Advanced Manufacturing of High-Purity Ambuprofen for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously demands higher standards for active pharmaceutical ingredient intermediates, particularly for non-steroidal anti-inflammatory drug derivatives. A pivotal advancement in this sector is documented in patent CN101531611A, which outlines a robust method for preparing Ambuprofen with exceptional purity. This technical breakthrough addresses critical bottlenecks in traditional synthesis pathways, offering a streamlined approach that enhances both product quality and process efficiency. For R&D directors and procurement specialists, understanding the nuances of this acyl chloride-mediated route is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent regulatory requirements. The methodology described eliminates the need for complex purification cycles often associated with older techniques, thereby reducing the overall environmental footprint and operational costs. By leveraging this specific chemical architecture, manufacturers can achieve consistent batch-to-batch reproducibility, a key metric for long-term supply chain stability in the global analgesic market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ambuprofen relied heavily on esterification pathways or direct salification methods that presented significant technical and economic challenges. Prior art, such as the methods described in Spanish patents ES2023585 and ES2028601, often involved reacting ibuprofen with methanol to form esters followed by ammonolysis, or direct salification with ethanolamine under reflux conditions. These conventional approaches frequently suffered from low yields, typically hovering around 85% to 88%, and generated complex mixtures that were difficult to purify effectively. The necessity to recover large quantities of unreacted ethanolamine or methanol added substantial complexity to the downstream processing units, increasing energy consumption and waste generation. Furthermore, the resulting product purity was often insufficient for direct pharmaceutical application, necessitating additional recrystallization steps that further eroded overall yield and increased production lead times. These inefficiencies created a fragile supply chain vulnerable to raw material fluctuations and regulatory scrutiny regarding residual solvents and impurities.
The Novel Approach
In stark contrast, the novel approach detailed in the referenced patent utilizes a two-step acyl chloride strategy that fundamentally restructures the synthesis logic for cost reduction in API manufacturing. By converting ibuprofen directly into its acyl chloride derivative using thionyl chloride and a catalytic amount of dimethylformamide in toluene, the process activates the carboxylic acid group more efficiently than esterification. This activated intermediate then undergoes amidation with ethanolamine in the presence of potassium carbonate as an acid scavenger in dichloromethane. This pathway bypasses the thermodynamic equilibrium limitations of direct amidation and avoids the formation of stubborn ester byproducts. The result is a significantly simplified workflow that enhances raw material utilization rates and minimizes the generation of hard-to-remove impurities. This strategic shift allows for a more predictable manufacturing timeline and reduces the dependency on extensive purification infrastructure, making it an ideal candidate for commercial scale-up of complex analgesic intermediates.
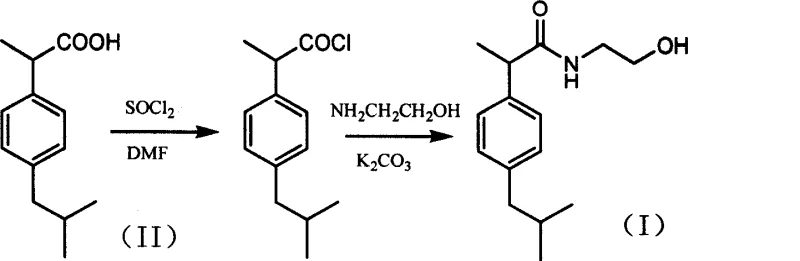
Mechanistic Insights into Acyl Chloride-Mediated Amidation
The core of this high-efficiency synthesis lies in the precise control of the acylation and subsequent amidation mechanisms. In the first step, dimethylformamide acts as a nucleophilic catalyst, reacting with thionyl chloride to form a highly reactive Vilsmeier-Haack type intermediate which rapidly converts the ibuprofen carboxylic acid into the corresponding acyl chloride. This reaction is conducted in toluene at temperatures between 60°C and 90°C, ensuring complete conversion while minimizing thermal degradation of the sensitive chiral center. The removal of toluene under reduced pressure yields a crude acyl chloride that is sufficiently pure for the next stage without distillation. In the second step, the crude acyl chloride is introduced into a suspension of ethanolamine and potassium carbonate in dichloromethane under ice bath conditions. The potassium carbonate serves a dual purpose: it neutralizes the hydrochloric acid byproduct generated during amide bond formation and maintains a basic environment that prevents the protonation of the nucleophilic amine, thereby driving the reaction to completion. This careful orchestration of reagents ensures that side reactions, such as the formation of N,N-disubstituted byproducts, are kept to a minimum.
Impurity control is inherently built into this mechanistic design, addressing a primary concern for quality assurance teams focused on high-purity Ambuprofen specifications. The use of potassium carbonate as a solid base in a heterogeneous system allows for easier separation of inorganic salts compared to soluble organic bases like triethylamine, which can be difficult to remove completely from the final product. Comparative data indicates that replacing organic bases with inorganic carbonates significantly reduces the presence of amine salts in the final organic layer. Additionally, the choice of dichloromethane as the amidation solvent provides excellent solubility for the reactants while facilitating efficient aqueous workup procedures. The washing sequence involving saturated sodium bicarbonate, dilute hydrochloric acid, and brine effectively removes residual acids, bases, and water-soluble impurities. This rigorous purification protocol embedded within the synthesis route ensures that the final product consistently meets purity thresholds exceeding 99% without the need for energy-intensive recrystallization, thereby preserving the crystalline integrity and bioavailability profile required for downstream pharmaceutical formulation.
