Advanced One-Pot Synthesis of Distamycin Derivatives for Commercial Scale-Up and High Purity
Advanced One-Pot Synthesis of Distamycin Derivatives for Commercial Scale-Up and High Purity
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing complex antitumor agents, and patent CN1190418C presents a groundbreaking methodology for the preparation of distamycin derivatives. This specific intellectual property outlines a robust chemical process designed to produce acryloyl-distamycin-guanidine derivatives, such as the potent antineoplastic agent PNU166196, with exceptional yield and purity. Unlike traditional synthetic routes that often suffer from low efficiency due to the necessity of isolating numerous unstable intermediates, this invention leverages a telescoped approach. By integrating condensation and reduction steps into a continuous flow or one-pot sequence, the process minimizes the generation of impurities and drastically simplifies the operational workflow. For R&D directors and procurement specialists alike, understanding this technology is crucial as it represents a significant leap forward in the manufacturing of high-value pharmaceutical intermediates.
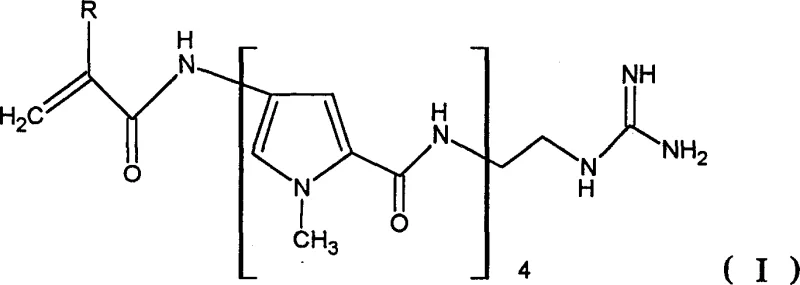
The core innovation lies in the ability to construct the polypyrrolamide backbone without interrupting the reaction sequence for purification. The target molecules, characterized by their specific interaction with DNA-AT sequences, are critical for interfering with tumor replication and transcription processes. The patent explicitly details how the formyl group of natural distamycin is replaced by an acryloyl group and the amidine group is substituted with nitrogen-containing terminal groups like guanidine. This structural modification enhances biological activity but traditionally posed significant synthetic challenges. The disclosed method overcomes these hurdles by utilizing activated carboxylic acid derivatives that react seamlessly with the guanidine-bearing backbone. This ensures that the final product meets the stringent quality standards required for clinical applications while maintaining a commercially viable production profile.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex pyrrole antibiotics like distamycin derivatives has been plagued by inefficiencies inherent in stepwise construction. Conventional methods, such as those referenced in prior art like WO98/04524, typically require the isolation of every intermediate species along the synthetic pathway. This step-by-step isolation is not only labor-intensive but also introduces significant risks of product degradation and contamination. Each purification step, often involving chromatography or recrystallization, results in material loss, thereby depressing the overall yield. Furthermore, the intermediates involved, particularly those containing reactive nitro or amino groups on the pyrrole ring, can be chemically unstable. Prolonged exposure to air or moisture during isolation can lead to oxidation or hydrolysis, generating difficult-to-remove impurities that compromise the safety profile of the final API. These factors collectively drive up the cost of goods sold and extend the lead time for manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in CN1190418C revolutionizes the synthesis by enabling a continuous, telescoped reaction sequence. The process allows for the direct conversion of starting materials into the final distamycin derivative through a series of in-situ transformations. Specifically, the method involves reacting 2-aminoethylguanidine with a nitropyrrole carboxylic acid derivative, followed immediately by reduction and further condensation without isolating the intermediate nitro or amino compounds. This strategy effectively bypasses the bottlenecks associated with traditional isolation. By keeping the reactive intermediates in solution, the process maintains their stability and directs the reaction equilibrium towards the desired product. The result is a streamlined workflow that significantly reduces solvent usage and waste generation. This efficiency translates directly into a more sustainable and cost-effective manufacturing protocol, making it an attractive option for large-scale industrial production.
