Industrial Scale Synthesis of Ursodeoxycholic Acid from Phytosterol Derivatives for Global Pharma Supply Chains
Industrial Scale Synthesis of Ursodeoxycholic Acid from Phytosterol Derivatives for Global Pharma Supply Chains
The pharmaceutical industry is currently witnessing a significant paradigm shift in the production of hepatobiliary therapeutics, driven by the urgent need for sustainable and scalable manufacturing processes. Patent CN115181150A, published in October 2022, introduces a groundbreaking methodology for the synthesis of Ursodeoxycholic Acid (UDCA), a critical Active Pharmaceutical Ingredient (API) used extensively in the treatment of cholesterol gallstones and various liver diseases. Unlike historical methods that relied on the ethically contentious extraction from bear bile or chemically intensive routes utilizing toxic heavy metals, this invention leverages phytosterol degradation products as a renewable starting material. The disclosed process integrates a series of sophisticated organic transformations, including ketal protection, Grignard coupling, and a novel catalytic allylic oxidation, to achieve high-purity UDCA. For R&D directors and procurement strategists, this patent represents a vital opportunity to secure a reliable pharmaceutical intermediates supplier capable of delivering cost-effective and environmentally compliant solutions. By transitioning away from animal-derived sources and chromium-laden chemistry, manufacturers can mitigate regulatory risks while ensuring a continuous supply of this essential medicine.
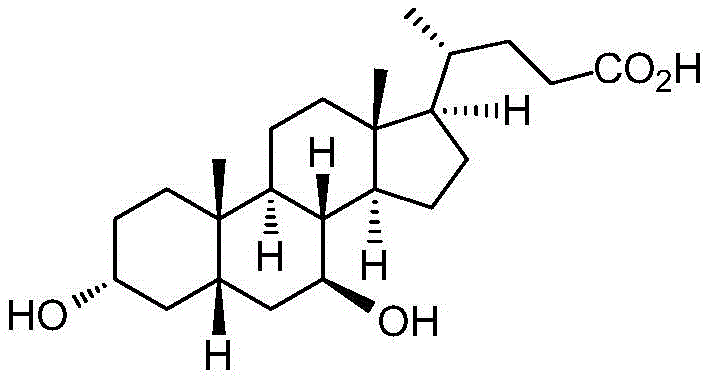
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Ursodeoxycholic Acid has been plagued by significant technical and ethical bottlenecks that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. Early methods involved the isolation of UDCA directly from black bear bile, a practice that is not only ethically problematic due to animal welfare concerns but also suffers from extremely low yields and inconsistent quality. On the chemical synthesis front, traditional routes starting from Cholic Acid (CA) or Hyocholic Acid (HCA) have relied heavily on stoichiometric amounts of chromium-based oxidants, such as chromium trioxide (CrO3) in acetic acid. As illustrated in prior art pathways, these processes generate massive quantities of toxic chromium waste, necessitating expensive and complex waste treatment protocols to meet modern environmental standards. Furthermore, certain legacy routes employ hazardous reagents like hydrazine hydrate at high temperatures or metallic sodium for reduction steps, which pose severe safety risks in a large-scale plant environment. The cumulative effect of these factors is a fragile supply chain characterized by high production costs, potential regulatory shutdowns, and difficulty in achieving the commercial scale-up of complex pharmaceutical intermediates required by the global market.
The Novel Approach
In stark contrast to these legacy methodologies, the invention disclosed in CN115181150A presents a streamlined and green synthetic strategy that fundamentally reimagines the construction of the UDCA molecule. The process initiates with UD-8, a readily available degradation product of phytosterols, thereby anchoring the supply chain in renewable biomass rather than limited animal sources or expensive bile acids. The core innovation lies in a meticulously designed sequence that avoids toxic heavy metals entirely. The route proceeds through ketal protection of the carbonyl group, followed by alcohol protection and a copper-catalyzed Grignard reaction to construct the necessary side chain. Crucially, the introduction of oxygen functionality is achieved via a catalytic allylic oxidation using N-hydroxyphthalimide (NHPI) and molecular air, a reaction that operates under mild conditions and produces water as the primary byproduct. This is followed by ketal deprotection, Pinnick oxidation to convert the terminal aldehyde to a carboxylic acid, and a final hydrogenation step. This holistic approach not only simplifies the operational workflow but also drastically reduces the environmental footprint, making it an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates while adhering to strict green chemistry principles.
