphthalene Efficiently
The synthesis of this advanced battery material involves a sequential three-step protocol that transitions from simple commodity chemicals to a complex functional polymer. The process initiates with the formation of the key diamine intermediate via palladium-catalyzed cross-coupling, followed by an acid-mediated cyclization to form the hexaazanaphthalene core, and concludes with oxidative polymerization. Each step requires precise control of temperature, atmosphere, and stoichiometry to maximize yield and purity, with the detailed standardized operating procedures outlined below for technical replication and scale-up assessment.
- Perform Suzuki coupling between 4-triphenylamine borate and 4-bromo-o-phenylenediamine using Pd(PPh3)2Cl2 catalyst at 80°C to form the diamine intermediate.
- Execute ring-closure condensation of the diamine intermediate with cyclohexanone in glacial acetic acid at 120°C to yield the hexaazanaphthalene monomer.
- Conduct oxidative polymerization of the monomer using anhydrous ferric chloride (FeCl3) in chloroform under nitrogen protection to obtain the final conjugated polymer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift towards this organic polymer architecture offers substantial strategic advantages by decoupling battery production from the volatile markets of transition metals. The reliance on abundant organic feedstocks such as boronic acids and brominated aromatics, which are widely available from the fine chemical industry, significantly mitigates the risk of raw material shortages that frequently plague the inorganic battery sector. Furthermore, the solution-processable nature of the polymer allows for flexible electrode fabrication techniques, potentially reducing the energy intensity and capital expenditure associated with the high-temperature sintering processes required for ceramic cathodes.
- Cost Reduction in Manufacturing: The elimination of expensive and scarce transition metals like cobalt and nickel from the active material formulation leads to a drastic reduction in the Bill of Materials (BOM) cost. Additionally, the synthesis utilizes standard organic reaction conditions that do not require specialized high-pressure equipment or extreme temperatures, thereby lowering the operational expenditure (OPEX) related to energy consumption and equipment maintenance. The use of ferric chloride as a polymerization agent is particularly cost-effective compared to noble metal catalysts often used in other conductive polymer syntheses, further enhancing the economic viability of large-scale production.
- Enhanced Supply Chain Reliability: By sourcing precursors from the established petrochemical and fine chemical supply chains, manufacturers can secure a more stable and diversified supply base that is less susceptible to the geopolitical tensions affecting mineral mining regions. The synthetic route is robust and tolerant to minor variations in reaction conditions, ensuring consistent batch-to-bquality which is vital for maintaining long-term supply contracts with automotive and consumer electronics clients. This reliability allows for better inventory planning and reduces the need for safety stock buffers that tie up working capital.
- Scalability and Environmental Compliance: The organic synthesis pathway generates waste streams that are generally easier to treat and manage compared to the heavy metal-laden effluents from inorganic cathode production, facilitating compliance with increasingly stringent environmental regulations. The ability to scale the reaction from gram-scale laboratory synthesis to multi-ton commercial production is supported by the use of common solvents like toluene and chloroform, for which established recovery and recycling infrastructure already exists in most chemical manufacturing facilities. This scalability ensures that the technology can meet the growing demand for energy storage without encountering bottlenecks in production capacity.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and performance of this novel organic electrode material, drawing directly from the experimental data and structural analysis provided in the patent documentation. These answers are intended to provide clarity on the material's stability, capacity metrics, and processing requirements for engineering teams evaluating its integration into existing battery architectures.
Q: How does this polymer overcome the low capacity limitations of pure polytriphenylamine?
A: By integrating a hexaazanaphthalene core with triphenylamine arms, the material leverages the multi-electron transfer capability of the nitrogen-rich heterocycle, theoretically boosting specific capacity from 109 mAh/g to 216 mAh/g while maintaining the high voltage platform of triphenylamine.
Q: What prevents the dissolution of the active material in the electrolyte during cycling?
A: The polymerization creates a large, conjugated macromolecular network that is insoluble in common organic electrolytes, unlike small molecule hexaazanaphthalenes which tend to dissolve and cause rapid capacity fading.
Q: Is the synthesis scalable for industrial battery production?
A: Yes, the route utilizes standard organic reactions like Suzuki coupling and oxidative polymerization which are amenable to scale-up, avoiding the need for rare earth metals or extreme high-pressure conditions typical of inorganic cathode synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Poly[2,8,14-tris(4-diphenylaminophenyl)-hexaazanaphthalene] Supplier
As the demand for high-performance organic battery materials accelerates, NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the sensitive oxidative polymerization and purification steps required for this conjugated polymer, ensuring stringent purity specifications and rigorous QC labs verify every batch against electrochemical performance benchmarks. We understand the critical nature of supply continuity in the energy sector and have optimized our logistics to ensure timely delivery of high-purity organic electrode materials to your manufacturing sites globally.
We invite you to engage with our technical team to discuss how this innovative polymer can enhance your product portfolio and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments that demonstrate the practical viability of integrating this material into your current cell assembly lines. Let us partner with you to drive the next generation of sustainable energy storage solutions forward.
