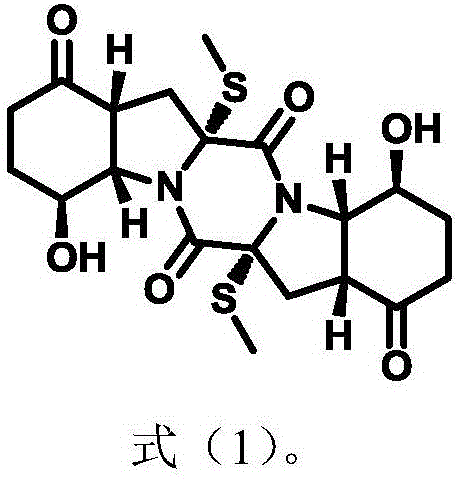
Advanced Asymmetric Synthesis of Epicoccin G for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust supply chains for complex natural products, particularly those exhibiting potent biological activities such as antitumor and antiviral properties. Patent CN111087402A discloses a groundbreaking method for the asymmetric synthesis of Epicoccin G, a member of the epithiodiketopiperazines (ETP) family. Historically, obtaining sufficient quantities of ETP alkaloids has been a significant bottleneck due to their low natural abundance in fungal sources and the arduous extraction processes required. This patent presents a transformative solution by enabling the gram-scale production of Epicoccin G and its derivatives through a concise, chemically elegant route. By leveraging cheap and readily available starting materials, this methodology not only secures the supply of this valuable high-purity pharmaceutical intermediate but also opens new avenues for structure-activity relationship (SAR) studies that were previously impossible. For R&D teams focused on oncology and infectious diseases, access to such reliable synthetic routes is critical for advancing drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for acquiring Epicoccin G rely heavily on fermentation and extraction from endophytic fungi such as Phoma species. This biological approach suffers from inherent variability, where yield is dictated by strain stability, fermentation conditions, and seasonal factors. Furthermore, the isolation of the target alkaloid from complex fermentation broths requires extensive chromatographic purification, leading to significant material loss and high operational costs. From a chemical synthesis perspective, prior art routes have often been plagued by excessive step counts, harsh reaction conditions, and poor stereocontrol, resulting in racemic mixtures that require difficult resolution. These inefficiencies translate directly into cost reduction in pharmaceutical intermediate manufacturing challenges, making large-scale clinical supply untenable. The reliance on scarce natural resources also poses a severe risk to supply chain reliability, potentially halting research projects due to material shortages.
The Novel Approach
The novel synthetic strategy outlined in the patent overcomes these hurdles through a streamlined, convergent design that prioritizes atom economy and stereochemical fidelity. The route initiates with a highly stereoselective [4+2] cycloaddition between 3,5-diiodo-2-pyrone and a chiral pyrrole derivative, effectively setting the multiple chiral centers early in the sequence. A standout feature of this methodology is the utilization of modern techniques such as microwave-assisted elimination and visible-light photocatalytic oxidation, which significantly accelerate reaction rates and improve yields compared to thermal methods. The process culminates in an oxidative dimerization and a silver-mediated sulfuration step to construct the characteristic disulfide bridge of the ETP core. This approach allows for the commercial scale-up of complex natural product analogues with unprecedented efficiency. By avoiding precious metal catalysts in the early stages and utilizing standard reagents like zinc and palladium on carbon, the process is inherently more scalable and cost-effective.