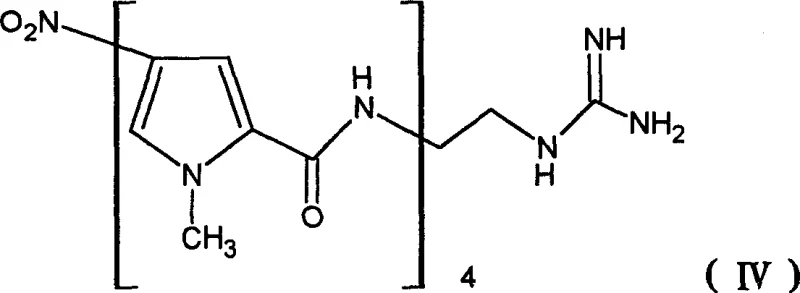
Mechanistic Insights into Telescoped Amide Condensation and Catalytic Hydrogenation
The chemical elegance of this process is rooted in its precise control over amide bond formation and nitro group reduction. The synthesis initiates with the nucleophilic attack of the amino group of 2-aminoethylguanidine on the activated carbonyl of the pyrrole derivative. This reaction is conducted under mild alkaline conditions, typically using bases like sodium carbonate or bicarbonate in solvents such as dioxane or THF. The choice of base and solvent is critical to ensure that the guanidine moiety remains intact while facilitating the coupling reaction. Following the initial coupling, the nitro group on the pyrrole ring is reduced to an amino group using catalytic hydrogenation. The patent highlights the use of heterogeneous catalysts like palladium on carbon (Pd/C) under hydrogen pressures ranging from 1 bar to 10 bar. This reduction step is highly selective, converting the nitro functionality without affecting other sensitive groups in the molecule.
Impurity control is inherently built into this mechanistic design. By avoiding the isolation of the amino-intermediate, the process prevents potential side reactions such as oxidative dimerization or reaction with atmospheric CO2. The subsequent condensation with another equivalent of the activated pyrrole acid chloride proceeds rapidly with the freshly generated amine. This 'generate-and-consume' strategy ensures that the concentration of the reactive amine remains low, minimizing self-condensation or polymerization side reactions. Finally, the terminal acylation with an alpha-haloacrylic acid introduces the pharmacologically active acryloyl warhead. The use of coupling agents like EDC or DDC in the final step ensures high conversion rates. The cumulative effect of these mechanistic choices is a product with an HPLC purity exceeding 98%, as demonstrated in the patent examples, which is essential for meeting regulatory requirements for antitumor drugs.
How to Synthesize Acryloyl-Distamycin-Guanidine Derivatives Efficiently
The synthesis of these high-value intermediates requires a deep understanding of reaction kinetics and thermodynamic control to maximize yield and purity. The patented route offers a standardized framework that can be adapted for various scales of production, from laboratory benchtop to industrial reactors. The key to success lies in the careful management of reaction conditions, particularly temperature and pH, during the telescoped steps. Maintaining the reaction mixture within the optimal range of 0°C to 50°C ensures that the exothermic condensation reactions proceed smoothly without thermal degradation of the sensitive pyrrole rings. Furthermore, the selection of appropriate solvents that can dissolve both the organic starting materials and the inorganic bases is vital for homogeneous reaction progress. Detailed standard operating procedures for executing this synthesis are provided below to guide technical teams in implementing this advanced methodology.
- React 2-aminoethylguanidine with a nitropyrrole carboxylic acid derivative under alkaline conditions to form the initial amide bond.
- Perform catalytic hydrogenation using Pd/C to reduce the nitro group to an amino group without isolating the intermediate.
