Scalable Production of Androst-2-ene-17-one: A Technical Breakthrough for Steroid Hormone Manufacturing
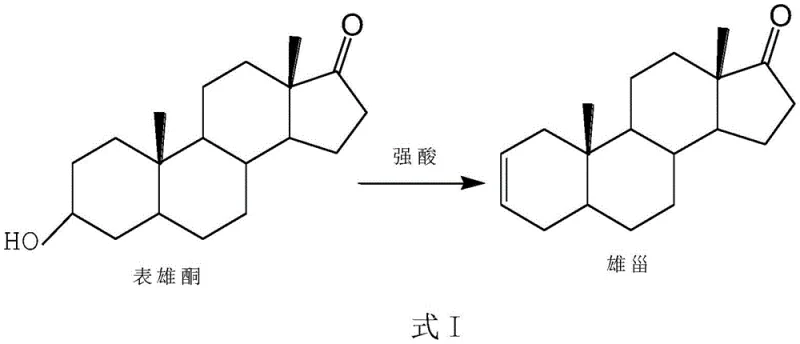
The pharmaceutical industry continuously seeks robust synthetic routes for critical steroid intermediates, particularly those serving as precursors for neuromuscular blocking agents. Patent CN112625077A introduces a transformative preparation method for Androst-2-ene-17-one, a pivotal building block in the synthesis of rocuronium bromide and vecuronium bromide. This innovation addresses long-standing inefficiencies in steroid chemistry by replacing complex multi-step sequences or heterogeneous catalytic systems with a streamlined, one-step dehydration protocol. By utilizing epiandrosterone as the starting material and employing strong acids as homogeneous catalysts, the process achieves high conversion rates while drastically simplifying post-reaction processing. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for high-value muscle relaxant medications, ensuring both economic viability and environmental compliance in modern API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Androst-2-ene-17-one has been plagued by operational complexities that hinder efficient commercial production. Traditional two-step methods typically involve an initial sulfonylation of the 3-position hydroxyl group followed by an elimination reaction mediated by organic amines or pyridine derivatives. These pathways necessitate the use of substantial quantities of organic alkalis, generating difficult-to-treat waste streams that impose heavy environmental burdens and disposal costs. Alternatively, existing one-step methods have relied on solid-phase acid catalysts supported on silica gel or alumina. While conceptually simple, these heterogeneous systems suffer from severe scalability issues; the presence of solid adsorbents complicates mixing and heat transfer in large reactors, often leading to inconsistent reaction kinetics and failure during process amplification. Furthermore, the requirement to filter and dispose of tons of silica gel waste renders these methods economically unattractive for large-scale operations.
The Novel Approach
The methodology disclosed in patent CN112625077A fundamentally reengineers this transformation by adopting a homogeneous acid-catalyzed dehydration strategy. Instead of relying on solid supports, the process utilizes soluble strong acids such as p-toluenesulfonic acid, methanesulfonic acid, or sulfuric acid directly in an organic solvent medium. This shift eliminates the physical barriers associated with solid-liquid interfaces, ensuring uniform reaction conditions throughout the vessel. The result is a process that not only shortens the synthetic timeline by consolidating steps but also removes the need for expensive adsorbents and excessive organic bases. For a reliable pharmaceutical intermediates supplier, this approach translates into a cleaner, more controllable workflow that minimizes the generation of hazardous waste and simplifies the isolation of the target ketone, thereby enhancing overall process operability and yield consistency.
Mechanistic Insights into Acid-Catalyzed Dehydration
The core chemical transformation involves the acid-catalyzed elimination of water from epiandrosterone to form the conjugated enone system characteristic of Androst-2-ene-17-one. In this mechanism, the strong acid catalyst protonates the hydroxyl group at the C3 position, converting it into a superior leaving group (water). Under thermal conditions, typically ranging from 60°C to 160°C, the molecule undergoes elimination, likely proceeding through an E1 or E2 pathway depending on the specific acid strength and solvent polarity. The formation of the double bond between C2 and C3 is driven by the thermodynamic stability of the resulting conjugated system with the C17 ketone. Crucially, the choice of catalyst and the control of reflux ratios allow for precise modulation of reaction kinetics, preventing over-reaction or degradation of the sensitive steroid skeleton. This mechanistic clarity enables chemists to fine-tune conditions to maximize the formation of the desired 2-ene isomer while suppressing potential byproducts.
Impurity control is another critical aspect addressed by this mechanistic understanding. The patent highlights that the use of specific strong acids in conjunction with controlled reflux parameters significantly enhances the purity profile of the crude product. Unlike solid-phase catalysts which may have uneven active site distribution leading to localized hot spots and side reactions, the homogeneous nature of the liquid acid ensures consistent catalytic activity. Following the reaction, a simple alkaline wash effectively neutralizes the acid catalyst, allowing for its removal without complex chromatographic steps. Subsequent recrystallization from solvents like ethanol or methanol further purifies the product by exploiting solubility differences between the target Androst-2-ene-17-one and any remaining stereoisomers or unreacted starting material. This rigorous control over the reaction environment and workup procedure is essential for delivering high-purity Androst-2-ene-17-one suitable for stringent pharmaceutical applications.