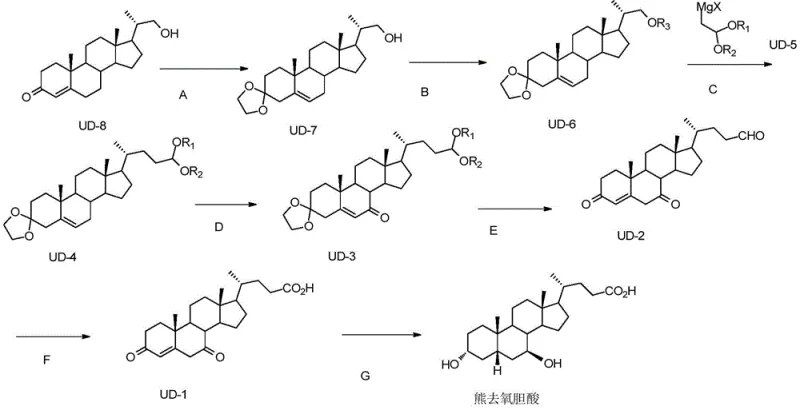
Mechanistic Insights into NHPI-Catalyzed Allylic Oxidation
From a technical perspective, the most distinguishing feature of this synthetic route is the implementation of an aerobic allylic oxidation catalyzed by N-hydroxyphthalimide (NHPI). In traditional steroid functionalization, introducing a ketone at the allylic position often requires harsh oxidants that lack selectivity, leading to over-oxidation or degradation of the sensitive steroid nucleus. The mechanism employed here utilizes the phthalimide N-oxyl (PINO) radical, generated in situ from NHPI by an initiator such as AIBN or benzoyl peroxide in the presence of oxygen. This radical species selectively abstracts an allylic hydrogen atom from the steroid substrate (UD-4.2), forming a resonance-stabilized allylic radical. Subsequent reaction with molecular oxygen and further propagation steps lead to the formation of the enone system found in intermediate UD-3.1. This catalytic cycle is highly efficient, operating effectively at moderate temperatures (30-80°C) and atmospheric pressure, which significantly lowers the energy input required for the reaction. For process chemists, this mechanism offers a robust pathway to install the critical 7-keto functionality without compromising the stereochemical integrity of the adjacent chiral centers, ensuring a high-quality impurity profile for the final API.
Furthermore, the strategic placement of protecting groups plays a pivotal role in controlling the impurity spectrum throughout the synthesis. The initial formation of the cyclic ketal at the C3 position protects the carbonyl group from unwanted nucleophilic attack during the subsequent Grignard addition, ensuring that the organometallic reagent reacts exclusively with the activated side-chain precursor. Similarly, the conversion of the primary alcohol to a sulfonate ester (mesylate or tosylate) activates the leaving group for the Grignard coupling while preventing elimination side reactions that could occur under basic conditions. The final deprotection steps are equally critical; the use of mild acidic conditions for ketal hydrolysis ensures that the newly formed ketone and the carboxylic acid functionalities remain intact. By avoiding strong bases or reducing agents like lithium in liquid ammonia until the very final stereoselective hydrogenation step, the process minimizes the formation of epimeric impurities. This rigorous control over reaction selectivity is essential for meeting the stringent purity specifications demanded by pharmacopeial standards for Ursodeoxycholic Acid.
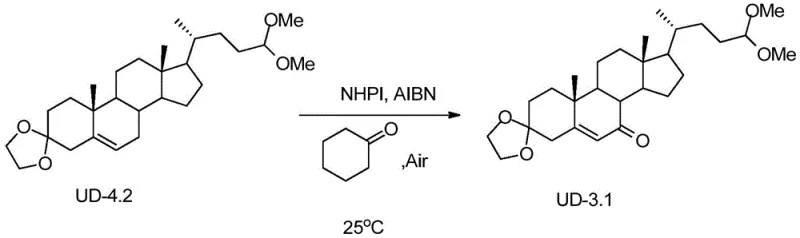
How to Synthesize Ursodeoxycholic Acid Efficiently
The execution of this synthetic route requires precise control over reaction parameters to maximize yield and minimize waste, particularly during the oxidation and coupling stages. The process begins with the protection of the starting material UD-8, where careful removal of water during ketal formation is essential to drive the equilibrium towards the protected product UD-7. Following activation of the side chain, the Grignard coupling must be performed under strictly anhydrous conditions to prevent quenching of the organometallic reagent. The subsequent oxidation step using NHPI and air is remarkably forgiving, allowing for operation at ambient pressure, which simplifies reactor requirements. Finally, the Pinnick oxidation provides a gentle method to convert the aldehyde to the acid without affecting the enone system, and the final hydrogenation over Raney Nickel establishes the correct 7-beta hydroxyl stereochemistry. For a detailed breakdown of the specific molar ratios, solvent choices, and workup procedures, please refer to the standardized protocol below.
- Protect the carbonyl group of starting material UD-8 using ethylene glycol and an acid catalyst to form ketal UD-7, followed by alcohol protection to generate sulfonate UD-6.
- Perform a copper-catalyzed Grignard reaction with reagent UD-5 to extend the side chain, yielding intermediate UD-4.
