Advanced Synthesis Of Homonojirimycin Glycosides For Commercial Scale-Up And High Purity Standards
Advanced Synthesis Of Homonojirimycin Glycosides For Commercial Scale-Up And High Purity Standards
The pharmaceutical industry continuously seeks robust synthetic pathways for potent antidiabetic agents, and Patent CN1016784B presents a significant advancement in the preparation of homonojirimycin glycosides. These compounds function as powerful inhibitors of carbohydrate digestive enzymes, offering a targeted approach to managing postprandial hyperglycemia without the systemic side effects associated with other metabolic interventions. The patent outlines a comprehensive chemical strategy that transforms protected glucopyranose derivatives into complex heptitol glycosides through a series of highly controlled reactions. This technical insight report analyzes the feasibility of this route for industrial application, focusing on the critical parameters that ensure high purity and reproducibility. For a reliable pharmaceutical intermediate supplier, understanding the nuances of this synthesis is essential for delivering high-purity antidiabetic intermediates that meet the rigorous demands of global drug developers. The following analysis dissects the chemical logic, operational advantages, and supply chain implications of this proprietary technology.
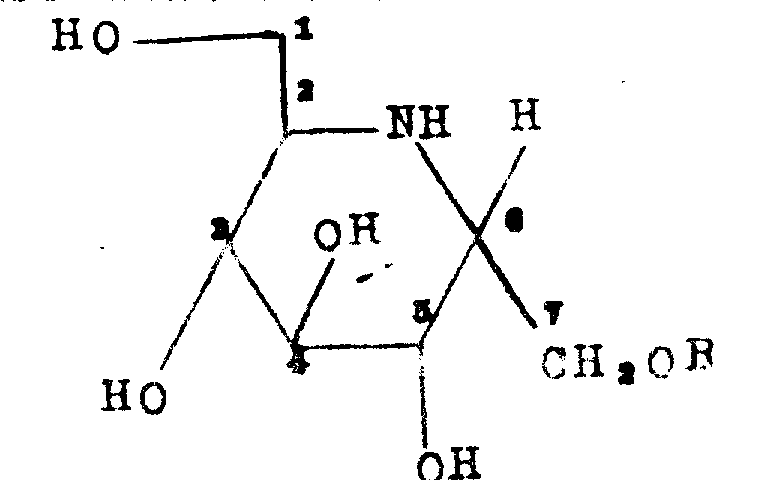
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing iminosugar derivatives often suffer from low stereoselectivity and cumbersome protection-deprotection sequences that hinder commercial scale-up of complex glycosides. Conventional routes may rely on non-selective glycosylation conditions that produce difficult-to-separate anomeric mixtures, significantly increasing downstream purification costs and waste generation. Furthermore, older methodologies frequently utilize harsh reaction conditions that can compromise the integrity of sensitive functional groups, leading to degraded product quality and inconsistent batch-to-batch performance. The reliance on inefficient cyclization strategies in prior art often results in poor yields of the core piperidine ring, which is the pharmacophore responsible for enzyme inhibition. These inefficiencies create bottlenecks in production capacity, making it challenging to secure a consistent supply of high-quality intermediates for clinical and commercial needs. Consequently, manufacturers face elevated operational expenses and extended lead times, which negatively impact the overall cost structure of the final active pharmaceutical ingredient.
The Novel Approach
The methodology described in Patent CN1016784B introduces a refined synthetic pathway that addresses these historical challenges through precise control of reaction conditions and reagent selection. By employing a specific sequence involving Wittig olefination followed by mercury-mediated cyclization, the process achieves superior stereocontrol during the formation of the critical heptitol backbone. This novel approach utilizes protected glycosyl halides in conjunction with mercury salts like mercuric bromide or cyanide to facilitate highly selective glycosidic bond formation under mild conditions. The use of molecular sieves as desiccants and weak bases further enhances reaction efficiency by scavenging acidic byproducts that could otherwise catalyze decomposition. This strategic design allows for the production of specific anomers with high purity, drastically simplifying the isolation process and reducing the need for extensive chromatographic purification. Such improvements translate directly into cost reduction in API manufacturing by minimizing material loss and maximizing throughput in standard reactor setups.
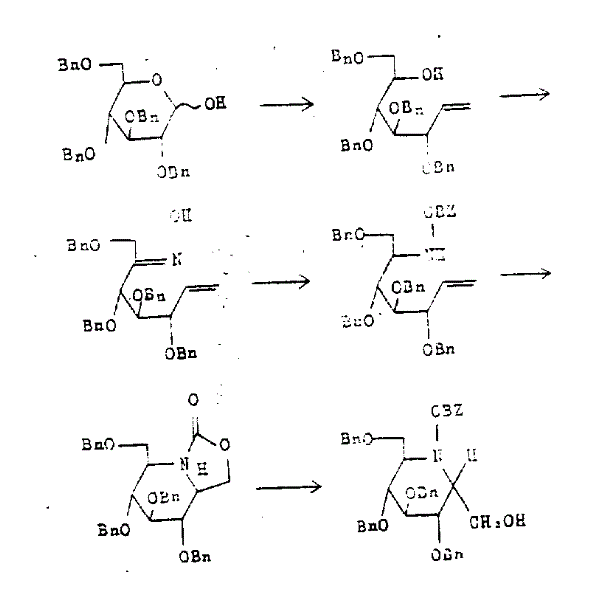
Mechanistic Insights into Mercury-Mediated Cyclization and Glycosylation
The core of this synthetic strategy lies in the sophisticated use of organomercury chemistry to construct the bicyclic framework essential for biological activity. The process begins with the conversion of protected glucopyranose into an unsaturated carbamate via a Wittig reaction, which extends the carbon chain to the required seven-carbon heptitol length. Subsequent oxidation and oxime formation set the stage for a critical reduction step using metal hydrides like lithium aluminum hydride, establishing the amine functionality needed for ring closure. The cyclization step is particularly noteworthy, as it employs mercury acetate or trifluoroacetate to promote the intramolecular attack of the nitrogen onto the unsaturated system, forming the stable piperidine ring with high fidelity. This mechanistic pathway ensures that the stereochemistry at the ring junction is strictly controlled, which is vital for the compound's ability to fit into the active site of carbohydrate digestive enzymes. Understanding this mechanism allows process chemists to optimize reagent stoichiometry and temperature profiles for maximum efficiency.