How to Synthesize Androst-2-ene-17-one Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to ensure optimal yield and safety. The process begins by dissolving epiandrosterone in a suitable organic solvent such as toluene, followed by the addition of a catalytic amount of a strong acid. The mixture is then heated to reflux, often utilizing a Dean-Stark trap or similar apparatus to remove the water generated during the dehydration, which drives the equilibrium towards product formation. Maintaining the correct temperature and monitoring the reaction progress are vital to prevent decomposition. Once the conversion is complete, the reaction mixture is cooled and subjected to a straightforward workup involving neutralization and solvent recovery. The detailed standardized synthesis steps see the guide below for specific operational protocols.
- Combine epiandrosterone with a strong acid catalyst (such as p-toluenesulfonic acid or methanesulfonic acid) in an organic solvent like toluene.
- Heat the mixture to reflux temperatures between 60°C and 160°C to facilitate the dehydration reaction, optionally using a water separator.
- Cool the reaction mixture, wash with alkaline water to neutralize the acid, concentrate under reduced pressure, and recrystallize the crude product to obtain high-purity Androst-2-ene-17-one.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers compelling strategic benefits that extend beyond mere chemical efficiency. The transition from heterogeneous solid-phase catalysis to a homogeneous liquid-phase system resolves critical bottlenecks associated with manufacturing scale-up. By eliminating the need for filtering massive quantities of silica gel or alumina, the process drastically reduces cycle times and equipment downtime. This streamlining of operations directly contributes to cost reduction in steroid hormone manufacturing, as it lowers labor requirements, minimizes solvent consumption for washing solids, and reduces the volume of solid waste requiring disposal. Furthermore, the avoidance of large amounts of organic alkalis simplifies wastewater treatment protocols, aligning production with increasingly strict environmental regulations and reducing the risk of regulatory shutdowns.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the elimination of expensive solid support materials and the reduction in reagent consumption. Traditional methods often incur high costs due to the purchase and disposal of silica gel carriers and the use of stoichiometric amounts of organic bases. By switching to a catalytic amount of soluble acid, the raw material costs are significantly lowered. Additionally, the simplified post-treatment process reduces energy consumption associated with filtration and drying steps. These cumulative efficiencies result in substantial cost savings per kilogram of produced intermediate, allowing for more competitive pricing in the global market for neuromuscular blocking agent precursors without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that are prone to failure during scale-up. The homogeneous nature of this new dehydration method ensures that reaction performance in a 100-liter pilot plant closely mirrors that of a multi-ton industrial reactor. This predictability reduces the risk of batch failures and ensures consistent delivery schedules for downstream API manufacturers. Moreover, the reagents required, such as toluene and common sulfonic acids, are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages. This reliability makes the supplier a more dependable partner for long-term contracts, ensuring that the production of critical anesthesia medications remains uninterrupted even during periods of market volatility.
- Scalability and Environmental Compliance: As the demand for steroid-based medications grows, the ability to scale production efficiently is paramount. This method is inherently scalable because it avoids the physical limitations of solid-liquid mixing, which often become problematic in large vessels. The reduction in three wastes (waste gas, waste water, and solid waste) is another major advantage, as it lowers the environmental footprint of the manufacturing facility. By generating less hazardous waste and avoiding the use of heavy metal catalysts or persistent organic pollutants, the process facilitates easier compliance with environmental protection laws. This sustainability profile not only protects the manufacturer from potential fines but also enhances the brand reputation of the supply chain partners who prioritize green chemistry initiatives in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Androst-2-ene-17-one using the patented dehydration method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on process capabilities and product quality. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their broader pharmaceutical supply networks.
Q: What are the primary advantages of the new acid-catalyzed dehydration method over traditional solid-phase catalysts?
A: The new method eliminates the need for expensive and bulky solid-phase carriers like silica gel, transitioning the process to a homogeneous system. This significantly improves scalability by avoiding filtration issues associated with heterogeneous mixtures and reduces the generation of solid waste, leading to a more environmentally friendly and cost-effective manufacturing process.
Q: How does this synthesis route impact the purity profile of the final steroid intermediate?
A: By utilizing specific strong acids like p-toluenesulfonic acid under controlled reflux conditions, the reaction achieves high conversion rates with minimal side reactions. Subsequent recrystallization steps, as detailed in the patent examples, allow for the removal of residual starting materials and isomers, consistently delivering product with purity levels exceeding 97%, which is critical for downstream pharmaceutical applications.
Q: Is this process suitable for large-scale commercial production of muscle relaxant intermediates?
A: Yes, the process is specifically designed for commercial scale-up. By removing the reliance on solid adsorbents that cause amplification failures in large reactors, the homogeneous acid-catalyzed system ensures consistent heat and mass transfer. This reliability makes it an ideal candidate for producing multi-ton quantities of rocuronium bromide intermediates required by the global anesthesia market.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Androst-2-ene-17-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team has extensively analyzed the pathway described in CN112625077A and possesses the expertise to implement this advanced dehydration technology at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Androst-2-ene-17-one meets the exacting standards required for the synthesis of rocuronium bromide and other potent steroid hormones.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to optimize their supply chains. By leveraging our technical capabilities, you can achieve significant efficiencies in your production of neuromuscular blocking agents. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized synthesis of Androst-2-ene-17-one can enhance your operational performance and reduce overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