- Execute allylic oxidation using NHPI catalyst and air to introduce the ketone functionality, followed by ketal deprotection, Pinnick oxidation of the aldehyde, and final Raney-Ni hydrogenation to obtain UDCA.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this phytosterol-based synthesis route offers transformative economic and logistical benefits that directly impact the bottom line. The primary advantage stems from the complete elimination of hexavalent chromium reagents, which are not only expensive to purchase but also incur substantial costs for disposal and environmental compliance. By replacing these hazardous oxidants with catalytic aerobic oxidation and mild Pinnick conditions, manufacturers can significantly reduce the overhead associated with waste treatment facilities and regulatory reporting. Moreover, the reliance on phytosterol degradation products as the starting material decouples the supply chain from the volatile and ethically sensitive market for animal bile acids. Phytosterols are abundant byproducts of the vegetable oil industry, ensuring a stable, renewable, and cost-effective feedstock that is immune to the supply shocks often seen with animal-derived raw materials. This shift enhances supply chain resilience and allows for long-term contracting with greater price stability.
- Cost Reduction in Manufacturing: The economic implications of this new route are profound, primarily driven by the simplification of the downstream processing units. Traditional chromium-based oxidations generate large volumes of heavy metal sludge that require specialized filtration and neutralization steps, adding both time and capital expenditure to the production cycle. By utilizing a chromium-free pathway, the process eliminates the need for expensive heavy metal scavengers and the associated analytical testing for residual metals in the final API. Additionally, the use of molecular air as the terminal oxidant in the allylic oxidation step replaces costly stoichiometric oxidants, further driving down the variable cost of goods sold (COGS). The high yields reported in the patent examples suggest that less raw material is wasted, contributing to a more efficient utilization of resources and a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for pharmaceutical buyers, and this synthetic method addresses several key vulnerabilities inherent in older technologies. The starting material, derived from phytosterols, is sourced from the massive global vegetable oil industry, providing a buffer against shortages that might affect niche animal byproducts. Furthermore, the reaction conditions are notably mild, with key steps like the NHPI oxidation and Pinnick oxidation occurring at or near room temperature. This reduces the risk of thermal runaways and equipment failures that can lead to unplanned plant downtime. The robustness of the chemistry means that batch-to-batch variability is minimized, ensuring consistent delivery schedules and reducing the likelihood of failed batches that disrupt inventory levels. This reliability makes the manufacturer a more attractive partner for long-term supply agreements.
- Scalability and Environmental Compliance: As regulatory bodies worldwide tighten restrictions on pharmaceutical manufacturing emissions, the environmental profile of a synthesis route becomes a decisive factor in vendor selection. This process is inherently scalable because it avoids the use of pyrophoric reagents like metallic sodium or hazardous gases like hydrazine, which require specialized containment and handling infrastructure. The aerobic oxidation step is particularly amenable to scale-up, as it can be efficiently managed in standard stirred-tank reactors with simple air sparging. From a compliance standpoint, the absence of persistent organic pollutants and heavy metals simplifies the permitting process for new manufacturing lines and reduces the liability associated with environmental remediation. This alignment with green chemistry principles not only future-proofs the supply chain against stricter regulations but also enhances the brand value of the final pharmaceutical product among environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel UDCA synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy methods in terms of safety, efficiency, and environmental impact. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply contracts based on the superior attributes of this green manufacturing process.
Q: Why is the phytosterol-based route preferred over traditional animal extraction for UDCA?
A: Traditional extraction from bear bile is limited by ethical concerns, low yield, and regulatory restrictions. The phytosterol-based chemical synthesis described in CN115181150A offers a sustainable, scalable, and ethically compliant alternative with easily obtained raw materials.
Q: How does this new method address environmental concerns associated with previous synthetic routes?
A: Conventional methods often rely on toxic chromium reagents (like Jones reagent or PCC) and dangerous reducing agents like metallic sodium. This novel route utilizes catalytic allylic oxidation with air and mild Pinnick oxidation, significantly reducing heavy metal waste and operational hazards.
Q: What are the key technical advantages for large-scale manufacturing of this API intermediate?
A: The process features mild reaction conditions (e.g., 25°C for oxidation), high overall yields, and avoids complex purification steps associated with chromium salt removal. This translates to lower production costs and enhanced supply chain reliability for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ursodeoxycholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just a regulatory necessity but a strategic imperative for maintaining competitiveness in the global pharmaceutical market. Our team of expert process chemists has thoroughly analyzed the methodology disclosed in CN115181150A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative UDCA synthesis to life. We are committed to delivering stringent purity specifications through our rigorous QC labs, ensuring that every batch of Ursodeoxycholic Acid meets the highest international standards for safety and efficacy. Our state-of-the-art facilities are equipped to handle the specific unit operations required by this route, from anhydrous Grignard couplings to catalytic aerobic oxidations, guaranteeing a seamless transfer from laboratory bench to industrial reactor.
We invite forward-thinking pharmaceutical companies and procurement leaders to collaborate with us to leverage this advanced technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this chromium-free process for your organization. We encourage you to contact our technical procurement team today to request specific COA data from our pilot runs and comprehensive route feasibility assessments. Let us help you secure a sustainable, cost-effective, and reliable source of high-purity Ursodeoxycholic Acid that aligns with your corporate sustainability goals and commercial objectives.
