Advanced Synthesis of Mequindox Metabolites for High-Purity Veterinary Drug Research and Development
The pharmaceutical and agrochemical industries are increasingly demanding rigorous standards for veterinary drug residue analysis, driven by global food safety regulations and the need for precise pharmacokinetic data. Patent CN101648917A introduces a groundbreaking methodology for the synthesis of 3-methyl-2-ethanolylquinoxaline-1,4-dioxide, a critical primary metabolite of the widely used veterinary antibacterial agent Mequindox. This innovation addresses a significant gap in the availability of high-purity reference standards, which are essential for establishing withdrawal periods and maximum residue limits in food-producing animals. The disclosed process utilizes a controlled reduction reaction followed by advanced recrystallization techniques to achieve purity levels exceeding 99%, ensuring that the synthesized compound is chemically identical to the metabolite found in vivo. For research directors and procurement specialists, this patent represents a pivotal shift from unreliable biological extraction to a robust, scalable chemical synthesis that guarantees consistency and supply continuity for essential veterinary drug intermediates.
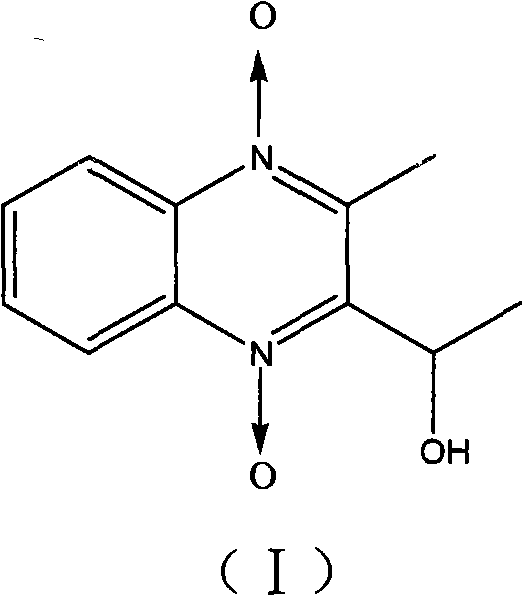
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, obtaining metabolites like 3-methyl-2-ethanolylquinoxaline-1,4-dioxide for research purposes relied heavily on extraction from biological samples or non-optimized synthetic routes that suffered from severe inefficiencies. Biological extraction is inherently limited by low yields, complex matrix interferences, and the inability to produce the quantities required for large-scale toxicological or residue studies. Furthermore, conventional chemical synthesis methods often lacked precise control over the reduction of the carbonyl group, leading to mixtures of by-products that compromised the purity of the final standard. These impurities could skew analytical results in high-performance liquid chromatography (HPLC) and mass spectrometry, leading to inaccurate pharmacokinetic modeling and potential regulatory non-compliance. The reliance on such inconsistent sources created a bottleneck for pharmaceutical companies aiming to validate residue detection methods or conduct comprehensive safety assessments for quinoxaline derivatives in the global market.
The Novel Approach
The methodology outlined in patent CN101648917A offers a transformative solution by employing a targeted reduction strategy that converts Mequindox directly into its metabolite with high selectivity and yield. By utilizing specific reducing agents such as sodium borohydride or sodium dithionite in common solvents like methanol, the process avoids the need for exotic catalysts or extreme reaction conditions that typically drive up costs and safety risks. The innovation lies in the rigorous monitoring of the reaction endpoint using HPLC, ensuring that the starting material is completely consumed before proceeding to purification. This is followed by a multi-step recrystallization process using ethanol, which effectively removes trace impurities and isomers. This approach not only simplifies the operational workflow but also ensures that the final product meets the stringent purity requirements necessary for use as a certified reference material in regulatory testing laboratories worldwide.
Mechanistic Insights into Carbonyl Reduction and Purification
The core of this synthesis lies in the selective reduction of the acetyl group on the quinoxaline ring to a hydroxyethyl group, a transformation that requires precise control to prevent over-reduction or degradation of the N-oxide functionalities. The reaction mechanism involves the nucleophilic attack of hydride ions from the reducing agent on the carbonyl carbon, facilitated by the solvent system which stabilizes the transition state. Maintaining the reaction temperature between 30°C and 40°C is critical, as higher temperatures could lead to side reactions affecting the quinoxaline dioxide core, while lower temperatures might result in incomplete conversion. The use of HPLC for real-time monitoring allows chemists to quench the reaction at the exact moment the Mequindox peak disappears, maximizing the yield of the desired metabolite while minimizing the formation of degradation products. This level of control is essential for maintaining the structural integrity of the molecule, which is vital for its function as a metabolic marker.
