Advanced Manufacturing of Fused Imidazopyridine Derivatives: Novel Low-Temperature Process and Stable Crystal Forms
The pharmaceutical industry constantly seeks robust synthetic routes for complex heterocyclic scaffolds that serve as critical building blocks for Central Nervous System (CNS) therapeutics. Patent CN1419557A introduces a significant technological advancement in the production of fused imidazopyridine derivatives, specifically detailing a novel process for synthesizing 2-(3-isoxazolyl)-3,6,7,9-tetrahydroimidazo[4,5-d]pyrano[4,3-b]pyridine and its phosphate salts. This innovation addresses long-standing challenges in thermal stability and process safety by replacing harsh high-temperature cyclization conditions with a milder, sulfinic acid salt-mediated pathway. The patent further discloses a novel prism crystal form of the phosphate monohydrate, which offers distinct physical advantages over previously known needle-like polymorphs. For R&D teams and supply chain managers, this represents a pivotal opportunity to optimize the manufacturing of high-value neurological agents.
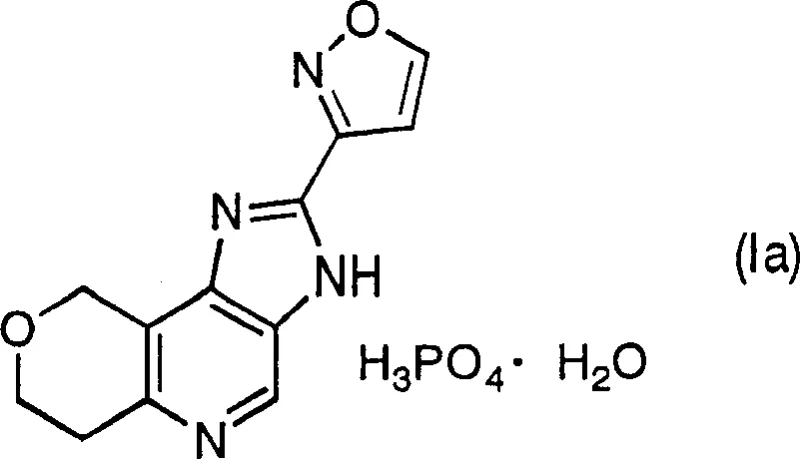
The core breakthrough lies in the structural integrity and stability of the final product, as depicted in the chemical structure above. This specific phosphate monohydrate configuration is not merely a salt formation but a carefully engineered crystal lattice that resists degradation. By securing a reliable pharmaceutical intermediate supplier capable of executing this specific crystallization protocol, manufacturers can ensure batch-to-batch consistency that is critical for regulatory approval. The transition from unstable needle-like crystals to robust prism crystals fundamentally alters the storage and handling logistics, reducing the risk of potency loss during long-term warehousing or transport in varying climatic zones.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for constructing the fused imidazopyridine core, such as those described in JP 1993/286973A, relied heavily on extreme thermal conditions to drive the cyclization reaction to completion. These legacy processes typically required reaction temperatures ranging from 150°C to 250°C, often utilizing high-boiling solvents like diphenyl ether or N-methylpyrrolidone without effective catalytic acceleration. Such aggressive thermal environments pose severe risks for industrial scale-up, including the potential for runaway exotherms, significant energy consumption, and the formation of complex impurity profiles due to thermal decomposition of sensitive heterocyclic rings. Furthermore, the isolation of the product from these high-temperature mixtures often resulted in the formation of needle-like crystals, which are notoriously difficult to filter and wash efficiently, leading to prolonged processing times and solvent retention issues that compromise overall yield and purity.
The Novel Approach
In stark contrast, the methodology outlined in CN1419557A utilizes a sulfinic acid salt, such as sodium p-toluenesulfinate, acting as a specialized reagent to facilitate the cyclization at drastically reduced temperatures. By operating at temperatures equal to or less than 120°C, and optimally around 100°C, this new route mitigates the thermal stress on the molecular framework. The addition of an acid or an organic base salt further refines the reaction environment, promoting cleaner conversion and minimizing side reactions. This shift enables cost reduction in pharmaceutical intermediate manufacturing by lowering energy demands and extending the lifespan of reactor equipment. Moreover, the process allows for the direct generation of the thermodynamically stable prism crystal form upon recrystallization from dilute phosphoric acid, bypassing the need for complex polymorph screening or post-synthesis stabilization treatments that are often required with the older, less controlled methods.
Mechanistic Insights into Sulfinic Acid Salt-Mediated Cyclization
The mechanistic elegance of this process centers on the role of the sulfinic acid salt as a nucleophilic catalyst or promoter that activates the halogenated precursor (Compound II) towards intramolecular cyclization. In traditional nucleophilic aromatic substitution or condensation reactions involving halogenated pyridines, high activation energy is typically required to displace the halogen atom. However, the presence of the sulfinic acid species likely generates a transient reactive intermediate that lowers this energy barrier, allowing the nitrogen atom of the imidazole ring to attack the electrophilic center of the pyridine moiety under mild conditions. This catalytic cycle avoids the need for stoichiometric amounts of strong bases or expensive transition metal catalysts, which can introduce heavy metal impurities that are difficult to remove to ppm levels required for pharmaceutical grades. The reaction proceeds smoothly in polar aprotic solvents like DMF or NMP, ensuring homogeneous mixing and efficient heat transfer throughout the reaction mass.
