Advanced Palladium-Catalyzed Carbonylation Process for Commercial-Scale Secondary Tertiary Amide Production
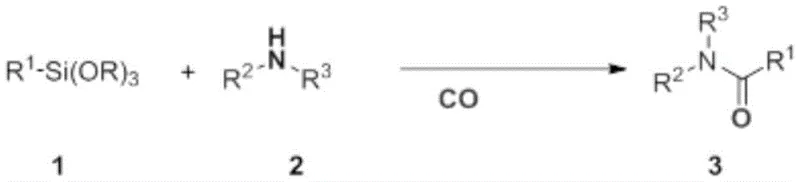
The patented methodology disclosed in CN109180518B represents a paradigm shift in secondary and tertiary amide synthesis, addressing critical limitations in traditional pharmaceutical intermediate manufacturing. This innovative carbonylation process utilizes aryl silanes as nucleophilic reagents under precisely controlled carbon monoxide pressure, achieving remarkable efficiency without requiring inert gas protection—a significant departure from conventional methods that demand stringent atmospheric controls. The reaction demonstrates exceptional atom economy through direct CO insertion into amine-silane bonds, enabling the production of structurally diverse amide compounds essential for drug development pipelines. With optimized conditions yielding up to 92% product purity, this approach eliminates multiple purification steps while maintaining compatibility with complex molecular architectures found in modern therapeutics. The methodology's robustness across varied substrates positions it as a transformative solution for pharmaceutical manufacturers seeking reliable, high-yield intermediate production without compromising on quality or scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional amide synthesis routes suffer from multiple critical constraints that impede efficient pharmaceutical intermediate production, including harsh reaction conditions requiring strong acids or elevated temperatures that degrade sensitive functional groups common in drug molecules. Conventional carbonylation methods necessitate high-pressure CO insertion systems operating above 5 MPa with mandatory inert gas shielding, creating significant safety hazards and infrastructure costs that limit accessibility for mid-sized manufacturers. The reliance on carboxylic acid derivatives often generates stoichiometric waste streams requiring extensive neutralization and disposal procedures, while halogenated hydrocarbon-based approaches introduce toxic byproducts that complicate regulatory compliance for pharmaceutical applications. Furthermore, these processes exhibit narrow substrate tolerance—particularly with sterically hindered amines—resulting in inconsistent yields and challenging purification requirements that increase production timelines by weeks for complex molecules. The cumulative effect of these limitations manifests as unreliable supply chains and elevated costs that directly impact drug development economics.
The Novel Approach
The patented methodology overcomes these constraints through a fundamentally redesigned catalytic system that operates under mild conditions (60–180°C, 0.1–5.0 MPa CO pressure) without inert gas requirements, leveraging aryl silanes as stable nucleophilic partners that react efficiently with amines under palladium catalysis. This innovation eliminates high-pressure CO insertion systems while maintaining exceptional substrate versatility across primary, secondary, and tertiary amines with diverse aromatic and aliphatic structures. The reaction's inherent atom economy minimizes waste generation by directly incorporating CO into the amide bond without intermediate activation steps, significantly reducing environmental impact compared to traditional condensation methods. Crucially, the process demonstrates consistent high yields (up to 92%) across structurally complex substrates including heterocyclic amines and sterically hindered compounds that typically fail in conventional systems, providing pharmaceutical manufacturers with unprecedented flexibility in intermediate design and production planning.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle initiates with oxidative addition of palladium(0) into the Si–O bond of aryl silane, forming a key palladium-silyl intermediate that facilitates transmetalation with the amine substrate. This step generates a palladium-amido species that undergoes migratory insertion with carbon monoxide to form an acyl-palladium complex—the critical intermediate enabling direct amide bond formation. Subsequent reductive elimination releases the secondary/tertiary amide product while regenerating the active palladium catalyst, completing the cycle with exceptional efficiency due to the synergistic role of copper additives that prevent palladium aggregation and maintain catalytic activity throughout the reaction. The absence of strong acid or base requirements preserves sensitive functional groups commonly found in pharmaceutical intermediates, while the controlled CO pressure environment prevents over-carbonylation side reactions that typically generate impurities in conventional processes.
Impurity control is achieved through precise regulation of the copper additive concentration (amine:catalyst:additive = 10:1:5–10:1:20), which suppresses undesired homocoupling reactions by modulating palladium oxidation states during the catalytic cycle. The solvent system—comprising polar aprotic options like DMF or NMP—facilitates optimal solubility of both organic substrates and inorganic additives while enabling straightforward product separation through standard extraction techniques. This dual mechanism of catalytic precision and simplified workup ensures consistent production of high-purity intermediates meeting stringent pharmaceutical specifications, with minimal residual metals or organic impurities that could compromise downstream drug synthesis.