- Conduct subsequent condensation with additional pyrrole units and final acylation with an alpha-haloacrylic acid to yield the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this streamlined synthesis process offers tangible strategic benefits beyond mere technical superiority. The elimination of multiple isolation and purification steps fundamentally alters the cost structure of manufacturing these complex intermediates. By reducing the number of unit operations, the process lowers the demand for labor, equipment time, and utilities. Moreover, the ability to recover and reuse solvents like dioxane and THF throughout the telescoped sequence contributes to substantial cost savings and aligns with green chemistry principles. This efficiency makes the supply of these critical antitumor intermediates more resilient against market fluctuations and raw material shortages. Companies adopting this technology can secure a more stable supply chain, ensuring continuity of production for downstream API manufacturing.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the drastic simplification of the workflow. Traditional methods involve multiple work-ups, extractions, and drying steps for each intermediate, each adding to the operational expense. By telescoping these steps, the new method eliminates the need for intermediate storage and handling, which are significant cost centers in fine chemical manufacturing. Additionally, the high yields reported in the patent examples mean that less raw material is wasted, directly improving the material cost efficiency. The reduction in solvent volume required per kilogram of product further decreases waste disposal costs, contributing to a leaner and more profitable production model without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain reliability is bolstered by the use of commercially available and stable starting materials. The patent notes that key reagents, such as the alpha-haloacrylic acids and the nitropyrrole derivatives, are either commercially available or easily prepared via known methods. This reduces the dependency on custom synthesis of exotic precursors, which can be a bottleneck in the supply chain. Furthermore, the robustness of the catalytic hydrogenation step using standard Pd/C catalysts ensures that the process is not reliant on scarce or specialized reagents. This accessibility of inputs allows for diversified sourcing strategies, mitigating the risk of supply disruptions and enabling faster response times to increased market demand for these oncology ingredients.
- Scalability and Environmental Compliance: Scalability is a critical factor for any process intended for commercial production, and this method excels in its adaptability from pilot to full-scale manufacturing. The reaction conditions, involving moderate temperatures and pressures, are well within the capabilities of standard stainless steel reactors found in most multipurpose chemical plants. The absence of cryogenic conditions or highly hazardous reagents simplifies the engineering controls required for scale-up. From an environmental perspective, the reduced solvent usage and waste generation facilitate easier compliance with increasingly stringent environmental regulations. The process generates less hazardous waste per unit of product, lowering the environmental footprint and reducing the liability associated with waste treatment and disposal, making it a sustainable choice for long-term production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this distamycin derivative synthesis process. These answers are derived directly from the technical specifications and experimental data provided in patent CN1190418C. They are designed to clarify the operational parameters and potential benefits for stakeholders evaluating this technology for integration into their manufacturing portfolios. Understanding these details is essential for making informed decisions about process adoption and resource allocation.
Q: What are the key advantages of the telescoped synthesis method described in CN1190418C?
A: The primary advantage is the elimination of multiple isolation steps for intermediates, which significantly reduces processing time, solvent consumption, and the formation of undesired by-products, leading to higher overall purity and yield.
Q: Which catalysts are preferred for the reduction steps in this distamycin derivative process?
A: The patent specifies the use of heterogeneous catalysts such as palladium on carbon (Pd/C) or platinum under hydrogen pressure ranging from 1 bar to 10 bar, ensuring efficient conversion of nitro groups to amino groups under mild conditions.
Q: How does this process impact the scalability of producing antitumor intermediates?
A: By utilizing common solvents like dioxane or THF and avoiding complex purification of unstable intermediates, the process is highly adaptable for commercial scale-up, allowing for consistent production of high-purity batches from kilogram to tonnage scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Distamycin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving antitumor therapies. Our team of expert chemists has extensively analyzed the methodology described in CN1190418C and possesses the technical capability to execute this complex synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the >98% HPLC purity targets outlined in the patent. We are committed to delivering distamycin derivatives that meet the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic route for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. By partnering with us, you gain access to our deep process knowledge and supply chain network. Please contact us today to request specific COA data and route feasibility assessments. Let us help you accelerate your path to market with a reliable, cost-effective, and high-quality supply of these essential pharmaceutical intermediates.