How to Synthesize Ambuprofen Efficiently
Implementing this synthesis route requires strict adherence to the optimized molar ratios and temperature profiles established in the patent data to ensure maximum yield and purity. The process begins with the activation of ibuprofen, where maintaining the molar ratio of ibuprofen to DMF to thionyl chloride within the range of 1:0.03:1.8 is critical for preventing over-chlorination or incomplete reaction. Following the removal of the solvent, the amidation step must be controlled carefully, with the addition rate of the acyl chloride solution managed to prevent exothermic spikes that could degrade the ethanolamine. Detailed standard operating procedures regarding stirring speeds, addition times, and phase separation techniques are vital for translating this laboratory-scale success into a robust manufacturing protocol. The standardized synthesis steps outlined below provide a foundational framework for process engineers to adapt this chemistry for larger reactor volumes while maintaining safety and quality standards.
- Perform acyl chloride reaction by reacting ibuprofen with thionyl chloride and DMF catalyst in toluene at 60-90°C for 4-10 hours.
- Remove toluene under reduced pressure to obtain crude ibuprofen acyl chloride intermediate.
- Conduct amidation by adding the crude acyl chloride to a suspension of ethanolamine and potassium carbonate in dichloromethane at room temperature.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, adopting this advanced synthesis methodology offers substantial benefits that extend beyond mere chemical yield, directly impacting the bottom line and supply chain resilience. The elimination of complex esterification and recovery steps translates into a drastically simplified production flow, which reduces the total cycle time per batch and increases the overall throughput of the manufacturing facility. For procurement managers, this efficiency means a more stable supply of high-purity Ambuprofen, mitigating the risks associated with production bottlenecks that often plague older, less efficient technologies. The use of common, commercially available solvents like toluene and dichloromethane ensures that raw material sourcing remains straightforward and cost-effective, avoiding the volatility associated with specialized or scarce reagents. Furthermore, the high selectivity of the reaction minimizes waste generation, aligning with increasingly stringent environmental regulations and reducing the costs associated with waste disposal and treatment.
- Cost Reduction in Manufacturing: The streamlined two-step process significantly lowers operational expenditures by removing the need for energy-intensive distillation columns required to recover excess alcohols in traditional methods. By utilizing potassium carbonate instead of expensive organic bases, the raw material costs are further optimized, and the downstream purification burden is lightened. This qualitative improvement in process economy allows for a more competitive pricing structure without compromising on the stringent quality standards required for pharmaceutical intermediates. The reduction in processing steps also decreases labor hours and equipment occupancy time, contributing to an overall leaner manufacturing model that enhances profit margins.
- Enhanced Supply Chain Reliability: The robustness of this chemical route ensures consistent output quality, which is paramount for maintaining trust with downstream API manufacturers. The high yield and purity reduce the likelihood of batch failures or rejections, ensuring a steady flow of materials into the supply chain. Additionally, the scalability of the process means that production volumes can be ramped up quickly to meet surges in demand without requiring significant capital investment in new specialized equipment. This flexibility is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing partners to respond agilely to market dynamics and regulatory changes.
- Scalability and Environmental Compliance: The reaction conditions, operating primarily at moderate temperatures and atmospheric pressure, are inherently safer and easier to scale from pilot plants to multi-ton commercial reactors. The simplified workup procedure reduces the volume of aqueous waste generated, easing the load on wastewater treatment facilities and ensuring compliance with environmental protection standards. The ability to recycle solvents like toluene and dichloromethane further enhances the sustainability profile of the manufacturing process. This alignment with green chemistry principles not only mitigates regulatory risk but also appeals to environmentally conscious stakeholders and end-users in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Ambuprofen synthesized via this patented method. These insights are derived directly from the experimental data and comparative analysis provided in the intellectual property documentation, ensuring accuracy and relevance for industry professionals. Understanding these details helps stakeholders make informed decisions regarding technology transfer, quality assurance, and long-term procurement strategies. The answers reflect the consensus on best practices for implementing this chemistry in a regulated manufacturing environment.
Q: What purity levels can be achieved with this Ambuprofen synthesis method?
A: The patented process consistently achieves product purity exceeding 93%, with optimized conditions reaching above 99% without requiring additional recrystallization steps.
Q: How does this method improve cost efficiency compared to traditional esterification routes?
A: By eliminating complex esterification and subsequent ammonolysis steps, this route reduces solvent consumption and simplifies downstream processing, leading to significant operational cost reductions.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes common solvents like toluene and dichloromethane and operates under mild conditions, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ambuprofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic routes to deliver high-value pharmaceutical intermediates to the global market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest international standards. Our facility is designed to handle complex chemistries safely and effectively, providing our partners with a secure and dependable source for their critical raw materials.
We invite you to collaborate with us to leverage this superior synthesis technology for your specific product needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency and product quality. Let us be your partner in achieving excellence in pharmaceutical manufacturing.