Impurity control is inherently built into this mechanism through the moderation of reaction kinetics. At lower temperatures (e.g., 100°C versus 200°C), the rate of degradation pathways for the isoxazole and dihydropyran rings is significantly suppressed. The isoxazole ring, in particular, can be susceptible to thermal ring-opening or rearrangement under harsh conditions; the mild protocol preserves this pharmacophore intact. Additionally, the subsequent crystallization step acts as a powerful purification engine. The specific lattice energy of the prism crystal form of the phosphate monohydrate selectively incorporates the target molecule while excluding structurally similar byproducts and unreacted starting materials. This phenomenon ensures that the final high-purity pharmaceutical intermediate meets stringent specifications without requiring multiple recrystallization cycles, thereby streamlining the downstream processing workflow and reducing solvent waste generation.
How to Synthesize 2-(3-isoxazolyl)-3,6,7,9-tetrahydroimidazopyranopyridine Efficiently
The synthesis of this complex fused heterocycle requires precise control over reaction parameters and crystallization dynamics to achieve the desired prism polymorph. The patent provides a clear roadmap for transitioning from the halogenated precursor to the final stable phosphate salt, emphasizing the importance of acid concentration and temperature gradients during the isolation phase. Operators must adhere to strict stoichiometric ratios of the sulfinic acid salt and phosphoric acid to ensure complete conversion and optimal crystal habit formation. The following guide outlines the critical operational phases derived from the patent examples, serving as a foundational reference for process engineers aiming to implement this technology.
- React the halogenated precursor (Compound II) with a sulfinic acid salt (e.g., sodium p-toluenesulfinate) in a polar aprotic solvent like DMF or NMP.
- Conduct the cyclization reaction at temperatures equal to or less than 120°C, preferably around 100°C, in the presence of an acid or organic base salt.
- Isolate the crude product and recrystallize from a dilute phosphoric acid aqueous solution to obtain the thermally stable prism crystal form of the phosphate monohydrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this novel synthetic route translates into tangible operational efficiencies and risk mitigation strategies. The shift away from extreme thermal conditions reduces the dependency on specialized high-temperature reactor vessels, allowing for the utilization of standard glass-lined or stainless steel equipment that is more readily available in the global CDMO network. This flexibility enhances supply chain resilience by broadening the pool of qualified manufacturing partners capable of producing the material. Furthermore, the improved filterability of the prism crystals directly impacts production throughput, as faster filtration and drying times reduce the overall cycle time per batch. This efficiency gain allows for increased production capacity without significant capital expenditure on new infrastructure, effectively de-risking the supply of this critical intermediate for downstream API manufacturing.
- Cost Reduction in Manufacturing: The elimination of high-temperature heating requirements results in substantial energy savings, as maintaining reactors at 100°C consumes significantly less utility resources than operating at 200°C or higher. Additionally, the avoidance of expensive transition metal catalysts removes the need for costly scavenging steps and rigorous heavy metal testing, which are major cost drivers in modern pharmaceutical production. The simplified workup procedure, driven by the superior physical properties of the prism crystals, further reduces labor costs and solvent consumption, contributing to a leaner and more economically viable manufacturing model that aligns with continuous improvement goals.
- Enhanced Supply Chain Reliability: The robust nature of the prism crystal form ensures that the material remains stable during extended storage and international shipping, minimizing the risk of quality deviations upon arrival at the customer's site. This stability reduces the likelihood of batch rejections and the need for expedited replacement shipments, which can disrupt production schedules. By securing a source of this intermediate produced via the patented low-temperature method, buyers can establish a more predictable inventory management system, knowing that the material's shelf life is maximized and its physical handling characteristics are optimized for automated dosing and processing equipment.
- Scalability and Environmental Compliance: The milder reaction conditions inherently generate fewer thermal degradation byproducts, simplifying the treatment of mother liquors and waste streams. This reduction in complex organic waste facilitates compliance with increasingly stringent environmental regulations regarding solvent discharge and hazardous waste disposal. The process is highly amenable to commercial scale-up of complex pharmaceutical intermediates, as the heat removal requirements are manageable even in large-volume reactors, preventing hot spots that could lead to safety incidents. This scalability ensures that supply can be rapidly ramped up to meet market demand for the final therapeutic agent without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this fused imidazopyridine derivative. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing clarity on the material's performance profile. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing drug development pipelines.
Q: What are the stability advantages of the prism crystal form over the needle-like form?
A: The prism crystal form of the phosphate monohydrate exhibits superior thermal and light stability compared to the needle-like form. Experimental data indicates that the prism crystals maintain their appearance and potency under accelerated stability conditions (40°C/75% RH) for extended periods, whereas needle-like crystals tend to discolor and degrade.
Q: How does the new sulfinic acid salt method improve industrial scalability?
A: The novel process operates at significantly lower temperatures (≤120°C) compared to conventional methods requiring 150-250°C. This reduction in thermal stress minimizes side reactions, simplifies reactor cooling requirements, and enhances safety profiles, making large-scale commercial production more feasible and cost-efficient.
Q: Is this intermediate suitable for CNS active pharmaceutical ingredients?
A: Yes, the fused imidazopyridine derivatives produced via this method are known intermediates for compounds with anxiolytic, narcotic antagonist, and brain function activator properties. The high purity and stable crystal form ensure consistent quality for downstream API synthesis targeting the central nervous system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(3-isoxazolyl)-3,6,7,9-tetrahydroimidazopyranopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation CNS therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to full-scale manufacturing is seamless and efficient. We are committed to delivering materials with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify crystal form, particle size distribution, and impurity profiles. Our facility is equipped to handle the specific solvent systems and crystallization protocols required for this patented process, guaranteeing a consistent supply of the stable prism crystal form.
We invite you to collaborate with us to leverage this advanced synthetic technology for your project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can enhance your project's economic viability. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey from discovery to commercial success with reliable, high-performance chemical solutions.