How to Synthesize Secondary Tertiary Amides Efficiently
This patented synthesis route represents a significant advancement over conventional amide formation techniques by eliminating multiple processing steps while maintaining exceptional product quality. The methodology leverages readily available aryl silanes and amines as starting materials, operating under conditions compatible with standard manufacturing equipment without requiring specialized high-pressure reactors or inert atmosphere systems. Detailed standardized synthesis procedures have been developed to ensure consistent implementation across different production scales, with specific parameters optimized for various substrate combinations to maximize yield and purity while minimizing processing time. The following section provides the essential operational guidelines for successful implementation of this innovative process.
- Precisely mix aryl silane (R¹-Si(OR)₃), amine compound (R²-NH-R³), palladium catalyst, and copper additive in solvent at controlled molar ratios (amine: silane = 1.0:0.5–1.0:10.0; amine:catalyst:additive = 10:1:5–10:1:20) to achieve optimal reaction kinetics.
- Heat the mixture under carbon monoxide pressure (0.1–5.0 MPa) at 60–180°C for 2–48 hours with continuous stirring, maintaining amine concentration at 0.1–0.2 mol/L without inert gas shielding.
- Purify the reaction mixture through ethyl acetate extraction and silica gel column chromatography to isolate high-purity secondary/tertiary amide products with minimal impurities.
Commercial Advantages for Procurement and Supply Chain Teams
This methodology delivers substantial strategic value for procurement and supply chain operations by addressing fundamental pain points in pharmaceutical intermediate sourcing through inherent process improvements rather than incremental optimizations. The elimination of inert gas protection requirements reduces both capital expenditure for specialized equipment and ongoing operational costs associated with gas handling systems, while the compatibility with standard solvents streamlines material sourcing logistics across global manufacturing networks. These advantages translate directly into enhanced supply chain resilience and cost efficiency without requiring significant infrastructure investments or process revalidation efforts.
- Cost Reduction in Manufacturing: The removal of high-pressure CO insertion systems and inert gas shielding requirements substantially lowers capital expenditure while reducing energy consumption during production cycles; simplified workup procedures using standard extraction solvents minimize solvent disposal costs and eliminate expenses associated with specialized waste treatment systems required by conventional methods; the high atom economy prevents raw material waste that typically necessitates additional purification steps in traditional amide synthesis.
- Enhanced Supply Chain Reliability: The use of stable, commercially available aryl silanes and amines eliminates dependency on hazardous or restricted reagents that often cause supply chain disruptions; consistent performance across diverse substrates enables single-source procurement strategies for multiple intermediate types; simplified process parameters facilitate rapid technology transfer between manufacturing sites without extensive revalidation.
- Scalability and Environmental Compliance: The absence of extreme pressure or temperature requirements enables seamless scale-up from laboratory to commercial production using existing reactor infrastructure; reduced waste generation through direct carbonylation aligns with green chemistry principles while lowering environmental compliance costs; consistent high yields across batch sizes ensure reliable delivery schedules even during demand surges.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations based on detailed analysis of the patented methodology's implementation parameters and performance data across multiple substrate classes. These insights derive directly from the experimental results documented in CN109180518B, providing evidence-based responses to common concerns regarding process adoption and integration into existing manufacturing workflows.
Q: How does this method eliminate inert gas protection requirements compared to conventional amide synthesis?
A: The reaction system operates under controlled CO pressure without oxygen-sensitive intermediates, eliminating the need for inert gas shielding. This simplifies reactor setup and reduces operational complexity while maintaining high atom economy.
Q: What enables the high yield (92%) and mild reaction conditions in this process?
A: The synergistic effect of palladium catalysts with copper additives facilitates efficient CO insertion at lower temperatures (60–180°C) and pressures (0.1–5.0 MPa), while the aryl silane nucleophile provides stable reaction pathways that minimize side products.
Q: How does this process support scalable commercial production of pharmaceutical intermediates?
A: The absence of high-pressure CO insertion requirements and compatibility with standard solvents enables straightforward scale-up from laboratory to industrial reactors, with demonstrated consistency across diverse substrate combinations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Secondary Tertiary Amide Supplier
Our company leverages this patented technology to deliver exceptional value in pharmaceutical intermediate manufacturing through extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through advanced QC labs equipped with state-of-the-art analytical instrumentation. We specialize in transforming complex synthetic routes into robust commercial processes that meet global regulatory standards while optimizing cost structures through continuous process refinement and raw material sourcing strategies tailored to each client's specific requirements.
Engage our technical procurement team today to request a Customized Cost-Saving Analysis demonstrating how this innovative methodology can enhance your supply chain resilience; we will provide specific COA data and route feasibility assessments to validate implementation timelines and quality metrics for your target molecules.