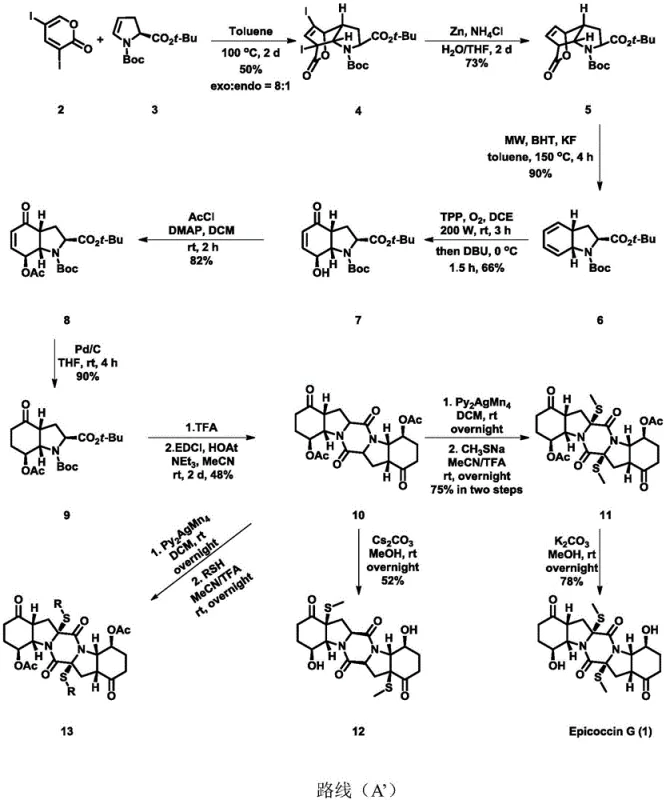
Mechanistic Insights into Photo-Oxidative Dimerization and Sulfuration
The core of this synthetic achievement lies in the precise manipulation of oxidation states to construct the densely functionalized diketopiperazine scaffold. The mechanism begins with the Diels-Alder reaction, where the electron-deficient pyrone acts as the dienophile against the electron-rich pyrrole, establishing the bicyclic framework with high exo selectivity. Following reductive dehalogenation, the system undergoes a retro-Diels-Alder-type elimination under microwave irradiation to reveal the conjugated diene system necessary for subsequent functionalization. A critical mechanistic step is the photo-oxidation using tetraphenylporphyrin (TPP) as a sensitizer. Under 200W illumination and oxygen atmosphere, singlet oxygen is generated, which reacts with the diene to form an endoperoxide intermediate. Subsequent treatment with DBU induces a rearrangement to the observed enone-alcohol motif. This photochemical step is superior to traditional chemical oxidants as it avoids over-oxidation and preserves the sensitive stereochemistry established in the first step.
Impurity control is rigorously managed through the choice of reagents and reaction conditions. The use of chiral starting materials ensures that the final product is enantiomerically pure, eliminating the need for chiral resolution which often halves the theoretical yield. During the dimerization step (conversion of compound 9 to 10), the use of EDCI and HOAt promotes efficient amide bond formation while minimizing racemization at the alpha-carbon. The final installation of the sulfur bridge utilizes bis(pyridine)silver permanganate, a mild oxidant that facilitates the formation of the carbon-sulfur bonds without degrading the sensitive ketone functionalities. This careful selection of oxidants ensures that the impurity profile remains clean, a crucial factor for a reliable Epicoccin G supplier aiming to meet stringent regulatory standards for clinical grade materials. The ability to swap thiol reagents in the final step further demonstrates the robustness of the mechanism, allowing for the generation of diverse libraries.
How to Synthesize Epicoccin G Efficiently
The synthesis of Epicoccin G described in this patent represents a significant leap forward in process chemistry, offering a clear path from grams to kilograms. The protocol is designed to be operationally simple, utilizing common organic solvents like toluene, dichloromethane, and acetonitrile, which facilitates easy solvent recovery and recycling in a plant setting. The sequence involves distinct phases: core construction, functional group manipulation, and final dimerization. Each step has been optimized for yield and purity, with specific attention paid to temperature control and reaction times to prevent side reactions. For process chemists looking to implement this route, the detailed conditions provided in the patent serve as a robust starting point for technology transfer. The following guide outlines the standardized synthesis steps derived from the patent data.
- Perform a [4+2] cycloaddition between 3,5-diiodo-2-pyrone and a chiral pyrrole derivative to establish the core stereochemistry.
- Execute reductive dehalogenation followed by microwave-assisted elimination to generate the cyclic alkene intermediate.
- Conduct photo-oxidation using tetraphenylporphyrin and oxygen, followed by acetylation and hydrogenation to prepare the monomer.