Impurity control is inherently built into this synthetic design through the use of robust protecting group strategies and selective deprotection methods. The benzyl and acetyl protecting groups used throughout the synthesis are orthogonal enough to allow for sequential removal without affecting the sensitive glycosidic linkage. The final deprotection step utilizes catalytic hydrogenation over palladium on carbon, which cleanly removes benzyl groups while simultaneously reducing any remaining unsaturated impurities. This dual function of the hydrogenation step significantly enhances the purity profile of the final product, ensuring that the resulting free amine meets stringent purity specifications required for pharmaceutical applications. Additionally, the ability to convert the free base into various pharmaceutically acceptable acid addition salts, such as hydrochlorides or tosylates, provides flexibility in formulation and stability. This level of control over the impurity profile is crucial for R&D directors evaluating the feasibility of this route for regulatory filing and commercial production.
How to Synthesize Homonojirimycin Glycosides Efficiently
Executing this synthesis requires careful attention to the specific reaction conditions outlined in the patent examples to ensure optimal yield and quality. The process involves multiple stages, starting from readily available sugar derivatives and progressing through oxidation, reduction, and cyclization before the final glycosylation and deprotection. Each step must be monitored closely to prevent side reactions, particularly during the mercury-mediated stages where moisture control is critical. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety considerations regarding mercury handling and waste disposal. Adhering to these protocols ensures that the commercial scale-up of complex glycosides can be achieved with consistent results.
- Prepare the core heptitol skeleton via Wittig reaction and oxidation of protected glucopyranose.
- Execute mercury-mediated cyclization to form the piperidine ring structure.
- Perform glycosylation with protected glycosyl halides followed by catalytic hydrogenation for deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial cost savings by utilizing common starting materials and avoiding exotic reagents that drive up expenses. The ability to perform key transformations at room temperature or under mild reflux conditions reduces energy consumption and lowers the operational burden on manufacturing facilities. Furthermore, the high selectivity of the glycosylation step minimizes the formation of byproducts, which means less solvent and silica gel are required for purification, leading to significant waste reduction. These factors combine to create a more economical process that enhances supply chain reliability by reducing the risk of production delays caused by purification bottlenecks. For supply chain heads, this translates to reducing lead time for high-purity pharmaceutical intermediates, ensuring that clinical trials and commercial launches can proceed without material shortages.
- Cost Reduction in Manufacturing: The elimination of complex chiral resolution steps and the use of efficient catalytic cycles significantly lower the overall cost of goods. By avoiding the need for expensive enzymatic resolutions or specialized chiral columns, manufacturers can achieve a leaner cost structure. The process relies on standard chemical transformations that can be performed in existing multipurpose reactors, avoiding the need for capital-intensive equipment upgrades. This operational efficiency allows for competitive pricing without compromising on the quality of the intermediate, making it an attractive option for cost-sensitive generic drug development projects.
- Enhanced Supply Chain Reliability: The starting materials, such as protected glucopyranose and common mercury salts, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality, ensuring consistent output even with different batches of reagents. This stability is critical for maintaining a continuous supply of critical intermediates, preventing disruptions that could impact downstream API production schedules. Procurement managers can rely on this chemistry to secure long-term supply agreements with confidence.
- Scalability and Environmental Compliance: While the process utilizes mercury salts, the amounts used are catalytic, and the workup procedures described ensure effective removal and recovery of heavy metals, aligning with environmental compliance standards. The final hydrogenation step is a green chemistry approach that uses hydrogen gas and produces minimal waste compared to stoichiometric reduction methods. The scalability of the route has been demonstrated through multi-gram examples in the patent, indicating a clear path to kilogram and ton-scale production. This scalability ensures that the supply can grow in tandem with the clinical and commercial demands of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these homonojirimycin derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. This section aims to clarify the capabilities of the manufacturing process and the quality attributes of the final product for potential partners.
Q: What is the primary mechanism of action for these compounds?
A: These compounds act as potent inhibitors of carbohydrate digestive enzymes, preventing the hydrolysis of complex carbohydrates into absorbable sugars, thereby reducing postprandial blood glucose levels without affecting systemic glucose transport.
Q: Are heavy metal catalysts used in the final product?
A: While mercury salts are utilized during the cyclization and glycosylation steps as catalysts, the final purification process involves rigorous workup and catalytic hydrogenation, ensuring that the final pharmaceutical intermediate meets stringent purity specifications with negligible residual metals.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the described methodology employs standard organic synthesis techniques such as Wittig reactions and catalytic hydrogenation, which are well-established in industrial settings, allowing for significant cost reduction in API manufacturing through scalable operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Homonojirimycin Glycosides Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced synthetic technology to support your drug development pipeline with high-quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from clinical phases to full-scale market supply. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of homonojirimycin glycosides meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of carbohydrate chemistry effectively, delivering products that facilitate your regulatory success.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for this specific intermediate. Request a Customized Cost-Saving Analysis to understand how our manufacturing efficiencies can benefit your project economics. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to meet your exact requirements. Partner with us to secure a reliable source of high-purity antidiabetic intermediates that will drive the success of your therapeutic programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