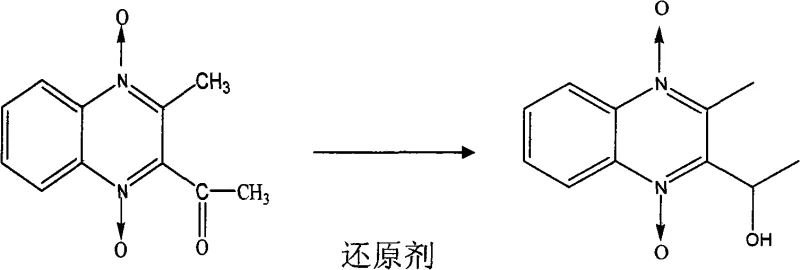
Impurity control is further enhanced through a sophisticated workup procedure that combines organic solvent extraction with repeated recrystallization. After the reduction is complete, the product is extracted into ethyl acetate, separating it from inorganic salts and water-soluble by-products. The subsequent drying and evaporation steps concentrate the crude material, which is then subjected to recrystallization in 95% ethanol. This purification step exploits the differential solubility of the target compound versus impurities at varying temperatures, effectively filtering out structurally similar analogues. The result is a pale yellow needle-like crystal with a purity profile that matches the in vivo metabolite as confirmed by UV and mass spectrometry data. This rigorous purification protocol ensures that the chemical standard is free from contaminants that could interfere with sensitive analytical instruments used in residue monitoring.
How to Synthesize 3-Methyl-2-Ethanolylquinoxaline-1,4-Dioxide Efficiently
Implementing this synthesis route requires adherence to strict operational parameters to ensure reproducibility and safety at scale. The process begins with the dissolution of Mequindox in methanol, followed by the controlled addition of the reducing agent under constant stirring. It is imperative to maintain the specified temperature range and sampling frequency to capture the optimal reaction endpoint. Once the reduction is confirmed via chromatography, the workup involves liquid-liquid extraction and solvent removal before the critical recrystallization phase. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and drying conditions, are outlined in the technical guide below to assist process engineers in replicating this high-yield pathway.
- Dissolve Mequindox in a suitable solvent such as methanol and initiate reduction using sodium borohydride at controlled temperatures between 30°C and 40°C.
- Monitor the reaction progress via HPLC analysis every 5 minutes until the starting material peak disappears completely to ensure full conversion.
- Extract the product with ethyl acetate, dry the organic phase, and perform multiple recrystallization cycles using 95% ethanol to achieve over 99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits regarding cost stability and supply reliability. Unlike biological extraction methods which are constrained by animal availability and ethical regulations, this chemical synthesis can be scaled indefinitely using readily available industrial raw materials. The reliance on common solvents such as methanol, ethyl acetate, and ethanol eliminates the need for specialized or hazardous reagents, simplifying logistics and reducing storage costs. Furthermore, the high yield and purity of the process minimize waste generation and the need for re-processing, leading to a more efficient use of resources. This operational efficiency translates into a more resilient supply chain capable of meeting the fluctuating demands of the veterinary pharmaceutical sector without the risk of shortages or quality deviations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of commodity chemicals significantly lower the direct material costs associated with production. By avoiding complex purification technologies like preparative HPLC and relying instead on standard recrystallization, the capital expenditure for manufacturing equipment is drastically reduced. This cost-effective approach allows for competitive pricing structures while maintaining high margins, making it an attractive option for large-scale commercial production of veterinary drug intermediates. The simplified process flow also reduces labor hours and energy consumption, contributing to overall operational expenditure savings.
- Enhanced Supply Chain Reliability: The use of stable, non-perishable raw materials ensures that production is not subject to the seasonal or biological constraints often associated with natural product extraction. This stability allows for better inventory planning and the ability to maintain safety stock levels to buffer against market volatility. Additionally, the robustness of the chemical process means that production can be easily transferred between different manufacturing sites without significant re-validation, providing flexibility in sourcing and reducing the risk of supply disruptions due to site-specific issues. This reliability is crucial for maintaining continuous operations in downstream analytical and research applications.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are standard in the fine chemical industry, such as stirred tank reactors and filtration systems. This facilitates a smooth transition from laboratory scale to multi-ton production without the need for process re-engineering. Moreover, the use of recyclable solvents and the generation of minimal hazardous waste align with modern environmental, health, and safety (EHS) standards. This compliance reduces the regulatory burden and disposal costs, ensuring that the manufacturing process remains sustainable and socially responsible in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this metabolite standard. These answers are derived directly from the patent specifications and are intended to clarify the feasibility and benefits of this synthesis route for potential partners. Understanding these details is essential for making informed decisions about integrating this intermediate into your research or supply chain.
Q: Why is synthetic 3-Methyl-2-Ethanolylquinoxaline-1,4-Dioxide preferred over biological extraction?
A: Synthetic routes provide significantly higher purity levels exceeding 99%, eliminating complex biological matrix interferences found in extraction methods, which is critical for accurate residue standard calibration.
Q: What are the scalability advantages of this reduction method?
A: The process utilizes common industrial solvents like methanol and ethyl acetate and avoids expensive transition metal catalysts, allowing for straightforward scale-up from laboratory to commercial tonnage without specialized equipment.
Q: How does this method ensure compliance with international residue testing standards?
A: By producing a chemically identical reference standard with verified mass spectrometry and UV profiles, this method supports the establishment of precise Maximum Residue Limits (MRLs) required by global regulatory bodies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methyl-2-Ethanolylquinoxaline-1,4-Dioxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of safe and effective veterinary medicines. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We adhere to stringent purity specifications and operate rigorous QC labs to verify that every batch of 3-methyl-2-ethanolylquinoxaline-1,4-dioxide meets the exacting standards required for regulatory submissions and research applications. Our commitment to technical excellence ensures that you receive a product that is consistent, reliable, and fully documented for your compliance needs.
We invite you to collaborate with us to optimize your supply chain and reduce your overall procurement costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume and purity requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can support your long-term strategic goals in the veterinary pharmaceutical sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