- Induce oxidative dimerization using EDCI/HOAt, followed by silver-mediated sulfuration and final deprotection to yield Epicoccin G.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The shift from extraction to total synthesis fundamentally alters the cost structure and risk profile of sourcing Epicoccin G. By relying on commodity chemicals rather than biological fermentation, the supply chain becomes decoupled from agricultural variables and biological contamination risks. This transition ensures a consistent, year-round availability of the material, which is essential for maintaining continuous clinical trial timelines. Furthermore, the shortened synthetic route reduces the overall inventory holding time and working capital requirements associated with long manufacturing cycles. The use of scalable technologies like microwave reactors and flow-compatible photochemistry suggests that the process can be readily adapted to continuous manufacturing environments, further enhancing efficiency.
- Cost Reduction in Manufacturing: The economic viability of this route is driven by the use of inexpensive starting materials such as 3,5-diiodo-2-pyrone and protected pyrroles, which are commercially available in bulk quantities. Unlike previous methods that may have required exotic chiral catalysts or cryogenic conditions, this process operates largely at room temperature or moderate heating, significantly lowering energy consumption. The elimination of complex purification steps associated with natural extraction translates to substantial cost savings in downstream processing. Additionally, the high yields observed in key steps, such as the microwave elimination and hydrogenation, maximize material throughput, reducing the cost per gram of the final active ingredient.
- Enhanced Supply Chain Reliability: Dependence on a single synthetic route with well-defined parameters mitigates the risk of supply disruption. The reagents used, including zinc powder, palladium on carbon, and standard coupling agents, are part of the global chemical infrastructure, ensuring that raw material shortages are unlikely. The modular nature of the synthesis allows for flexible production scheduling; intermediates like compound 10 can potentially be stockpiled and converted to the final product or its derivatives on demand. This flexibility is crucial for responding to fluctuating market demands in the pharmaceutical sector. By establishing a domestic or regional synthesis capability, companies can reduce lead time for high-purity pharmaceutical intermediates and avoid international logistics bottlenecks.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing catalytic amounts of reagents where possible and avoiding stoichiometric heavy metal waste where alternatives exist. The photo-oxidation step, for instance, uses oxygen from the air as the terminal oxidant, generating water as the only byproduct, which is a significant environmental advantage over chromium or manganese-based oxidations. The scalability is evidenced by the successful gram-scale execution described in the examples, providing confidence for ton-scale production. Waste streams are primarily organic solvents which can be distilled and reused, aligning with modern sustainability goals and reducing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into their supply chains. The following questions address common inquiries regarding the scalability, purity, and versatility of the patented method. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making. Whether you are concerned about regulatory compliance or the ability to produce specific analogues, this section clarifies the capabilities of the technology.
Q: Why is synthetic Epicoccin G preferred over natural extraction?
A: Natural extraction yields are extremely low due to the low abundance of the producing fungus and difficult isolation processes. Synthetic routes provide consistent supply, higher purity, and the ability to create structural derivatives for SAR studies.
Q: What is the key stereochemical control step in this synthesis?
A: The stereochemistry is primarily controlled at the beginning of the sequence using a chiral (S)-2,3-dihydro-1H-pyrrole derivative. This chirality is preserved through the Diels-Alder cycloaddition and subsequent transformations.
Q: Can this synthetic route produce Epicoccin G derivatives?
A: Yes, the route is highly modular. By varying the thiol reagent in the final sulfuration step (using different mercaptans instead of sodium methyl mercaptide), various tolylthio or alkylthio derivatives can be synthesized for biological screening.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epicoccin G Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing high-quality intermediates for advanced drug development. While the patent CN111087402A provides the intellectual foundation, translating this chemistry into a robust commercial process requires deep expertise in process optimization and quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. We are committed to meeting stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify the identity and potency of every batch. Our facility is equipped to handle the specific unit operations required by this synthesis, including photochemical reactors and microwave-assisted processing.
We invite pharmaceutical partners to collaborate with us to leverage this innovative synthetic route for their Epicoccin G requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs and timeline constraints. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can accelerate the development of ETP-based therapeutics and bring life-saving medicines to patients faster. Let us be your trusted partner in navigating the complexities of natural product synthesis.
